Abstract
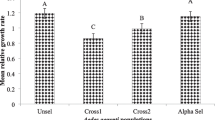
Aedes aegypti is a globally spread disease vector of supreme concern, primarily controlled by chemical insecticides. Current study investigates the comparative rate of acetamiprid and deltamethrin resistance development in Ae. aegypti larvae and; possible correlation between resistance and cuticular thickening. The larvae were selected with LC90 level of the respective insecticide for 10 successive generations and the level of resistance induced was estimated. The larvae of parent (PS), acetamiprid-selected (ACSF-10) and deltamethrin-selected (DLSF-10) strains were sectioned through first abdominal segment to elucidate the variation in cuticular thickness. The PS larvae of Ae. aegypti were 229.26-fold higher susceptible to deltamethrin as compared to acetamiprid, exhibiting corresponding LC50 values of 0.00082 mg/L and 0.18799 mg/L. Larval selections induced 9.91-fold and 19.74-fold resistance to deltamethrin and acetamiprid, respectively; indicating multifactorial and heterozygous pattern. Cuticular analysis of the larval sections demonstrated a significantly thicker cuticle in selected strains, the mean thickness in PS, ACSF-10 and DLSF-10 was 2.891 ± 0.243, 4.288 ± 0.508 and 5.695 ± 0.437 μm, respectively revealing 1.48-fold and 1.97-fold thicker cuticle in the ACSF-10 and DLSF-10 strains, respectively in comparison to PS strain. The increased cuticular thickness possibly inhibited the insecticide penetration in selected larvae resulting in the resistance development. This is the first such report which demonstrates the association between acetamiprid/deltamethrin resistance in Ae. aegypti larvae with the cuticular thickness.





Similar content being viewed by others
Data availability
Not Applicable.
References
Abbott WS (1925) A method of computing the effectiveness of an insecticide. J Econ Entomol 18:265–267
Balabanidou V, Grigoraki L, Vontas J (2018) Insect cuticle: a critical determinant of insecticide resistance. Curr Opin Insect Sci 27:68–74. https://doi.org/10.1016/j.cois.2018.03.001
Bass C, Puinean AM, Andrews M, Cutler P, Daniels M, Elias J, Paul VL, Crossthwaite AJ, Denholm I, Field LM, Foster SP (2011) Mutation of a nicotinic acetylcholine receptor β subunit is associated with resistance to neonicotinoid insecticides in the aphid Myzus persicae. BMC Neurosci 12:51. https://doi.org/10.1186/1471-2202-12-51
Benelli G, Conti B, Garreffa R, Nicoletti M (2014) Shedding light on bioactivity of botanical by-products: neem cake compounds deter oviposition of the arbovirus vector Aedes albopictus (Diptera: Culicidae) in the field. Parasitol Res 113:933–940. https://doi.org/10.1007/s00436-013-3725-4
Bhatt S, Gething PW, Brady OJ, Messina JP, Farlow AW, Moyes CL, Drake JM, Brownstein JS, Hoen AG, Sankoh O, Myers MF, George DB, Jaenisch T, Wiliam Wint GR, Simmons CP, Scott TW, Farrar JJ, Hay SI (2013) The global distribution and burden of dengue. Nature 496:504–507. https://doi.org/10.1038/nature12060
Brady OJ, Gething PW, Bhatt S, Messina JP, Brownstein JS, Hoen AG, Moyes CL, Farlow AW, Scott TW, Hay SI (2012) Refining the global spatial limits of dengue virus transmission by evidence-based consensus. PLoS Negl Trop Dis 6:6e1760. https://doi.org/10.1371/journal.pntd.0001760
