Abstract
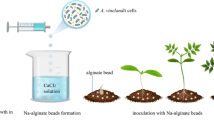
Immobilizarion of PGPR for agricultural applications aims to provide temporary physical protection from stressful environmental conditions and the gradual release of cells for successful root colonization, release the cells gradually. In this work, we immobilized Bradyrhizobium sp. SEMIA6144 or Azospirillum brasilense Az39 cells in 2% alginate beads prepared by ionic gelation process, and then stored up to 12 months at 4 °C. Alginate matrix showed interaction with the immobilized bacteria (FTIR), allowed a constant release of cells, and improved their viability and capability to interact with Arachis hypogaea. Cell number into beads reached 107 CFU.bead−1; however, viability decreased from 4 months of storage for Az39, while it was maintained up to 12 months for SEMIA6144, showing a low metabolic activity measured by the MTT assay. Adhesion of SEMIA6144 and Az39 from new beads to peanut root was 11.5% and 16%, respectively, higher than non-immobilized bacteria. Peanut inoculation with 12 months storage SEMIA6144 beads significantly increased root length and biomass at 30 days of growth, and under restrictive water condition (RWC), nodulation and total plant N content increased compared with liquid inoculation. Our results demonstrate that immobilization of SEMIA6144 and Az39 in alginate matrix is a potential alternative to enhance peanut growth even under RWC.
Key points
• Alginate encapsulation enhances viability of SEMIA6144 or Az39 under storage at 4 °C for 1 year.
• Alginate beads 2% ensure the gradual release of the microorganisms.
• Cells from beads stored for long periods present chemotaxis and adhesion to peanut root.
• Peanut inoculation with 1-year-old SEMIA6144 beads improves nodulation and growth in RWC.










Similar content being viewed by others
References
Ahemad M, Khan, MS (2009) Effect of insecticide-tolerant and plant growth promoting Mesorhizobium on the performance of chickpea grown in insecticide stressed alluvial soils. JCSB 12(4):213–222. https://doi.org/10.1007/s12892-009-0130-8
Albareda M, Dardanelli M, Sousa C, Megías M, Temprano F, Rodriguez-Navarro (2006) Factors affecting the attachment of rhizospheric bacteria to bean and soybean roots. FEMS Microbiol Lett 259:68–72. https://doi.org/10.1111/j.1574-6968.2006.00244.x
Albareda M, Rodrıguez-Navarro DN, Camacho M, Temprano FJ (2008) Alternatives to peat as a carrier for rhizobia inoculants: Solid and liquid formulations. Soil Biol Biochem 40:1–9. https://doi.org/10.1016/j.soilbio.2008.07.021
Almeida FF, Araújo AP, Rodrigues Alves BJ (2013) Seeds with high molybdenum concentration improved growth and nitrogen acquisition of Rhizobium-inoculated and Nitrogen- fertilized common bean plants. Rev Bras Ciênc Solo 37:378. https://doi.org/10.1590/S0100-06832013000200008
Amer GA, Utkhede RS (2000) Developments of formulations of biological agents for management of root of lettuce and cucumber. Can J Microbiol 46:809–816. https://doi.org/10.1139/cjm-46-9-809
Angelini J, Taurian T, Morgante C, Ibáñez F, Castro S, Fabra A (2005) Peanut nodulation kinetics in response to low pH. Plant Physiol Biochem 43:754–759. https://doi.org/10.1016/j.plaphy.2005.05.012
Arora NK, Khare E, Maheshwari DK (2011) Plant growth promoting rhizobacteria: constraints in bioformulation, commercialization, and future strategies. In: Maheshwari D (ed) Plant growth and health promoting bacteria. Microbiology Monographs, vol 18. Springer, Berlin, pp 97–116
