Abstract
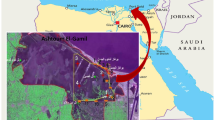
A deep investigation on the trophic structure of a fish assemblage in a reference condition river is important since allow the comparison and determination of the extent in which other sites are degraded. It is especially relevant when we consider a reference condition river located in a basin under influence of several anthropogenic activities, as is the case of the Rio Cipó, an important tributary of the polluted Rio das Velhas Basin, Brazil. We aimed to characterize the trophic structure of the fish assemblage of the Rio Cipó using the carbon (δ13C) and nitrogen (δ15N) stable isotopes of fish and several available food resources sampled at two sites along the Rio Cipó in the dry and wet seasons. We found that the δ15N of fishes change among trophic guilds, but not change between sites and seasons. The δ13C values in turn, varies among trophic guilds and between seasons, and the changes between sites are not very expressive. Detritivorous fish presented the greatest variation in δ13C values. Although all detritivorous species appeared to consume the same basal resources (algae, periphyton, POM and CPOM), the proportion of assimilation of each resource varied among them. Two individuals of Myleus micans (herbivorous) and two of Piabarchus stramineus (omnivorous), were found to differ substantially within the food web by presenting the most enriched values of δ15N and δ13C in the wet season at the site close to the confluence with other rivers. These outliers had influence in the broad isotopic niches of herbivorous and omnivorous fish. This result suggest that these species have a large spatial range and may be feeding in places with resources enriched in δ15N and δ13C, outside of the Rio Cipó, as well as highlights the importance of considering the influence of downstream polluted areas on the trophic structure of this reference condition river.





Similar content being viewed by others
References
Abelha MCF, Agostinho AA, Goulart E (2001) Plasticidade trófica em peixes de água doce. Acta Sci Biol Sci 23:425–434. https://doi.org/10.4025/actascibiolsci.v23i0.2696
Adams J (1985) The definition and interpretation of guild structure in ecological communities. J Anim Ecol 54:43. https://doi.org/10.2307/4619
Albrecht MP, Silveira CM (2001) Alimentação de Loricariichthys anus nas lagoas Marcelino e Peixoto Planicie Costeira do RS.pdf. Acta Limnol Bras 13:7985
Alonso MB, de Carvalho DR, Alves CBM, Moreira MZ, Pompeu PS (2019) Changes in trophic characteristics of two fish species of Astyanax (Teleostei: Characidae) in response to aquatic pollution. Zoologia 36:1–12. https://doi.org/10.3897/zoologia.36.e30445
Alves CBM, Pompeu PS (2005) Historical changes in the Rio das Velhas fish Fauna—Brazil. Am Fish Soc Symp 45:587–602
Alves CBM, Pompeu PS (2010) A fauna de peixes da bacia do rio das Velhas no final do século XX. In: Alves CBM, Pompeu PS (eds) Peixes do rio das Velhas: passado e presente. ARGVMENTVM, Belo Horizonte, pp 167–189
Alves C, Leal C, Brito M, Santos A (2008) Biodiversidade e conservação de peixes do Complexo do Espinhaço. Megadiversidade 4:201–220
Anderson C, Cabana G (2007) Estimating the trophic position of aquatic consumers in river food webs using stable nitrogen isotopes. J N Am Benthol Soc 26:273–285. https://doi.org/10.1899/0887-3593(2007)26[273:ETTPOA]2.0.CO;2
Arias-González J, Morand S (2006) Trophic functioning with parasites: a new insight for ecosystem analysis. Mar Ecol Prog Ser 320:43–53. https://doi.org/10.3354/meps320043
Barbosa FAR, Callisto M, Galdean N (2001) The diversity of benthic macroinvertebrates as an indicator of water quality and ecosystem health: a case study for Brazil. Aquat Ecosyst Health Manag 4:51–59. https://doi.org/10.1080/146349801753569270
Barrie A, Prosser SJ (1996) Mass spectrometry of soils. In: mass spectrometry of soils. Marcel Dekker, Inc., p 536
Baumgartner SD, Robinson CT (2017) Changes in macroinvertebrate trophic structure along a land-use gradient within a lowland stream network. Aquat Sci 79:407–418. https://doi.org/10.1007/s00027-016-0506-z
Bearhop S, Adams CE, Waldron S et al (2004) Determining trophic niche width: a novel approach using stable isotope analysis. J Anim Ecol 73:1007–1012. https://doi.org/10.1111/j.0021-8790.2004.00861.x
