Abstract
Purpose
To explore changes in functional connectivity (FC) within the sensorimotor network (SMN) and the relationship between the SMN and bilateral thalamus in patients with thalamic infarction (TI) using resting state functional magnetic resonance imaging (rs-fMRI). Also determined was whether those measures are useful for monitoring the functional recovery of somatosensory deficits.
Methods
The study included 31 patients with TI presenting somatosensory dysfunction and 31 controls who underwent clinical assessments and MRI scanning at 6 months after a stroke. An independent component analysis was used to identify the SMN. The mean time courses of SMN activity were extracted for each subject, and FC with the bilateral thalamus was assessed. Differences in connectivity strength were compared between groups. Finally, we correlated the altered FC values with clinical data from patients with TI.
Results
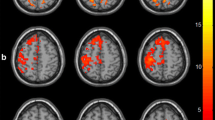
Compared to controls, patients with TI showed decreases in FC within SMN in the ipsilesional posterior central gyrus (PCG) (Z-score = −4.581, cluster size = 171), but presented increased FC within the SMN in the ipsilesional supplementary motor area (SMA) (Z-score = 4.648, cluster size = 46). The FC values of the ipsilesional SMA correlated with the somatosensory function score of patients with TI (r = 0.426, P = 0.027). Increased FC was observed between the SMN and bilateral thalamus in patients with TI. The region exhibiting increased FC was adjacent to the lesion in the affected thalamus, while the area with increased FC overlapped the location of the lesion when the lesion was mirrored onto the unaffected thalamus.
Conclusion
The increased FC in the ipsilesional SMA and between the SMN and perilesional thalamus might reflect functional reorganization in patients with TI presenting somatosensory deficits.



Similar content being viewed by others
References
Milosevic L, Kalia SK, Hodaie M, Lozano AM, Popovic MR, Hutchison WD. Physiological mechanisms of thalamic ventral intermediate nucleus stimulation for tremor suppression. Brain. 2018;141:2142–55. Erratum in: Brain. 2018;141:e72.
Li S, Kumar Y, Gupta N, Abdelbaki A, Sahwney H, Kumar A, Mangla M, Mangla R. Clinical and Neuroimaging Findings in Thalamic Territory Infarctions: A Review. J Neuroimaging. 2018;28:343–9.
Schmahmann JD. Vascular syndromes of the thalamus. Stroke. 2003;34:2264–78.
Kim JH, Greenspan JD, Coghill RC, Ohara S, Lenz FA. Lesions limited to the human thalamic principal somatosensory nucleus (ventral caudal) are associated with loss of cold sensations and central pain. J Neurosci. 2007;27:4995–5004.
Vartiainen N, Perchet C, Magnin M, Creac’h C, Convers P, Nighoghossian N, Mauguière F, Peyron R, Garcia-Larrea L. Thalamic pain: anatomical and physiological indices of prediction. Brain. 2016;139:708–22.
Schubert V, Lauxmann S, Bender B, Lerche H. Thalamic transitory ischemic attacks presenting as Jacksonian sensory march. J Neurol. 2017;264:2158-61.
Morel A. The thalamus and behavior: effects of anatomically distinct strokes. Neurology. 2007;68:1640; author reply 1640–1.
Kessner SS, Bingel U, Thomalla G. Somatosensory deficits after stroke: a scoping review. Top Stroke Rehabil. 2016;23:136–46.
Kishi M, Sakakibara R, Nagao T, Terada H, Ogawa E. Thalamic infarction disrupts spinothalamocortical projection to the mid-cingulate cortex and supplementary motor area. J Neurol Sci. 2009;281:104–7.
Ohara S, Lenz FA. Reorganization of somatic sensory function in the human thalamus after stroke. Ann Neurol. 2001;50:800–3.
Staines WR, Black SE, Graham SJ, McIlroy WE. Somatosensory gating and recovery from stroke involving the thalamus. Stroke. 2002;33:2642–51.
Lee MY, Kim SH, Choi BY, Chang CH, Ahn SH, Jang SH. Functional MRI finding by proprioceptive input in patients with thalamic hemorrhage. NeuroRehabilitation. 2012;30:131–6.
Dacosta-Aguayo R, Graña M, Iturria-Medina Y, Fernández-Andújar M, López-Cancio E, Cáceres C, Bargalló N, Barrios M, Clemente I, Toran P, Forés R, Dávalos A, Auer T, Mataró M. Impairment of functional integration of the default mode network correlates with cognitive outcome at three months after stroke. Hum Brain Mapp. 2015;36:577–90.