Brengues C, Hawkes NJ, Chandre F, McCarroll L, Duchon S, Guillet P, Manguin S, Morgan JC, Hemingway J (2003) Pyrethroid and DDT cross-resistance in Aedes aegypti is correlated with novel mutations in the voltage-gated sodium channel gene. Med Vet Entomol 17:87–94. https://doi.org/10.1046/j.1365-2915.2003.00412.x
Casida JE, Durkin KA (2013) Neuroactive insecticides: targets, selectivity, resistance, and secondary effects. Annu Rev Entomol 58:99–117. https://doi.org/10.1146/annurev-ento-120811-153645
Casida JE, Quistad GB (2004) Why insecticides are more toxic to insects than people: the unique toxicology of insects. J Pestic Sci 29:81–86. https://doi.org/10.1584/jpestics.29.81
Corbel V, Duchon S, Zaim M, Hougard JM (2004a) Dinotefuran: a potential neonicotinoid insecticide against resistant mosquitoes. J Med Entomol 41:712–717. https://doi.org/10.1603/0022-2585-41.4.712
Corbel V, Raymond M, Chandre F, Darriet F, Hougard JM (2004) Efficacy of insecticide mixtures against larvae of Culex quinquefasciatus (Say) (Diptera: Culicidae) resistant to pyrethroids and carbamates. Pest Manag Sci 60:375–380. https://doi.org/10.1002/ps.809
Djouaka RF, Bakare AA, Coulibaly ON, Akogbeto MC, Ranson H, Hemingway J, Strode C (2008) Expression of the cytochrome P450s, CYP6P3 and CYP6M2 are significantly elevated in multiple pyrethroid resistant populations of Anopheles gambiae ss. from southern Benin and Nigeria. BMC Genomics 9:538. https://doi.org/10.1186/1471-2164-9-538
Elbert A, Nauen R, Leicht W (1998) Imidacloprid, a novel chloronicotinyl insecticide: biological activity and agricultural importance. In: Ishaaya I, Degheele D (eds) Insecticides with novel modes of action. Springer, Berlin, Heidelberg, pp 50–73. https://doi.org/10.1007/978-3-662-03565-8_4
Elbert A, Haas M, Springer B, Thielert W, Nauen R (2008) Applied aspects of neonicotinoid uses in crop protection. Pest Manag Sci 64:1099–1105. https://doi.org/10.1002/ps.1616
Fine BC, Godin PJ, Thain EM (1963) Penetration of pyrethrin I labelled with Carbon-14 into susceptible and pyrethroid resistant houseflies. Nature 199:927–928. https://doi.org/10.1038/199927b0
Georghiou GP, Metcalf RL, March RB (1961) The development and characterization of resistance to carbamate insecticides in the house fly, Musca domestica. J Econ Entomol 54:132–140. https://doi.org/10.1093/jee/54.1.132
Goulson D (2013) An overview of the environmental risks posed by neonicotinoid insecticides. J Appl Ecol 50:977–987. https://doi.org/10.1111/1365-2664.12111
Hemingway J, Field L, Vontas J (2002) An overview of insecticide resistance. Science 298:96–97. https://doi.org/10.1126/science.1078052
Ilias A, Lagnel J, Kapantaidaki DE, Roditakis E, Tsigenopoulos CS, Vontas J, Tsagkarakou A (2015) Transcription analysis of neonicotinoid resistance in Mediterranean (MED) populations of B. tabaci reveal novel cytochrome P450s, but no nAChR mutations associated with the phenotype. BMC Genomics 16:939. https://doi.org/10.1186/s12864-015-2161-5
Jian-chu MO, Tian-ci YA, Jia-an CH, Xiao-gang SO (2002) Lethal and sublethal effects of acetamiprid on the larvae of Culex pipiens pallens. Insect Sci 9:45–49. https://doi.org/10.1111/j.1744-7917.2002.tb00153.x
Kasai S, Komagata O, Itokawa K, Shono T, Ng LC, Kobayashi M, Tomita T (2014) Mechanisms of pyrethroid resistance in the dengue mosquito vector, Aedes aegypti: target site insensitivity, penetration, and metabolism. PLoS Negl Trop Dis 8:e2948. https://doi.org/10.1371/journal.pntd.0002948
