Abstract
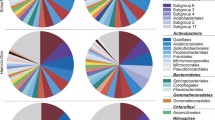
Soil pH has shown to predict bacterial diversity, but mechanisms are still poorly understood. To investigate how bacteria distribute themselves as a function of soil pH, we assessed community composition, diversity, assembly, and gene abundance across local (ca. 1 km) scale gradients in soil pH from ~ 3.8 to 6.5 created by differences in soil parent material in three northern forests. Plant species were the same on all sites, with no evidence of agriculture in the past. Concentrations of extractable calcium, iron, and phosphorus also varied significantly across the pH gradients. Among taxa, Alphaproteobacteria and Acidobacteria were more common in soils with acidic pH values. Overall richness and diversity of OTUs peaked at intermediate pH values. Variations in OTU richness and diversity also had a quadratic fit with concentrations of extractable calcium and phosphorus. Community assembly was via homogeneous deterministic processes in soils with acidic pH values, whereas stochastic processes dominated in soils with near-neutral pH values. Although we expected selection via genes for acid tolerance response in acidic soils, genes for genetic information processing were more selective. Taxa in higher pH soils had differential abundance of transporter genes, suggesting adaptation to acquire metabolic substrates from soils. Soil bacterial communities in northern forest soils are incredibly diverse, and we still have much to learn about how soil pH and co-varying soil parameters directly drive gene selection in this critical component of ecosystem structure.




Similar content being viewed by others
References
Beales N (2004) Adaptation of microorganisms to cold temperatures, weak acid preservatives, low pH, and osmotic stress: a review. Compr Rev Food Sci Food Saf 3:1–20
Fierer N, Jackson RB (2006) The diversity and biogeography of soil bacterial communities. Proc Natl Acad Sci U S A 103:626–631
Malik AA, Thomson BC, Whiteley AS, Bailey M, Griffiths RI (2017) Bacterial physiological adaptations to contrasting edaphic conditions identified using landscape scale metagenomics. MBio 8:e00799–e00717
Tripathi BM, Stegen JC, Kim M, Dong K, Adams JM, Lee YK (2018) Soil pH mediates the balance between stochastic and deterministic assembly of bacteria. The ISME journal 12:1072–1083
Lauber CL, Hamady M, Knight R, Fierer N (2009) Pyrosequencing-based assessment of soil pH as a predictor of soil bacterial community structure at the continental scale. Appl Environ Microbiol 75:5111–5120
Rousk J, Bååth E, Brookes PC, Lauber CL, Lozupone C, Caporaso JG, Knight R, Fierer N (2010) Soil bacterial and fungal communities across a pH gradient in an arable soil. ISME J 4:1340–1351
Krulwich TA, Sachs G, Padan E (2011) Molecular aspects of bacterial pH sensing and homeostasis. Nat Rev Microbiol 9:330–343
Talley K, Alexov E (2010) On the pH-optimum of activity and stability of proteins. Proteins 78:2699–2706
Kashket ER (1985) The proton motive force in bacteria: a critical assessment of methods. Annu Rev Microbiol 39:219–242
Lund P, Tramonti A, De Biase D (2014) Coping with low pH: molecular strategies in neutralophilic bacteria. FEMS Microbiol Rev 38:1091–1125
Xu Y, Zhao Z, Tong W, Ding Y, Liu B, Shi Y, Wang J, Sun S, Liu M, Wang Y, Qi Q (2020) An acid-tolerance response system protecting exponentially growing Escherichia coli. Nat Commun 11:1–3
Nguyen NL, Yu WJ, Gwak JH, Kim SJ, Park SJ, Herbold CW, Kim JG, Jung MY, Rhee SK (2018) Genomic insights into the acid adaptation of novel methanotrophs enriched from acidic forest soils. Front Microbiol 9:1982
Vergara E, Neira G, González C, Cortez D, Dopson M, Holmes DS (2020) Evolution of predicted acid resistance mechanisms in the extremely acidophilic Leptospirillum genus. Genes 11:389
