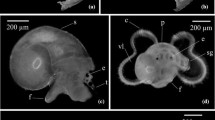
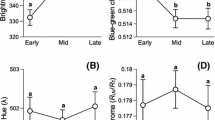
Abstract
Phenotypic polymorphisms are common in nature, but the mechanism through which natural selection maintains them can be difficult to identify. Heterozygote advantage, negative frequency-dependent selection, and variation in selective environment each can explain polymorphisms in some systems. Although first observed nearly 100 years ago, the egg mass color polymorphism in the spotted salamander (Ambystoma maculatum) still is not well understood. Females of this species lay egg masses with either clear or white jelly. White egg masses are preyed upon less than clear egg masses in some environments, suggesting an advantage to white masses, whereas an advantage to clear masses has not been identified. We propose that embryos in clear masses have an advantage in some light environments, mediated by the symbiotic green alga, Oophila amblystomatis, that grows in the egg capsules. The transparent jelly of clear masses may allow for greater algal growth, which could have cascading effects on embryonic development and larval performance. We asked whether larvae from clear and white masses differ in performance or survivorship and, if so, whether this difference is mediated by differences in algal density. We paired clear and white masses in a pond during the embryonic period and quantified egg capsule algal density at hatching. A portion of the larvae were subject to swim trials and were measured for size and shape. The remaining larvae were placed in mesocosms to quantify survival in the presence and absence of newt predators. There was no difference in algal density between membranes of clear and white masses. Similarly, there were no differences in larval performance, morphology, or survivorship between morphs. Our data do not support our hypothesis that variation in predator density and light availability among microhabitats could maintain the two phenotypes within populations.





Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available in the Dryad Digital Repository, https://doi.org/10.5061/dryad.69p8cz8xq.
References
Arendt JD (2009) Influence of sprint speed and body size on predator avoidance in New Mexican spadefoot toads (Spea multiplicata). Oecologia 159:455–461. https://doi.org/10.1007/s00442-008-1210-z
Arendt JD, Reznick DN (2005) Evolution of juvenile growth rates in female guppies (Poecilia reticulata): predator regime or resource level? Proc R Soc B Biol Sci 272:333–337
Banta AM, Gortner RA (1914) A milky white amphibian egg jell. Biol Bull 27:259–261. https://doi.org/10.2307/1536185
Bianchini K, Tattersall GJ, Sashaw J et al (2012) Acid water interferes with salamander-green algae symbiosis during early embryonic development. Physiol Biochem Zool 85:470–480. https://doi.org/10.1086/667407
Bishop JA (1972) An experimental study of the cline of industrial melanism in Biston betularia (L.) between urban Liverpool and rural North Wales. J Anim Ecol 41:209–243
Bishop CD, Miller AG (2014) Dynamics of the growth, life history transformation and photosynthetic capacity of Oophila amblystomatis (Chlorophyceae), a green algal symbiont associated with embryos of the northeastern yellow spotted salamander Ambystoma maculatum (Amphibia). Symbiosis 63:47–57. https://doi.org/10.1007/s13199-014-0287-x
Borash DJ, Gibbs AG, Joshi A, Mueller LD (1998) A genetic polymorphism maintained by natural selection in a temporally varying environment. Am Nat 151:148–156. https://doi.org/10.1086/286108
Boyd AE, Preusser A (2016) Vascular flora of the Sandy Bottom Wetland Preserve, Buncombe County, North Carolina. Castanea 81:323–332. https://doi.org/10.2179/16-094