Khan HA, Akram W (2019) Resistance status to Deltamethrin, Permethrin, and Temephos along with preliminary resistance mechanism in Aedes aegypti (Diptera: Culicidae) from Punjab, Pakistan. J Med Entomol 56:1304–1311. https://doi.org/10.1093/jme/tjz057
Kimura-Kuroda J, Komuta Y, Kuroda Y, Hayashi M, Kawano H (2012) Nicotine-like effects of the neonicotinoid insecticides acetamiprid and imidacloprid on cerebellar neurons from neonatal rats. PLoS One 7:e32432. https://doi.org/10.1371/journal.pone.0032432
Kumar S, Thomas A, Sahgal A, Verma A, Samuel T, Pillai MK (2002) Effect of the synergist, piperonyl butoxide, on the development of deltamethrin resistance in yellow fever mosquito, Aedes aegypti L. (Diptera: Culicidae). Arch Insect Biochem Physiol 50:1–8. https://doi.org/10.1002/arch.10021
Kumar S, Thomas A, Samuel T, Sahgal A, Verma A, Pillai MK (2009) Diminished reproductive fitness associated with the deltamethrin resistance in an Indian strain of dengue vector mosquito, Aedes aegypti L. Trop Biomed 26:155–164
Kumar S, Mishra M, Wahab N, Warikoo R (2014) Larvicidal, repellent, and irritant potential of the seed-derived essential oil of Apium graveolens against dengue vector, Aedes aegypti L. (Diptera: Culicidae). Front Public Health 2:147. https://doi.org/10.3389/fpubh.2014.00147
Lee CY, Yap HH (2003) Overview on urban pests: a Malaysian perspective. In: Chong NL, Lee CY, Jaal Z, Yap HH (eds) Urban Pest control - a Malaysian perspective Vol 2. Vector Control Research Unit, Universiti Sains Malaysia, Penang, Malaysia
Li CX, Kaufman PE, Xue RD, Zhao MH, Wang G, Yan T, Guo XX, Zhang YM, Dong YD, Xing D, Zhang HD (2015) Relationship between insecticide resistance and kdr mutations in the dengue vector Aedes aegypti in southern China. Parasites Vectors 8:325. https://doi.org/10.1186/s13071-015-0933-z
Lilly DG, Latham SL, Webb CE, Doggett SL (2016) Cuticle thickening in a pyrethroid-resistant strain of the common bed bug, Cimex lectularius L. (Hemiptera: Cimicidae). PLoS ONE 11:e0153302. https://doi.org/10.1371/journal.pone.0153302
Liu N (2015) Insecticide resistance in mosquitoes: impact, mechanisms, and research directions. Annu Rev Entomol 60:537–559. https://doi.org/10.1146/annurev-ento-010814-020828
Liu Z, Williamson MS, Lansdell SJ, Denholm I, Han Z, Millar NS (2005) A nicotinic acetylcholine receptor mutation conferring target-site resistance to imidacloprid in Nilaparvata lugens (brown planthopper). Proc Natl Acad Sci 102:8420–8425. https://doi.org/10.1073/pnas.0502901102
Liu N, Xu Q, Zhu F, Zhang LE (2006) Pyrethroid resistance in mosquitoes. Insect Sci 13:159–166. https://doi.org/10.1111/j.1744-7917.2006.00078.x
Liu Z, Yao X, Zhang Y (2008) Insect nicotinic acetylcholine receptors (nAChRs): important amino acid residues contributing to neonicotinoid insecticides selectivity and resistance. Afr J Biotechnol 7:4935–4939
Matsuda K, Kanaoka S, Akamatsu M, Sattelle DB (2009) Diverse actions and target-site selectivity of neonicotinoids: structural insights. Mol Pharmacol 76:1–10. https://doi.org/10.1124/mol.109.055186
Matsumura F, Brown AW (1961) Biochemical study of a malathion tolerant strain of Aedes aegypti. Mosq News. 21:192–194
Matsumura F, Brown AW (1963) Studies on Organophosphorus-tolerance in Aedes aegypti. Mosq News 23:26–31
Moore MT, Cooper CM, Smith S Jr, Cullum RF, Knight SS, Locke MA, Bennett ER (2009) Mitigation of two pyrethroid insecticides in a Mississippi Delta constructed wetland. Environ Pollut 157:250–256. https://doi.org/10.1016/j.envpol.2008.07.025