Rao M, Streur TL, Aldwell FE, Cook GM (2001) Intracellular pH regulation by Mycobacterium smegmatis and Mycobacterium bovis BCG. Microbiology 147:1017–1024
Castanie-Cornet MP, Penfound TA, Smith D, Elliott JF, Foster JW (1999) Control of acid resistance in Escherichia coli. J Bacteriol 181:3525–3535
Pommerening-Röser A, Koops HP (2005) Environmental pH as an important factor for the distribution of urease positive ammonia-oxidizing bacteria. Microbiol Res 160:27–35
Gong L, Ren C, Xu Y (2020) GlnR negatively regulates glutamate-dependent acid resistance in Lactobacillus brevis. Appl Environ Microbiol 86:e02615–e02619
Barnett SE, Youngblut ND, Buckley DH (2020) Soil characteristics and land-use drive bacterial community assembly patterns. FEMS Microbiol Ecol 96:fiz194
Chase JM, Myers JA (2011) Disentangling the importance of ecological niches from stochastic processes across scales. Philos Trans R Soc London Ser B 366:2351–2363
Nemergut DR, Schmidt SK, Fukami T, O'Neill SP, Bilinski TM, Stanish LF, Knelman JE, Darcy JL, Lynch RC, Wickey P, Ferrenberg S (2013) Patterns and processes of microbial community assembly. Microbiol Mol Biol Rev 77:342–356
Zhou J, Ning D (2017) Stochastic community assembly: does it matter in microbial ecology? Microbiol Mol Biol Rev 81:e00002–e00017
Driscoll CT, Lawrence GB, Bulger AJ et al (2001) Acidic deposition in the northeastern United States: sources and inputs, ecosystem effects, and management strategies: the effects of acidic deposition in the northeastern United States include the acidification of soil and water, which stresses terrestrial and aquatic biota. BioScience 51:180–198
Buckley DH, Schmidt TM (2001) The structure of microbial communities in soil and the lasting impact of cultivation. Microb Ecol 42:11–21
Ogilvie IH (1902) Glacial phenomena in the Adirondacks and Champlain Valley. J Geol 10:397–412
Dyer JM (2006) Revisiting the deciduous forests of eastern North America. BioScience 56:341–352
Bailey SW, Brousseau PA, McGuire KJ, Ross DS (2014) Influence of landscape position and transient water table on soil development and carbon distribution in a steep, headwater catchment. Geoderma 226:279–289
Darby BA, Goodale CL, Chin NA, Fuss CB, Lang AK, Ollinger SV, Lovett GM (2020) Depth patterns and connections between gross nitrogen cycling and soil exoenzyme activities in three northern hardwood forests. Soil Biol Biochem 147:107836
Paratley RD, Fahey TJ (1986) Vegetation-environment relations in a conifer swamp in central New York. Bull Torrey Bot Club 113:357–371
Shanley JB, Sundquist ET, Kendall C (1995) Water, energy, and biogeochemical budget research at Sleepers River Research Watershed, Vermont. US Geological Survey
Christopher SF, Page BD, Campbell J, Mitchell MJ (2006) Contrasting stream water NO3− and Ca2+ in two nearly adjacent catchments: the role of soil Ca and forest vegetation. Glob Chang Biol 12:364–381
Karp NA, Lilley KS (2009) Investigating sample pooling strategies for DIGE experiments to address biological variability. Proteomics 9:388–397
Mehlich A (1984) Mehlich 3 soil test extractant: a modification of Mehlich 2 extractant. Commun Soil Sci Plant Anal 15:1409–1416
Kozich JJ, Westcott S, Baxter NT, Highlander SK, Schloss PD (2013) Development of a dual-index sequencing strategy and curation pipeline for analyzing amplicon sequence data on the MiSeq Illumina sequencing platform. Appl Environ Microbiol 79:5112–5120
Pepe-Ranney C, Campbell AN, Koechli CN, Berthrong S, Buckley DH (2016) Unearthing the ecology of soil microorganisms using a high resolution DNA-SIP approach to explore cellulose and xylose metabolism in soil. Front Microbiol 7:703
Zhang J, Kobert K, Flouri T, Stamatakis A (2014) PEAR: a fast and accurate Illumina Paired-End reAd mergeR. Bioinformatics 30:614–620