Bouleau G, Pont D (2015) Did you say reference conditions? Ecological and socio-economic perspectives on the European water framework directive. Environ Sci Pol 47:32–41. https://doi.org/10.1016/j.envsci.2014.10.012
Buss DF, Carlisle DM, Chon TS, Culp J, Harding JS, Keizer-Vlek HE, Robinson WA, Strachan S, Thirion C, Hughes RM (2015) Stream biomonitoring using macroinvertebrates around the globe: a comparison of large-scale programs. Environ Monit Assess 187:4132. https://doi.org/10.1007/s10661-014-4132-8
Casatti L, Langeani F, Castro RMC (2002) Peixes de riacho do parque estadual Morro do Diabo, Bacia do Alto Rio Paraná, SP. Biota Neotrop 1:1–15. https://doi.org/10.1590/s1676-06032001000100005
Casatti L, de Paula FC, Carvalho FR (2009) Grass-dominated stream sites exhibit low fish species diversity and dominance by guppies: an assessment of two tropical pasture river basins. Hydrobiologia 632:273–283. https://doi.org/10.1007/s10750-009-9849-y
Castro DMP, De Carvalho DR, Pompeu PDS et al (2016) Land use influences niche size and the assimilation of resources by benthic macroinvertebrates in tropical headwater streams. PLoS One 11:e0150527. https://doi.org/10.1371/journal.pone.0150527
Cataldo D (2015) Trophic relationships of Limnoperna Fortunei with adult fishes. In: Boltovskoy D (ed) Limnoperna Fortunei. Springer International Publishing, Cham, pp 231–248
CBH Rio das Velhas 2016 Revista Rio das Velhas: A bacia do rio Cipó e a sua importância para a qualidade das águas do rio das Velhas 2016. Available at: http://cbhvelhas.org.br/noticias/a-bacia-do-rio-cipo-e-a-sua-importancia-para%E2%80%A8a-qualidade-das-aguas-do-rio-das-velhas/. Accessed May 2 2019
Cohen JE, Newman CM, Briand F (1985) A stochastic theory of community food webs. II. Individual webs. Proc R Soc London, Ser B 224:449–461. https://doi.org/10.1098/rspb.1985.0043
Davis AM, Blanchette ML, Pusey BJ et al (2012) Gut content and stable isotope analyses provide complementary understanding of ontogenetic dietary shifts and trophic relationships among fishes in a tropical river. Freshw Biol 57:2156–2172. https://doi.org/10.1111/j.1365-2427.2012.02858.x
de A Teixeira JL, Gurgel HDCB (2004) Dinâmica da nutrição e alimentação natural de Steindachnerina notonota (Pisces, Curimatidae), Açude de Riacho da Cruz, Rio Grande do Norte, Brasil. Rev Bras Zoociências 6:19–28
de Carvalho DR, Flecker AS, Alves CBM, Sparks JP, Pompeu PS (2019) Trophic responses to aquatic pollution of native and exotic livebearer fishes. Sci Total Environ 681:503–515. https://doi.org/10.1016/j.scitotenv.2019.05.092
de Carvalho DR, Alves CBM, Flecker AS et al (2020a) Using δ15N of periphyton and fish to evaluate spatial and seasonal variation of anthropogenic nitrogen inputs in a polluted Brazilian river basin. Ecol Indic 115:106372. https://doi.org/10.1016/j.ecolind.2020.106372
de Carvalho DR, Alves CBM, Moreira MZ, Pompeu PS (2020b) Trophic diversity and carbon sources supporting fish communities along a pollution gradient in a tropical river. Sci Total Environ 738:139878. https://doi.org/10.1016/j.scitotenv.2020.139878
de M Vazzoler AEA, Agostinho AA, Hahn NS (1997) A Planície de Inundação do Alto Rio Paraná: Aspectos físicos, biológicos e socioeconômicos. Nupélia, Maringá, EDUEM
Delariva R (2001) Relationship between morphology and diets of six neotropical loricariids. J Fish Biol 58:832–847. https://doi.org/10.1006/jfbi.2000.1499
DeNiro MJ, Epstein S (1978) Influence of diet on the distribution of carbon isotopes in animals. Geochim Cosmochim Acta 42:495–506. https://doi.org/10.1016/0016-7037(78)90199-0
DeNiro M, Epstein S (1981) Influence of diet on the distribution of nitrogen isotopes in animals. Geochim Cosmochim Acta 45:341–351
do Prado MR, de Carvalho DR, Alves CBM et al (2020) Convergent responses of fish belonging to different feeding guilds to sewage pollution. Neotrop Ichthyol 18:e190041. https://doi.org/10.1590/1982-0224-2019-0045
Echevarría G, González N (2018) Fish taxonomic and functional diversity in mesohabitats of the river Kakada, Caura National Park, Venezuela. Nat Conserv Res 3:21–39. https://doi.org/10.24189/ncr.2018.048