Bais HP, Weir TL, Perry LG, Gilroy S, Vivanco JM (2006) The role of root exudates in rhizosphere interactions with plants and other organisms. Annu Re Plant Biol 57:233–266. https://doi.org/10.1146/annurev.arplant.57.032905.105159
Bashan Y (1998) Inoculants of plant growth-promoting bacteria for use in agriculture. Biotechnol Adv 4:729–770. https://doi.org/10.1016/S0734-9750(98)00003-2
Bashan Y, Gonzalez LE (1999) Long-term survival of the plant-growth-promoting bacteria Azospirillum brasilense and Pseudomonas fluorescens in dry alginate inoculant. Appl Microbiol Biotechnol 51:262–266. https://doi.org/10.1007/s002530051391
Bashan Y, Hernandez JP, Leyva-Macario Bacilio LA (2002) Alginate microbeads as inoculant carriers for plant growth-promoting bacteria. Biol Fertil Soils 35:359–368. https://doi.org/10.1007/s00374-002-0481-5
Bashan Y, de-Bashan LE, Prabhu SR, Hernandez JP (2014) Advances in plant growth-promoting bacterial inoculant technology: formulations and practical perspectives (1998–2013). Plant Soil 378:1–33. https://doi.org/10.1007/s11104-013-1956-x
Berney M, Hammes F, Bosshard F, Weilenmann H, Egli T (2007) Assessment and interpretation of bacterial viability by using the LIVE/DEAD BacLight Kit in combination with flow cytometry. Appl Environ Microbiol 73(10):3283–3290. https://doi.org/10.1128/AEM.02750-06
Berninger T, López O, Bejarano A, Preininger C, Sessitsch A (2018) Maintenance and assessment of cell viability in formulation of non-sporulating bacterial inoculants. Microb Biotechnol 11(2):277–301. https://doi.org/10.1111/1751-7915.12880
Bianucci E, Furlan A, Rivadeneira J, Sobrino-Plata J, Carpena-Ruiz R, Tordable C, Fabra A, Hernández B, Castro S (2013) Influence of cadmium on the symbiotic interaction established between peanut (Arachis hypogaea L.) and sensitive or tolerant bradyrhizobial strains. J Environ Manag 130:126–134. https://doi.org/10.1016/j.jenvman.2013.08.056
Boogerd FC, van Rossum D (1997) Nodulation of groundnut by Bradyrhizobium: a simple infection process by crack entry. FEMS Microbiol Rev 21:5–27. https://doi.org/10.1111/j.1574-6976.1997.tb00342.x
Caetano-Anollés G (1998) Molecular dissection and improvement of the nodule symbiosis in legumes. Field Crop Res 53:47–68. https://doi.org/10.1016/S0378-4290(97)00022-1
Cassidy MB, Lee H, Trevors JT (1996) Environmental applications of immobilized microbial cells: a review. J Ind Microbiol 16:79–101. https://doi.org/10.1007/BF01570068
Celikol A, Ercan M, Kavas L, Yildiz C, Yilmaz H, Oktem A, Yucel M (2010) Drought induced oxidative damage and antioxidant responses in peanut (Arachis hypogaea L.) seedlings. Plant Growth Regul 61:21–22. https://doi.org/10.1007/s10725-010-9445-1
Cesari AB, Paulucci NS, Biasutti M, Reguera Y, Gallarato L, Kilmurray C, Dardanelli MS (2016) Reorganization of Azospirillum brasilense cell membrane is mediated by lipid composition adjustment to maintain optimal fluidity during water deficit. J Appl Microbiol 120:185–194. https://doi.org/10.1111/jam.12994
Cesari AB, Paulucci NS, Biasutti M, Morales G, Dardanelli MS (2018) Changes in the lipid composition of Bradyrhizobium cell envelope reveal a rapid response to water deficit involving lysophosphatidylethanolamine synthesis from phosphatidylethanolamine in outer membrane. Res Microbiol 169:303–312. https://doi.org/10.1016/j.resmic.2018.05.008