Schloss PD, Westcott SL, Ryabin T, Hall JR, Hartmann M, Hollister EB, Lesniewski RA, Oakley BB, Parks DH, Robinson CJ, Sahl JW, Stres B, Thallinger GG, van Horn D, Weber CF (2009) Introducing mothur: open-source, platform-independent, community-supported software for describing and comparing microbial communities. Appl Environ Microbiol 75:7537–7541
Edgar RC (2010) Search and clustering orders of magnitude faster than BLAST. Bioinformatics 26:2460–2461
Caporaso JG, Kuczynski J, Stombaugh J, Bittinger K, Bushman FD, Costello EK, Fierer N, Peña AG, Goodrich JK, Gordon JI, Huttley GA, Kelley ST, Knights D, Koenig JE, Ley RE, Lozupone CA, McDonald D, Muegge BD, Pirrung M, Reeder J, Sevinsky JR, Turnbaugh PJ, Walters WA, Widmann J, Yatsunenko T, Zaneveld J, Knight R (2010) QIIME allows analysis of high-throughput community sequencing data. Nat Methods 7:335–336
Knight R, Maxwell P, Birmingham A et al (2007) PyCogent: a toolkit for making sense from sequence. Genome Biol 8:R171
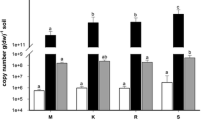
Nadeau SA, Roco CA, Debenport SJ, Anderson TR, Hofmeister KL, Walter MT, Shapleigh JP (2019) Metagenomic analysis reveals distinct patterns of denitrification gene abundance across soil moisture, nitrate gradients. Environ Microbiol 21:1255–1266
Roco CA, Dörsch P, Booth JG, Pepe-Ranney C, Groffman PM, Fahey TJ, Yavitt JB, Shapleigh JP (2019) Using metagenomics to reveal landscape scale patterns of denitrifiers in a montane forest ecosystem. Soil Biol Biochem 138:107585
Gotelli NJ, Colwell RK (2011) Estimating species richness. In: Magurran AE, McGill BJ (eds) Biological diversity: frontiers in measurement and assessment. Oxford University Press, Oxford, pp 39–54
Chao A, Gotelli NJ, Hsieh TC, Sander EL, Ma KH, Colwell RK, Ellison AM (2014) Rarefaction and extrapolation with Hill numbers: a framework for sampling and estimation in species diversity studies. Ecol Monogr 84:45–67
Camargo JA (1993) Must dominance increase with the number of subordinate species in competitive interactions? J Theor Biol 161:537–542
Stegen JC, Lin X, Konopka AE, Fredrickson JK (2012) Stochastic and deterministic assembly processes in subsurface microbial communities. ISME J 6:1653–1664
Oksanen J, Blanchet FG, Kindt R, Legendre P, Minchin PR, O’hara RB, Simpson GL, Solymos P, Stevens MH, Wagner H (2018) vegan: Community ecology package
R Core Team (2018) R: A Language and Environment for Statistical Computing
Kembel SW, Cowan PD, Helmus MR, Cornwell WK, Morlon H, Ackerly DD, Blomberg SP, Webb CO (2010) Picante: {R} tools for integrating phylogenies and ecology. Bioinformatics 26:1463–1464
Levins R (1968) Evolution in changing environments. Princeton University Press, Princeton
Pandit SN, Kolasa J, Cottenie K (2009) Contrasts between habitat generalists and specialists: an empirical extension to the basic metacommunity framework. Ecology 90:2253–2262
Kim J, Kim MS, Koh AY, Xie Y, Zhan X (2016) FMAP: functional mapping and analysis pipeline for metagenomics and metatranscriptomics studies. BMC Bioinformatics 17:420
Buchfink B, Xie C, Huson DH (2015) Fast and sensitive protein alignment using DIAMOND. Nat Methods 12:59–60
Kanehisa M, Goto S, Kawashima S, Okuno Y, Hattori M (2004) The KEGG resource for deciphering the genome. Nucleic Acids Res 32(suppl_1):D277–D280
Kanehisa M, Araki M, Goto S, Hattori M, Hirakawa M, Itoh M, Katayama T, Kawashima S, Okuda S, Tokimatsu T, Yamanishi Y (2007) KEGG for linking genomes to life and the environment. Nucleic Acids Res 36:D480–D484
Nakagawa S, Cuthill IC (2007) Effect size, confidence interval and statistical significance: a practical guide for biologists. Biol Rev 82:591–605
Dalman MR, Deeter A, Nimishakavi G, Duan ZH (2012) Fold change and p-value cutoffs significantly alter microarray interpretations. BMC Bioinformatics 13:S11