Bright KL, Rausher MD (2008) Natural selection on a leaf-shape polymorphism in the ivy leaf morning glory (Ipomoea hederacea). Evolution 62:1978–1990. https://doi.org/10.1111/j.1558-5646.2008.00416.x
Burns JA, Kerney R, Duhamel S (2020) Heterotrophic carbon fixation in a salamander-alga symbiosis. Front Microbiol 11:1815. https://doi.org/10.3389/fmicb.2020.01815
Cain AJ, Sheppard PM (1950) Selection in the polymorphic land snail Cepaea nemoralis. Heredity 4:275–294
Cain AJ, Cook LM, Currey JD (1990) Population size and morph frequency in a long-term study of Cepaea nemoralis. Proc R Soc Lond B 240:231–250
Carey K, Ganders FR (1980) Heterozygote advantage at the fruit wing locus in Plectoritis congesta (Valerianaceae). Evolution 34:601–607
Cook LM (2008) Variation with habitat in Cepaea nemoralis: the Cain & Sheppard diagram. J Molluscan Stud 74:239–243. https://doi.org/10.1093/mollus/eyn011
Cook LM, Saccheri IJ (2013) The peppered moth and industrial melanism: evolution of a natural selection case study. Heredity 110:207–212
Cook LM, Grant BS, Saccheri IJ, Mallet J (2012) Selective bird predation on the peppered moth: the last experiment of Michael Majerus. Biol Lett 8:609–612. https://doi.org/10.1098/rsbl.2011.1136
Dykens BYJA, Shick JM, Benoit C et al (1992) Oxygen radical production in the sea anemone Anthopleura elegantissima and its endosymbiotic algae. J Exp Biol 168:219–241
Ferrari MCO, Messier F, Chivers DP (2008) Larval amphibians learn to match antipredator response intensity to temporal patterns of risk. Behav Ecol 19:980–983. https://doi.org/10.1093/beheco/arn056
Fitzpatrick BM, Benard MF, Fordyce JA (2003) Morphology and escape performance of tiger salamander larvae (Ambystoma tigrinum mavortium). J Exp Zool Part A Comp Exp Biol 297A:147–159. https://doi.org/10.1002/jez.a.10254
Furla P, Allemand D, Shick JM et al (2005) The symbiotic anthozoan: a physiological chimera between alga and animal. Integr Comp Biol 45:595–604. https://doi.org/10.1093/icb/45.4.595
Gilbert PW (1942) Observations on the eggs of Ambystoma maculatum with especial reference to the green algae found within the egg envelopes. Ecology 23:215–227
Gilbert PW (1944) The alga-egg relationship in Ambystoma maculatum, a case of symbiosis. Ecology 25:366–369
Goff L, Stein J (1976) Preliminary studies on the green alga Oophila in salamander egg masses. J Phycol 12:23–23
Graham ER, Fay SA, Davey A, Sanders RW (2013) Intracapsular algae provide fixed carbon to developing embryos of the salamander Ambystoma maculatum. J Exp Biol 216:452–459. https://doi.org/10.1242/jeb.076711
Graham ER, McKie-Krisberg ZM, Sanders RW (2014) Photosynthetic carbon from algal symbionts peaks during the latter stages of embryonic development in the salamander Ambystoma maculatum. BMC Res Notes 7:764. https://doi.org/10.1186/1756-0500-7-764
Gregoire DR, Gunzburger MS (2008) Effects of predatory fish on survival and behavior of larval gopher frogs (Rana capito) and southern leopard frogs (Rana sphenocephala). J Herpetol 42:97–103. https://doi.org/10.1670/07-039.1
Gulisija D, Kim Y, Plotkin JB (2016) Phenotypic plasticity promotes balanced polymorphism in periodic environments by a genomic storage effect. Genetics 202:1437–1448. https://doi.org/10.1534/genetics.115.185702
Hale RE, Kennedy C, Winkelman D, Brown C (2017) An advantage of clear over white egg mass morphs in metabolically demanding microhabitats suggests a role of symbiotic algae in the maintenance of a polymorphism in the spotted salamander (Ambystoma maculatum). Evol Ecol Res 18:637–650
Halliwell B, Gutteridge JMC (1999) Free radicals in biology and medicine, 3rd edn. Oxford University Press, Oxford