Cesari AB, Paulucci NS, López-Gómez M, Hidalgo-Castellanos J, Lluch Plá C, Dardanelli MS (2019a) Performance of Bradyrhizobium and Bradyrhizobium–Azospirillum in alleviating the effects of water-restrictive conditions during the early stages of Arachis hypogaea growth. J Plant Growth Regul 38:1362–1374. https://doi.org/10.1007/s00344-019-09939-4
Cesari AB, Paulucci NS, López-Gómez M, Hidalgo-Castellanos J, Lluch Plá C, Dardanelli MS (2019b) Restrictive water condition modifies the root exudates composition during peanut-PGPR interaction and conditions early events, reversing the negative effects on plant growth. Plant Physiol Biochem 142:519–527. https://doi.org/10.1016/j.plaphy.2019.08.015
Cesari AB, Loureiro M, Vale M, Yslas EI, Dardanelli MS, Marques AC (2019c) Polycaprolactone microcapsules containing citric acid and naringin for plant growth and sustainable agriculture: physico-chemical properties and release behavior. Sci Total Environ:703. https://doi.org/10.1016/j.scitotenv.2019.135548
Cooper JE (2007) Early interactions between legumes and rhizobia: disclosing complexity in a molecular dialogue. J Appl Microbiol 103:1355–1365. https://doi.org/10.1111/j.1365-2672.2007.03366.x
Córdoba AL, Deladino L, Martino M (2013) Effect of starch filler on calcium-alginate hydrogels loaded with yerba mate antioxidants. Carbohydr Polym 95:315–323. https://doi.org/10.1016/j.carbpol.2013.03.019
da Silva R (1996) Tecnica de Microgota para contagem de celulas bacterianas viaveisem uma suspensao. Vi1osa, Minas Gerais, Brazil: Universidade Federal de Vi1osa
Dardanelli M, Fernández F, Espuny M, Rodríguez Carvajal M, Soria Díaz M, Gil Serrano M, Okon Y, Megías M (2008) Effect of Azospirillum brasilense coinoculated with Rhizobiumon Phaseolus vulgaris flavonoids and Nod factor production under salt stress. Soil Biol Biochem 40:2713–2721. https://doi.org/10.1016/j.soilbio.2008.06.016
Di Rienzo J, Casanoves F, Balzarini M, Gonzalez L, Tablada M, Robledo C (2016) InfoStat version. InfoStat Group. National University of Córdoba, Argentina. https://www.infostat.com.ar
Döbereiner J, Day J (1976) Associative symbioses in tropical grasses: characterization of microorganisms and dinitrogen- fixing sites. In: Nyman W (ed) Proceedings of the 1st International Symposium on Nitrogen Fixation, vol 2. Pullman Washington State University Press, Washington, pp 518–538
Donthidi AR, Tester RF, Aidoo KE (2010) Effect of lecithin and starch on alginate-encapsulated probiotic bacteria. J Microencapsul 27:67–77. https://doi.org/10.3109/02652040902982183
Fabra A, Castro S, Taurian T, Angelini J, Ibañez F, Dardanelli M, Tonelli M, Bianucci E, Valetti L (2010) Interaction among Arachis hypogaea L. (peanut) and beneficial soil microorganisms: how much is it known? Crit Rev Microbiol 36:179–194. https://doi.org/10.3109/10408410903584863
Fenice M, Selbman L, Federici F, Vassilev N (2000) Application of encapsulated Penicillium variabile P16 in solubilization of rock phosphate. Bioresour Technol 73:157–162. https://doi.org/10.1016/S0960-8524(99)00150-9
Glickmann E, Dessaux Y (1995) A critical examination of the specificity of the Salkowski Reagent for indolic compounds produced by phytopathogenic bacteria. Appl Environ Microbiol 61:793–796. https://doi.org/10.1128/AEM.61.2.793-796.1995