Kaiser K, Wemheuer B, Korolkow V, Wemheuer F, Nacke H, Schöning I, Schrumpf M, Daniel R (2016) Driving forces of soil bacterial community structure, diversity, and function in temperate grasslands and forests. Sci Rep 6:33696
Baker SR, Watmough SA, Eimers MC (2015) Phosphorus forms and response to changes in pH in acid-sensitive soils on the Precambrian Shield. Can J Soil Sci 95:95–108
Kabata-Pendias A (2010) Trace elements in soils and plants. CRC press, Boca Raton
Zhang Y, Shen H, He X, Thomas BW, Lupwayi NZ, Hao X, Thomas MC, Shi X (2017) Fertilization shapes bacterial community structure by alteration of soil pH. Front Microbiol 8:1325
Hurlbert SH (1971) The nonconcept of species diversity: a critique and alternative parameters. Ecology 52:577–586
Morris EK, Caruso T, Buscot F, Fischer M, Hancock C, Maier TS, Meiners T, Müller C, Obermaier E, Prati D, Socher SA (2104) Choosing and using diversity indices: insights for ecological applications from the German Biodiversity Exploratories. Ecol Evol 4:3514–3524
MacDonald ZG, Nielsen SE, Acorn JH (2017) Negative relationships between species richness and evenness render common diversity indices inadequate for assessing long-term trends in butterfly diversity. Biodivers Conserv 26:617–629
Stirling G, Wilsey B (2001) Empirical relationships between species richness, evenness, and proportional diversity. Am Nat 158:286–299
Jost L (2006) Entropy and diversity. Oikos 113:363–375
Wang J, Meier S, Soininen J, Casamayor EO, Pan F, Tang X, Yang X, Zhang Y, Wu Q, Zhou J, Shen J (2017) Regional and global elevational patterns of microbial species richness and evenness. Ecography 40:393–402
Griffiths RI, Thomson BC, James P, Bell T, Bailey M, Whiteley AS (2011) The bacterial biogeography of British soils. Environ Microbiol 13:1642–1654
Brislawn CJ, Graham EB, Dana K, Ihardt P, Fansler SJ, Chrisler WB, Cliff JB, Stegen JC, Moran JJ, Bernstein HC (2019) Forfeiting the priority effect: turnover defines biofilm community succession. ISME J 13:1865–1877
Azuma Y, Ota M (2009) An evaluation of minimal cellular functions to sustain a bacterial cell. BMC Syst Biol 3:111
Rees DC, Johnson E, Lewinson O (2009) ABC transporters: the power to change. Nat Rev Mol Cell Biol 10:218–227
Andersson S, Nilsson SI (2001) Influence of pH and temperature on microbial activity, substrate availability of soil-solution bacteria and leaching of dissolved organic carbon in a mor humus. Soil Biol Biochem 33:1181–1191
Allison SD, Vitousek PM (2005) Responses of extracellular enzymes to simple and complex nutrient inputs. Soil Biol Biochem 37:937–944
Acknowledgments
This work was supported by a US Department of Agriculture (USDA), National Institute of Food and Agriculture (NIFA), Hatch project. Any opinions, findings, conclusions, or recommendations expressed in this publication are those of the author(s) and do not necessarily reflect the view of NIFA or the USDA. We thank the ad hoc reviewers for comments and suggestions that greatly improved the clarity of the presentation.
Funding
This work was supported by a US Department of Agriculture (USDA), National Institute of Food and Agriculture (NIFA), Hatch project (1007286). Any opinions, findings, conclusions, or recommendations expressed in this publication are those of the author(s) and do not necessarily reflect the view of NIFA or the USDA.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of Interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
ESM 1
(DOCX 275 kb)
Rights and permissions
About this article
Cite this article
Yavitt, J.B., Roco, C.A., Debenport, S.J. et al. Community Organization and Metagenomics of Bacterial Assemblages Across Local Scale pH Gradients in Northern Forest Soils. Microb Ecol 81, 758–769 (2021). https://doi.org/10.1007/s00248-020-01613-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-020-01613-7