Hardy LM, Lucas MC (1991) A crystalline protein is responsible for dimorphic egg jellies in the spotted salamander, Ambystoma maculatum (Shaw) (Caudata: Ambystomatidae). Comp Biochem Physiol Part A Physiol 100:653–660. https://doi.org/10.1016/0300-9629(91)90385-P
Harrison RG (1969) Harrison stages and description of the normal development of the spotted salamander, Ambystoma punctatum (Linn.). In: Wilens S (ed) Organization and Development of the Embryo. Yale University Press, New Haven, pp 44–66
Heath DJ (1975) Colour, sunlight and internal temperatures in the land-snail Cepaea nemoralis (L.). Oecologia 19:29–38
Hector KL, Bishop PJ, Nakagawa S (2012) Consequences of compensatory growth in an amphibian. J Zool 286:93–101. https://doi.org/10.1111/j.1469-7998.2011.00850.x
Hedrick PW (2006) Genetic polymorphism in heterogeneous environments: the age of genomics. Annu Rev Ecol Evol Syst 37:67–93. https://doi.org/10.1146/annurev.ecolsys.37.091305.110132
Hedrick PW, Ginevan ME, Ewing EP (1976) Genetic polymorphism in heterogeneous environments. Annu Rev Ecol Syst 7:1–32
Hoekstra HE, Drumm KE, Nachman MW (2004) Ecological genetics of adaptive color polymorphism in pocket mice: geographic variation in selected and neutral genes. Evolution 58:1329–1341
Jacobson C (2015) Predation and mutualism: conflicting selection pressures maintain spotted salamander (Ambystoma maculatum). Undergraduate thesis, Appalachian State University
Kats LB, Ferrer RP (2003) Alien predators and amphibian declines: review of two decades of science and the transition to conservation. Divers Distrib 9:99–110. https://doi.org/10.1046/j.1472-4642.2003.00013.x
Kerney R (2011) Symbioses between salamander embryos and green algae. Symbiosis 54:107–117. https://doi.org/10.1007/s13199-011-0134-2
Kerney R, Kim E, Hangarter RP et al (2011) Intracellular invasion of green algae in a salamander host. Proc Natl Acad Sci 108:6497–6502. https://doi.org/10.1073/pnas.1018259108
Kettlewell HBD (1955) Selection experiments on industrial melanism in the Lepidoptera. Heredity 9:323–342
King SL (2016) Predator and environmental effects on the polymorphic egg masses of spotted salamanders (Ambystoma maculatum). Undergraduate thesis, Appalachian State University
Koskella B, Lively CM (2009) Evidence for negative frequency-dependent selection during experimental coevolution of a freshwater snail and a sterilizing trematode. Evolution. https://doi.org/10.1111/j.1558-5646.2009.00711.x
Landberg T, Azizi E (2010) Ontogeny of escape swimming performance in the spotted salamander. Funct Ecol 24:576–587. https://doi.org/10.1111/j.1365-2435.2009.01662.x
Lushchak VI, Bagnyukova TV (2006) Effects of different environmental oxygen levels on free radical processes in fish. Comp Biochem Physiol Part B Biochem Mol Biol 144:283–289
Majerus M, Brunton C, Stalker J (2000) A bird’s eye view of the peppered moth. J Evol Biol 13:155–159
Mani GS (1980) A theoretical study of morph ratio clines with special reference to melanism in moths. Proc R Soc Lond Ser B Biol Sci 210:299–316
Marchinko KB, Matthews B, Arnegard ME et al (2014) Maintenance of a genetic polymorphism with disruptive natural selection in stickleback. Curr Biol 24:1289–1292. https://doi.org/10.1016/j.cub.2014.04.026
Mills NE, Barnhart MC (1999) Effects of hypoxia on embryonic development in two Ambystoma and two Rana species. Physiol Biochem Zool 72:179–188. https://doi.org/10.1086/316657
Murillo-Rincón AP, Kolter NA, Laurila A, Orizaola G (2017) Intraspecific priority effects modify compensatory responses to changes in hatching phenology in an amphibian. J Anim Ecol 86:128–135. https://doi.org/10.1111/1365-2656.12605
Muto K, Nishikawa K, Kamikawa R, Miyashita H (2017) Symbiotic green algae in eggs of Hynobius nigrescens, an amphibian endemic to Japan. Phycol Res 65:171–174. https://doi.org/10.1111/pre.12173