Mouhamadou CS, de Souza SS, Fodjo BK, Zoh MG, Bli NK, Koudou BG (2019) Evidence of insecticide resistance selection in wild Anopheles coluzzii mosquitoes due to agricultural pesticide use. Infect Dis Poverty 8:64. https://doi.org/10.1186/s40249-019-0572-2
National Vector Borne Disease Control Programme (NVBDCP) (2020a) Dengue/DHF situation in India. [Online] Available at:https://nvbdcp.gov.in/index4.php?lang=1&level=0&linkid=431&lid=3715. Accessed 7 May 2020
National Vector Borne Disease Control Programme (NVBDCP) (2020b) Chikungunya situation in India. [Online] Available at:https://nvbdcp.gov.in/index4.php?lang=1&level=0&linkid=486&lid=3765. Accessed 7 May 2020
Paul A, Harrington LC, Scott JG (2006) Evaluation of novel insecticides for control of dengue vector Aedes aegypti (Diptera: Culicidae). J Med Entomol 43:55–60. https://doi.org/10.1093/jmedent/43.1.55
Pedrini N, Mijailovsky SJ, Girotti JR, Stariolo R, Cardozo RM, Gentile A, Juárez MP (2009) Control of pyrethroid-resistant Chagas disease vectors with entomopathogenic fungi. PLoS Negl Trop Dis 3:e434. https://doi.org/10.1371/journal.pntd.0000434
Ponlawat A, Scott JG, Harrington LC (2005) Insecticide susceptibility of Aedes aegypti and Aedes albopictus across Thailand. J Med Entomol 42:821–825. https://doi.org/10.1093/jmedent/42.5.821
Rao MS, Murty US, Gangadasu B, Raju BC, Ramesh CH, Kumar SB, Rao VJ (2008) Larvicidal efficacy of neonicotinoid classes of compounds on Culex quinquefasciatus. J Entomol 5:45–50. https://doi.org/10.3923/je.2008.45.50
Samal RR, Kumar S (2018) Susceptibility status of Aedes aegypti L. against different classes of insecticides in New Delhi, India to formulate mosquito control strategy in fields. Open Parasitol J 6:52–62. https://doi.org/10.2174/1874421401806010052
Sanchez-Bayo FP (2012) Insecticides mode of action in relation to their toxicity to non-target organisms. J Environ Anal Toxicol S4:002. https://doi.org/10.4172/2161-0525.S4-002
Sawicki RM, Lord KA (1970) Some properties of a mechanism delaying penetration of insecticides into houseflies. Pestic Sci 1:213–217. https://doi.org/10.1002/ps.2780010512
Schmuck R (2001) Ecotoxicological profile of the insecticide thiacloprid. Pflanzenschutz Nachrichten-Bayer-English Edition 54:161–184
Shetty V, Sanil D, Shetty NJ (2013) Insecticide susceptibility status in three medically important species of mosquitoes, Anopheles stephensi, Aedes aegypti and Culex quinquefasciatus, from Bruhat Bengaluru Mahanagara Palike, Karnataka, India. Pest Manag Sci 69:257–267. https://doi.org/10.1002/ps.3383
Takahashi H, Mitsui J, Yano M, Take T, Asai M, Yamada T (1999) Efficacy of acetamiprid 2% granule against green peach aphid [Myzus persicae] on cabbage [Brassica oleracea] by various soil application methods. J Pestic Sci 24:270–274
Tomizawa M, Casida JE (2005) Neonicotinoid insecticide toxicology: mechanisms of selective action. Annu. Rev Pharmacol Toxicol 45:247–268. https://doi.org/10.1146/annurev.pharmtox.45.120403.095930
Uragayala S, Verma V, Natarajan E, Velamuri PS, Kamaraju R (2015) Adulticidal and larvicidal efficacy of three neonicotinoids against insecticide susceptible & resistant mosquito strains. Indian J Med Res 142:S64–S70. https://doi.org/10.4103/0971-5916.176624