Wang C, Qin W, Zhang J, Tian T, Li Y, Meng L, Zhang X, Yu C. Altered functional organization within and between resting-state networks in chronic subcortical infarction. J Cereb Blood Flow Metab. 2014;34:597–605.
Wang L, Yu C, Chen H, Qin W, He Y, Fan F, Zhang Y, Wang M, Li K, Zang Y, Woodward TS, Zhu C. Dynamic functional reorganization of the motor execution network after stroke. Brain. 2010;133:1224–38.
Louis ED, Yousefzadeh E, Barnes LF, Yu Q, Pullman SL, Wendt KJ. Validation of a portable instrument for assessing tremor severity in epidemiologic field studies. Mov Disord. 2000;15:95–102.
Lindmark B, Hamrin E. Evaluation of functional capacity after stroke as a basis for active intervention. Presentation of a modified chart for motor capacity assessment and its reliability. Scand J Rehabil Med. 1988;20:103–9.
Friston KJ, Williams S, Howard R, Frackowiak RS, Turner R. Movement-related effects in fMRI time-series. Magn Reson Med. 1996;35:346–55.
Calhoun VD, Liu J, Adali T. A review of group ICA for fMRI data and ICA for joint inference of imaging, genetic, and ERP data. Neuroimage. 2009;45(1 Suppl):S163–72.
Smith SM, Fox PT, Miller KL, Glahn DC, Fox PM, Mackay CE, Filippini N, Watkins KE, Toro R, Laird AR, Beckmann CF. Correspondence of the brain’s functional architecture during activation and rest. Proc Natl Acad Sci U S A. 2009;106:13040–5.
Li YO, Adah T, Calhoun VD. Sample dependence correction for order selection in fMRI analysis. 2006 3rd Ieee International Symposium on Biomedical Imaging: From Nano to Macro. Arlington, USA 2006. p. 1072–75.
Calhoun VD, Adali T, Pearlson GD, Pekar JJ. A method for making group inferences from functional MRI data using independent component analysis. Hum Brain Mapp. 2001;14:140-51. Erratum in: Hum Brain Mapp 2002;16:131.
Carey LM, Abbott DF, Harvey MR, Puce A, Seitz RJ, Donnan GA. Relationship between touch impairment and brain activation after lesions of subcortical and cortical somatosensory regions. Neurorehabil Neural Repair. 2011;25:443–57.
Tahmasian M, Bettray LM, van Eimeren T, Drzezga A, Timmermann L, Eickhoff CR, Eickhoff SB, Eggers C. A systematic review on the applications of resting-state fMRI in Parkinson’s disease: Does dopamine replacement therapy play a role? Cortex. 2015;73:80–105.
Loubinoux I, Carel C, Pariente J, Dechaumont S, Albucher JF, Marque P, Manelfe C, Chollet F. Correlation between cerebral reorganization and motor recovery after subcortical infarcts. Neuroimage. 2003;20:2166–80.
Kwon YH, Lee MY. Changes in cortical activation patterns accompanying somatosensory recovery in a stroke patient: a functional magnetic resonance imaging study. Neural Regen Res. 2014;9:1485–8.
Chen L, Luo T, Wang K, Zhang Y, Shi D, Lv F, Li Y, Li Y, Li Q, Fang W, Zhang Z, Peng J, Yang H. Effects of thalamic infarction on the structural and functional connectivity of the ipsilesional primary somatosensory cortex. Eur Radiol. 2019;29:4904–13.
Funding
This study was supported by Sichuan Science and Technology Program (2019YJ0380), Key project Sichuan Provincial Department of Education (18ZA0211), Nanchong Science and Technology Program (NSMC20170433) and the doctoral scientific funds of North Sichuan Medical College (CBY16-QD04).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
M. He, J. Song, T. Luo, K. Wang, Y. Li, F. Lv and L. Chen declare that they have no competing interests.
Caption Electronic Supplementary Material
Rights and permissions
About this article
Cite this article
He, M., Song, J., Luo, T. et al. Alteration of Resting-state Functional Connectivity in the Sensorimotor Network in Patients with Thalamic Infarction. Clin Neuroradiol 31, 721–728 (2021). https://doi.org/10.1007/s00062-020-00966-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00062-020-00966-3