Gombotz WR, Wee SF (2012) Protein release from alginate matrices. Adv Drug Deliv Rev 64:194–205. https://doi.org/10.1016/s0169-409x(97)00124-5
Gomes Germano M, Gomes Germano M, Menna P, Menna F, Mostasso F, Mostasso M, Hungria M (2006) RFLP analysis of the rRNA operon of a Brazilian collection of bradyrhizobial strains from 33 legume species. Int J Sysst Evol Micr 56:217–229. https://doi.org/10.1099/ijs.0.02917-0
He Y, Wu Z, Ye B, Wang Y, Guan X, Zhang J (2016) Viability evaluation of alginate-encapsulated Pseudomonas putida Rs-198 under simulated salt-stress conditions and its effect on cotton growth. Eur J Soil Biol 75:135–141. https://doi.org/10.1016/j.ejsobi.2016.05.002
Hoagland D, Arnon D (1938) The water-culture method for growing plants without soil, vol 347. Circular California University Agricultural Experiment Station, pp 1–39
Hungria M, Nogueira M, Araujo R (2015) Alternative methods and time for soybean inoculation to overcome adverse conditions at sowing. Afr J Agric Res 10(23):2329–2338. https://doi.org/10.5897/AJAR2014.8687
Hungria M, Nogueira M, Araujo R (2016) Inoculation of Brachiaria spp. with the plant growth-promoting bacterium Azospirillum brasilense: an environment-friendly component in the reclamation of degraded pastures in the tropics. Agric Ecosyst Environ 221:125–131. https://doi.org/10.1016/j.agee.2016.01.024
INTA (2018) Informe de sequía en el territorio sudoeste de Córdoba. Campaña 2017–2018. https://inta.gob.ar/sites/default/files/inta_informe_sequia_pretso_cba.pdf
INTA (2020) Análisis de la evolución del área sembrada de Maní en la provincia de Córdoba. Campaña 2019-2020. https://inta.gob.ar/documentos/analisis-de-la-evolucion-del-area-sembrada-de-mani-en-la-provincia-de-cordoba
Joe MM, Karthikeyan B, Chauhan PS, Shagol C, Islam R, Deiveekasundarama R, Sa T (2012) Survival of Azospirillum brasilense flocculated cells in alginate and its inoculation effect on growth and yield of maize under water deficit conditions. Eur J Soil Biol 50:198–206. https://doi.org/10.1016/j.ejsobi.2012.03.002
Joe MM, Saravanan VS, Islam MR, Sa T (2014) Development of alginate-based aggregate inoculants of Methylobacterium sp. and Azospirillum brasilense tested under in vitro conditions to promote plant growth. J Appl Microbiol 116(2):408–423. https://doi.org/10.1111/jam.12384
John RP, Tyagi RD, Brar SK, Surampalli RY, Prévost D (2011) Bio-encapsulation of microbial cells for targeted agricultural delivery. Crit Rev Biotechnol 30:211–226. https://doi.org/10.3109/07388551.2010.513327
Kim KI, Baek YJ, Yoon YH (1996) Effects of rehydration media and immobilization in calcium-alginate on the survival of Lactobacillus casei and Bifidobacterium bifidum. Korean J Dairy Sci 18:193–198. https://doi.org/10.1016/j.idairyj.2005.10.005
Kris-Etherton P, Hu FB, Ros E, Sabaté J (2008) The role of tree nuts and peanuts in the prevention of coronary heart disease: Multiple potential mechanisms. J Nutr 138:1746–1751. https://doi.org/10.1093/jn/138.9.1746S
Liang T, Yanhui H, Hongbing Y, Zhansheng W, Lijuan Y (2015) Preparation and characterization of alginate–gelatin microencapsulated Bacillus subtilis SL-13 by emulsification/internal gelation. J Biomat Sci Polymer Ed 26:735–749. https://doi.org/10.1080/09205063.2015.1056075