US Environmental Protection Agency (US-EPA) Factsheet-Acetamiprid (2002) https://www3.epa.gov/pesticides/chem_search/reg_actions/registration/fs_PC-099050_15-Mar-02.pdf. pp 23
Valle D, Bellinato DF, Viana-Medeiros PF, Lima JB, Martins Junior AD (2019) Resistance to temephos and deltamethrin in Aedes aegypti from Brazil between 1985 and 2017. Mem Inst Oswaldo Cruz 114:1–7. https://doi.org/10.1590/0074-02760180544
Vinson BS, Brazzel JR (1966) The penetration and metabolism of C14-labeled DDT in resistant and susceptible tobacco budworm larvae, Heliothis virescens (F.). J Econ Entomol 59:600–604. https://doi.org/10.1093/jee/59.3.600
Vontas J, David JP, Nikou D, Hemingway J, Christophides GK, Louis C, Ranson H (2007) Transcriptional analysis of insecticide resistance in Anopheles stephensi using cross-species microarray hybridization. Insect Mol Biol 16:315–324. https://doi.org/10.1111/j.1365-2583.2007.00728.x
Warikoo R, Ray A, Sandhu JK, Samal R, Wahab N, Kumar S (2012) Larvicidal and irritant activities of hexane leaf extracts of Citrus sinensis against dengue vector Aedes aegypti L. Asian Pac J Trop Biomed 2:152–155. https://doi.org/10.1016/S2221-1691(11)60211-6
Wood OR, Hanrahan S, Coetzee M, Koekemoer LL, Brooke BD (2010) Cuticle thickening associated with pyrethroid resistance in the major malaria vector Anopheles funestus. Parasit Vectors 3:67. https://doi.org/10.1186/1756-3305-3-67
World Health Organization (2005) Guidelines for laboratory and field testing of mosquito larvicides (No. WHO/CDS/WHOPES/GCDPP/2005.13). World Health Organization. https://apps.who.int/iris/bitstream/handle/10665/69101/WHO_CDS_WHOPES_GCDPP_2005.13.pdf
Yang T, Liu N (2014) Permethrin resistance variation and susceptible reference line isolation in a field population of the mosquito, Culex quinquefasciatus (Diptera: Culicidae). Insect Sci 21:659–666. https://doi.org/10.1111/1744-7917.12071
Zhu F, Gujar H, Gordon JR, Haynes KF, Potter MF, Palli SR (2013) Bed bugs evolved unique adaptive strategy to resist pyrethroid insecticides. Sci Rep 3:1456. https://doi.org/10.1038/srep01456
Acknowledgements
The authors are highly grateful to the Council of Scientific Research (CSIR), New Delhi, India for providing financial assistance to carry out the present investigations. We are also highly grateful to the Principal, Acharya Narendra Dev College for providing infrastructure and research facilities.
Funding
This research was supported by the contingent grant from Council of Scientific and Industrial Research, New Delhi, India (Award No. 08/529(0003)/2015-EMR-I).
Author information
Authors and Affiliations
Contributions
RRS conceived the idea, conducted the experiments and wrote the manuscript. SK designed and guided the experiments. RRS analysed the results and SK helped in the statistical analysis. Both the authors were involved in the finalization of manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Authors declare no conflict of interest.
Ethics approval
Not Applicable.
Code availability
Not Applicable.
Consent of participation
Not Applicable.
Consent for publication
Not Applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Samal, R.R., Kumar, S. Cuticular thickening associated with insecticide resistance in dengue vector, Aedes aegypti L.. Int J Trop Insect Sci 41, 809–820 (2021). https://doi.org/10.1007/s42690-020-00271-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42690-020-00271-z