Oksanen J, Blanchet FG, Friendly M, et al (2019) vegan: community ecology package v. 2.5-4
Ożgo M (2012) Shell polymorphism in the land-snail Cepaea nemoralis (L.) along a West-East transect in continental Europe. Folia Malacol 20:181–253. https://doi.org/10.2478/v10125-012-0015-1
Petranka JW (1998) Salamanders of the United States and Canada. Smithsonian Institution Press, Washington
Petranka JW, Just JJ, Crawford EC (1982) Hatching of amphibian embryos: the physiological trigger. Science 217:257–259
Petranka JW, Rushlow AW, Hopey ME (1998) Predation by tadpoles of Rana sylvatica on embryos of Ambystoma maculatum: implications of ecological role reversals by Rana (predator) and Ambystoma (prey). Herpetologica 54:1–13
Pinder AW, Friet SC (1994) Oxygen transport in egg masses of the amphibians Rana sylvatica and Ambystoma maculatum: convection, diffusion and oxygen production by algae. J Exp Biol 197:17–30
Pinheiro J, Bates D, DebRoy S, et al (2015) nlme: linear and nonlinear mixed effect models. R package version 31-122
Pintar MR, Resetarits WJ (2017) Persistence of an egg mass polymorphism in Ambystoma maculatum: differential performance under high and low nutrients. Ecology 98:1349–1360. https://doi.org/10.1002/ecy.1789
R Development Core Team (2018) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Richier S, Furla P, Plantivaux A et al (2005) Symbiosis-induced adaptation to oxidative stress. J Exp Biol 208:277–285. https://doi.org/10.1242/jeb.01368
Rowe CL, Sadinski WJ, Dunson WA (2006) Predation on larval and embryonic amphibians by acid-tolerant caddisfly larvae (Ptilostomis postica). J Herpetol 28:357. https://doi.org/10.2307/1564535
Rueden CT, Schindelin J, Hiner MC et al (2017) Image J2: ImageJ for the next generation of scientific image data. BMC Bioinform 18:529. https://doi.org/10.1186/s12859-017-1934-z
Ruth BC, Dunson WA, Rowe CL, Hedges SB (1993) A molecular and functional evaluation of the egg mass color polymorphism of the spotted salamander, Ambystoma maculatum. J Herpetol 27:306–314
Salthe S (1963) The egg capsules in the amphibia. J Morphol 113:161–171
Samollow PB, Soulé ME (1983) A case of stress related heterozygote superiority in nature. Evolution 37:646–649
Schmidt PS, Bertness MD, Rand DM et al (2000) Environmental heterogeneity and balancing selection in the acorn barnacle Semibalanus balanoides. Proc R Soc B Biol Sci 267:379–384
Sheppard PM (1951) Fluctuations in the selective value of certain phenotypes in the polymorphic land snail Cepaea nemoralis (L.). Heredity 5:125–134
Small DP, Bennett RS, Bishop CD (2014) The roles of oxygen and ammonia in the symbiotic relationship between the spotted salamander Ambystoma maculatum and the green alga Oophila amblystomatis during embryonic development. Symbiosis 64:1–10. https://doi.org/10.1007/s13199-014-0297-8
Starnes SM, Kennedy CA, Petranka JW (2000) Sensitivity of embryos of southern Appalachian amphibians to ambient solar UV-B radiation. Conserv Biol 14:277–282. https://doi.org/10.1046/j.1523-1739.2000.98596.x
Stock MK, Metcalfe J (1987) Modulation of growth and metabolism of the chick embryo by a brief (72-hr) change in oxygen availability. J Exp Zool Suppl Publ Auspices Am Soc Zool Div Comp Physiol Biochem 1:351–356
Sun BJ, Wang TT, Pike DA et al (2014) Embryonic oxygen enhances learning ability in hatchling lizards. Front Zool 11:21. https://doi.org/10.1186/1742-9994-11-21
Svensson EI, Abbott J, Härdling R (2005) Female polymorphism, frequency dependence, and rapid evolutionary dynamics in natural populations. Am Nat 165:567–576. https://doi.org/10.1086/429278
Takahashi Y, Yoshimura J, Morita S, Watanabe M (2010) Negative frequency-dependent selection in female color polymorphism of damselfly. Evolution 64:3620–3628