Liu X, Germaine K, Ryan D, Dowling D (2007) Development of an alginate encapsulated gfp-based biosensor for detecting the bioavailability and biodegradation of polychlorinated biphenyls and chlorobenzoates. J Environ Eng Landsc Manag 4:261–268. https://doi.org/10.1080/16486897.2007.9636939
Loureiro MV, Ciriminna R, Lourenço MJ, Santos LF, De Schrijver A, Bordado JC, Pagliaro M, Marques AC (2017) Organically-modified silica based microspheres for self-curing polyurethane one component foams. Microporous Mesoporous Mater 244:244–250. https://doi.org/10.1016/j.micromeso.2016.10.039
Manivannan P, Jaleel CA, Sankar B, Kishorekumar A, Somasundaram R, Lakshmanan GMA, Panneerselvam R (2007) Growth, biochemical modifications and proline metabolism in Helianthus annuus L. as induced by drought stress. Colloid Surface B 59:141–149. https://doi.org/10.1016/j.colsurfb.2007.05.002
Markusen JF, Mason C, Hull DA, Town MA, Tabor AB, Clements M, Boshoff CH, Dunnill P (2006) Behavior of adult human mesenchymal stem cells entrapped in alginate- GRGDY Beads. Tissue Eng 12(4):821–830. https://doi.org/10.1089/ten.2006.12.821
Medeot D, Sohlenkamp C, Dardanelli MS, Geiger O, García M, Lopez-Lara I (2010) Phosphatidylcholine levels of peanut-nodulating Bradyrhizobium sp. SEMIA6144 affect cell size and motility. FEMS Microbiol Lett 303:123–131. https://doi.org/10.1111/j.1574-6968.2009.01873.x
Mengual C, Roldán A, Caravaca F, Schoebitz M (2014) Advantages of inoculation with immobilized rhizobacteria versus amendment with olive-mill waste in the afforestation of a semiarid area with Pinus halepensis Mill. Ecol Eng 73:1–8. https://doi.org/10.1016/j.ecoleng.2014.09.007
Morra MJ, Chaverra MH, Dandurand LM, Orser CS (1998) Survival of Pseudomonas fluorescens 2-79RN10 in clay powders undergoing drying. Soil Sci Soc Am J 62:663–670. https://doi.org/10.2136/sssaj1998.03615995006200030017x
Nava Saucedo JE, Audras B, Jan S, Bazinet CE, Barbotin JN (1994) Factors affecting densities, distribution and growth patterns of cells inside immobilization supports. FEMS Microbiology Reviews 14:93–98. https://doi.org/10.1111/j.1574-6976.1994.tb00079.x
Parke D, Rivelli M, Ornston LN (1985) Chemotaxis to aromatic and hydroaromatic acids: comparison of Bradyrhizobium japonicum and Rhizobium trifolii. J Bacteriol 163:417–422. https://doi.org/10.1128/AEM.57.9.2635-2639.1991
Paul E, Fages J, Blanc P, Goma G, Pareilleux A (1993) Survival of alginate-entrapped cells of Azospirillum lipoferum during dehydration and storage in relation to water properties. Appl Microbiol Biotechnol 40:34–39. https://doi.org/10.1007/BF00170425
Paulucci N, Gallarato L, Reguera Y, Vicario C, Cesari A, García M, Dardanelli M (2015) Arachis hypogaea PGPR isolated from Argentine soil modifies its lipids components in response to temperature and salinity. Microbiol Res 173:1–9. https://doi.org/10.1016/j.micres.2014.12.012
Power B, Liu X, Germaine K, Ryan D, Brazil D, Dowling D (2011) Alginate beads as a storage, delivery and containment system for genetically modified PCB degrader and PCB biosensor derivatives of Pseudomonas fluorescens F113. J Appl Microbiol 110(5):1351–1358. https://doi.org/10.1111/j.1365-2672.2011.04993.x
Rivera D, Obando M, Rojas-Tapias D, Buitrago RB, Barbosa H (2016) Entrapment of Rhizobium sp. by fluidized bed technique using polymers as coating materials. Univ Sci 21:117–128. https://doi.org/10.1007/s10725-017-0356-2
Rodriguez Cáceres E (1982) Improved medium for isolation of Azospirillum spp. Appl Environ Microbiol 44:990–991 https://europepmc.org/backend/ptpmcrender.fcgi?accid=PMC242127&blobtype=pdf
Roy A, Bajpai J, Bajpai K (2009) Dynamics of controlled release of chlorpyrifos from swelling and eroding biopolymeric microspheres of calcium alginate and starch. Carbohydr Polym 76(2):222–231. https://doi.org/10.1016/j.carbpol.2008.10.013
Sánchez MT, Ruiz MA, Lasserrot A, Hormigo M, Morales ME (2017) An improved ionic gelation method to encapsulate Lactobacillus spp. bacteria:Protection, survival and stability study. Food Hydrocoll 69:67–75.https://doi.org/10.1016/j.foodhyd.2017.01.019
Schoebitz M, Lopez Belch MD (2016) Encapsulation techniques for plant growth-promoting rhizobacteria. In: Bioformulations: for Sustainable Agriculture, pp 251–265. https://doi.org/10.1007/978-81-322-2779-3_14
Schoebitz M, Simonin H, Poncelet D (2012) Starch filler and osmoprotectants improve the survival of rhizobacteria in dried alginate beads. J Microencapsul 29:532–538. https://doi.org/10.3109/02652048.2012.665090
Schoebitz M, Mengual C, Roldán A (2013) Combined effects of clay immobilized Azospirillum brasilense and Pantoea dispersa and organic olive residue on plant performance and soil properties in the revegetation of a semiarid area. Sci Total Environ 466–467:67–73. https://doi.org/10.1016/j.scitotenv.2013.07.012
Schottroff F, Fröhling A, Zunabovic-Pichler M, Krottenthaler A, Schlüter O, Jäger H (2018) Sublethal Injury and Viable but Non-culturable (VBNC) State in microorganisms during preservation of food and biological materials by non-thermal processes. Front Microbiol 9:2773. https://doi.org/10.3389/fmicb.2018.02773
Smidsrød O, Skjak-Braek G (1990) Alginate as immobilization matrix for cells. Trends Biotechnol 8:71–78. https://doi.org/10.1016/0167-7799(90)90139-O
Somasegaran P, Hoben H (1994) Handbook for rhizobia. Methods in Legume- Rhizobium Technology, 1st edn. Spring-Verlag, New York, pp 332–341. https://doi.org/10.1007/978-1-4613-8375-8
Steenhoudt O, Vanderleyden J, Janssens FA (2011) Azospirillum, a free-living nitrogen-fixing bacterium closely associated with grasses: genetic, biochemical and ecological aspects. World J Microbiol Biotechnol 27:197–205. https://doi.org/10.1016/S0168-6445(00)00036-X
Tavares Salgado L, Tomazetto R, Paes L, Cinelli FM, Amado Filho G (2007) The influence of brown algae alginates on phenolic compounds capability of UV absorption in vitro. Braz J Oceanogr 55(2):145–154. https://doi.org/10.1590/S1679-87592007000200007
Trivedi P, Pandey A, Palni LMS (2005) Carrier-based preparations of plant growth-promoting bacterial inoculants suitable for use in cooler regions. World J Microbiol Biotechnol 21:19–23. https://doi.org/10.1007/s11274-004-6820-y
Vejan P, Khadiran T, Abdullah R, Ismail S, Dadrasnia A (2019) Encapsulation of plant growth promoting Rhizobacteria prospects and potential in agricultural sector: a review. J Plant Nutr 42:2600–2623. https://doi.org/10.1080/01904167.2019.1659330
Vincent J (1970) A manual for the practical study of root nodule bacteria. In: Internat. Biol. Progr. Handbook No. Blackwell Scientific Publications Ltd, Oxford
Wu Z, Zhao Y, Kaleem I, Li C (2011) Preparation of calcium–alginate microcapsuled microbial fertilizer coating Klebsiella oxytoca Rs5 and its performance under salinity stress. Eur J Soil Biol 47:152–159. https://doi.org/10.1016/j.ejsobi.2010.11.008