Tattersall GJ, Spiegelaar N (2008) Embryonic motility and hatching success of Ambystoma maculatum are influenced by a symbiotic alga. Can J Zool 86:1289–1298. https://doi.org/10.1139/Z08-115
Urban MC (2007) Risky prey behavior evolves in risky habitats. Proc Natl Acad Sci USA 104:14377–14382. https://doi.org/10.1073/pnas.0704645104
Urban MC, Lewis LA, Fučíková K, Cordone A (2015) Population of origin and environment interact to determine oomycete infections in spotted salamander populations. Oikos 124:274–284. https://doi.org/10.1111/oik.01598
Valls JH, Mills NE (2007) Intermittent hypoxia in eggs of Ambystoma maculatum: embryonic development and egg capsule conductance. J Exp Biol 210:2430–2435. https://doi.org/10.1242/jeb.003541
van Buskirk J, McCollum SA (1999) Plasticity and selection explain variation in tadpole phenotype between ponds with different predator composition. Oikos 85:31–39
Walls S (1995) Differential vulnerability to predation and refuge use in competing larval salamanders. Oecologia 101:86–93
Warkentin KM (2011) Plasticity of hatching in amphibians: evolution, trade-offs, cues and mechanisms. Integr Comp Biol 51:111–127. https://doi.org/10.1093/icb/icr046
Watkins TB (1996) Predator-mediated selection on burst swimming performance in tadpoles of the Pacific tree frog, Pseudacris regilla. Physiol Zool 69:154–167
Wente WH, Phillips JB (2003) Fixed green and brown color morphs and a novel color-changing morph of the Pacific tree frog Hyla regilla. Am Nat 162:461–473. https://doi.org/10.1086/378253
Williams PJ, Brown JA (1991) Developmental changes in foraging-predator avoidance trade-offs in larval lumpfish Cyclopterus lumpus. Mar Ecol Prog Ser 76:53–60. https://doi.org/10.3354/meps076053
Wilson RS, Kraft PG, van Damme R (2005) Predator-specific changes in the morphology and swimming performance of larval Rana lessonae. Funct Ecol 19:238–244. https://doi.org/10.1111/j.1365-2435.2005.00958.x
Wood J, Goodwin O (1954) Observations on the abundance, food, and feeding behavior of the newt, Notophthalmus viridescens viridescens (Rafinesque), in Virginia. J Elisha Mitchell Sci Soc 70:27–30
Acknowledgements
The authors express their appreciation of Megan Kline, Ruvim But-Gusaim, Jacob Boone, Dr. James Perkins, and Dr. Graham Reynolds for assistance in data collection, data analysis, and presentation of findings. The authors also thank Dr. Ryan Kerney and an anonymous reviewer for detailed feedback on the manuscript. This research was conducted with permission of the University of North Carolina Asheville Endowment Fund Board, which owns the study site at Sandy Bottom Preserve, and with a Wildlife Collection License from the North Carolina Wildlife Resources Commission. This project was funded by the UNC Asheville Undergraduate Research Program awarded to M. D’Errico (Spring 2018).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Data were collected by MD, with assistance from CK and RH. Data were analyzed by RH. MD prepared the first manuscript draft, which was revised by CK and RH. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The research followed the guidelines of the NSF for responsible and ethical conduct of research, and of the Animal Welfare Act, with approval of the UNC Asheville Institutional Animal Care and Use Committee. The authors declare no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
D’Errico, M., Kennedy, C. & Hale, R.E. Egg mass polymorphism in Ambystoma maculatum is not associated with larval performance or survival, or with cell density of the algal symbiont Oophila amblystomatis. Evol Ecol 34, 981–997 (2020). https://doi.org/10.1007/s10682-020-10083-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10682-020-10083-9