Wu Z, Guo L, Qin S, Li C (2012) Encapsulation of R. planticola Rs-2 from alginate-starch-bentonite and its controlled release and swelling behavior undersimulated soil conditions. J Ind Microbiol Biotechnol 39:317–327. https://doi.org/10.1007/s10295-011-1028-2
Wu Z, Lib X, Liua L, Dongb J, Fan D, Xu X, He Y (2020) Membrane shell permeability of Rs-198 microcapsules and their ability for growth promoting bioactivity compound releasing. RSC Adv 10:1159–1171. https://doi.org/10.1039/c9ra06935f
Young C, Rekha P, Wei-An L, Arun A (2006) Encapsulation of plant growth-promoting bacteria in alginate beads enriched with humic acid. Biotechnol Bioeng 95(1):76–83. https://doi.org/10.1002/bit.20957
Yuan J, Zhang N, Huang Q, Raza W, Li R, Vivanco JM (2015) Organic acids from root exudates of banana help root colonization of PGPR strain Bacillus amyloliquefaciens NJN-6. Sci Rep 5:1–8. https://doi.org/10.1038/srep13438
Zhou J, Deng B, Zhang Y, Cobb AB, Zhang Z (2017) Molybdate in rhizobial seed-coat formulations improves the production and nodulation of alfalfa. PLoS One. https://doi.org/10.1371/journal.pone.0170179
Zohar-Perez C, Ritte E, Chernin L, Chet I, Nussinovitch A (2002) Preservation of Chitinolytic Pantoae agglomerans in a viable form by cellular dried alginate-based carriers. Biotechnol Prog 18:1133–1140. https://doi.org/10.1021/bp025532t
Zommere Z, Nikolajeva V (2017) Immobilization of bacterial association in alginate beads for bioremediation of oil-contaminated lands. Environ Exp Biol 15:105–111. https://doi.org/10.2174/1874285801509010048
Zur J, Wojcieszynska D, Guzik U (2016) Metabolic responses of bacterial cells to immobilization. Molecules 21:958. https://doi.org/10.3390/molecules21070958
Acknowledgments
We thank Dr. Fabiana Deramo (UNRC) for the Nitrogen analysis service, Dr. Diego Acevedo (UNRC) for preparing the FTIR, and Dr. Ana Marques (IST, Lisbon) for allowing the use of SEM. Finally, we are also grateful to the anonymous editors and reviewers for their comments and suggestions.
Funding
Financial support was provided by PIP CONICET 112-201501-00232, SECYT UNRC N° 161/16, PID 144/2018, PICT-2016-0417, and GRFT-2019. AC is a fellow of CONICET-Argentina. MD, EY, and NP are members of the Research Career of CONICET Argentina.
Author information
Authors and Affiliations
Contributions
AC, NP, EY, and MD conceived and designed the experiments. AC and EY performed the MTT measurements, and interpretation of living and dead cells. AC and NP performed chemotaxis and adhesion experiments. AC synthesized the alginate microspheres and carried out physico-chemical analysis and viable count and conducted plant inoculation tests and statistical analysis. AC, NP, EY, and MD wrote the manuscript, with contributions from all authors. All authors read and approved.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical statement
This article does not contain any study with human participants or animals performed by any of the author.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Cesari, A.B., Paulucci, N.S., Yslas, E.I. et al. Immobilization of Bradyrhizobium and Azospirillum in alginate matrix for long time of storage maintains cell viability and interaction with peanut. Appl Microbiol Biotechnol 104, 10145–10164 (2020). https://doi.org/10.1007/s00253-020-10910-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-020-10910-7