Feio MJ, Reynoldson TB, Ferreira V, Graça MAS (2007) A predictive model for freshwater bioassessment (Mondego River, Portugal). Hydrobiologia 589:55–68. https://doi.org/10.1007/s10750-006-0720-0
Feio MJ, Ferreira WR, Macedo DR, Eller AP, Alves CBM, França JS, Callisto M (2015) Defining and testing targets for the recovery of tropical streams based on macroinvertebrate communities and abiotic conditions. River Res Appl 31:70–84. https://doi.org/10.1002/rra.2716
Ferreira W, Paiva L, Callisto M (2011) Development of a benthic multimetric index for biomonitoring of a neotropical watershed. Braz J Biol 71:15–25. https://doi.org/10.1590/S1519-69842011000100005
Fiorentino F, Cicala D, Careddu G, Calizza E, Jona-Lasinio G, Rossi L, Costantini ML (2017) Epilithon δ15N signatures indicate the origins of nitrogen loading and its seasonal dynamics in a volcanic Lake. Ecol Indic 79:19–27. https://doi.org/10.1016/j.ecolind.2017.04.007
Fry B, Sherr EB (1984) δ13C measurements as indicators of carbon flow in marine and freshwater ecosystems. In: Contributions in Marine Science. pp. 13–47
Giora J, Fialho CB (2003) Biologia alimentar de Steindachnerina brevipinna do rio Ibicuí-Mirim, Rio Grande do Sul, Brasil. Iheringia, Sér Zool Porto Alegre 93:277–281
Gomiero LM, de S Braga FM (2005) Uso do grau de preferência alimentar para a caracterização da alimentação de peixes na APA de São Pedro e Analândia. Acta Sci Biol Sci 27:265–270. https://doi.org/10.4025/actascibiolsci.v27i3.1337
Hesslein RH, Hallard KA, Ramlal P (1993) Replacement of sulfur, carbon, and nitrogen in tissue of growing broad whitefish (Coregonus nasus) in response to a change in diet traced by δ34S, δ13C, and δ15N. Can J Fish Aquat Sci 50:2071–2076. https://doi.org/10.1139/f93-230
Hidrotec. 2012. Atlas digital das águas de Minas. Ruralminas/Igam/UFV. Available at: http://www.atlasdasaguas.ufv.br. Accessed August 15 2013
Ibañez C, Tedesco PA, Bigorne R, Hugueny B, Pouilly M, Zepita C, Zubieta J, Oberdorff T (2007) Dietary-morphological relationships in fish assemblages of small forested streams in the Bolivian Amazon. Aquat Living Resour 20:131–142. https://doi.org/10.1051/alr:2007024
IBGE (2018) Estimativas de população. In: Inst. Bras. Geogr. e Estatística. https://www.ibge.gov.br/estatisticas-novoportal/sociais/populacao/9103-estimativas-de-populacao.html?=&t=resultados. Accessed 26 Feb 2019
ICMBio (2020) Espécies ameaçadas de extinção da fauna aquática da bacia do Rio São Francisco
IGAM - Instituto Mineiro de Gestão das águas (2016) Qualidade das águas superficiais de Minas Gerais em 2015. Belo Horizonte
IGAM - Instituto Mineiro de Gestão das águas (2017) Monitoramento de Qualidade das Águas. In: Monit. Qual. das Águas. http://portalinfohidro.igam.mg.gov.br/monitoramento-de-qualidade-das-aguas. Accessed 16 Jan 2019
Jackson AL, Inger R, Parnell AC, Bearhop S (2011) Comparing isotopic niche widths among and within communities: SIBER - stable isotope Bayesian ellipses in R. J Anim Ecol 80:595–602. https://doi.org/10.1111/j.1365-2656.2011.01806.x
Jepsen DB, Winemiller KO (2002) Structure of tropical river food webs revealed by stable isotope ratios. Oikos 96:46–55
Karr JR (1981) Assessment of biotic integrity using fish communities. Fisheries 6:21–27. https://doi.org/10.1577/1548-8446(1981)006<0021:AOBIUF>2.0.CO;2
Karr JR, Dudley DR (1981) Ecological perspective on water quality goals. Environ Manag 5:55–68. https://doi.org/10.1007/BF01866609
Layman CA, Arrington DA, Montaña CG, Post DM (2007) Can stable isotope ratios provide for community-wide measures of trophic structure? Ecology 88:42–48. https://doi.org/10.1890/0012-9658(2007)88[42:CSIRPF]2.0.CO;2
Leal CG, Junqueira NT, Alves CBM, Pompeu PS (2014) Morphological space stability in Rivers under different disturbance regimes. Copeia 2014:149–159. https://doi.org/10.1643/CE-12-096
Lobón-Cerviá J, Mazzoni R, Rezende CF (2016) Effects of riparian forest removal on the trophic dynamics of a Neotropical stream fish assemblage. J Fish Biol 89:50–64. https://doi.org/10.1111/jfb.12973
López-Fernández H, Winemiller KO (2005) Status of Dionda Diaboli and report of established populations of exotic fish species in lower San Felipe Creek, Val Verde County, Texas. Southwest Nat 50:246–251. https://doi.org/10.1894/0038-4909(2005)050[0246:soddar]2.0.co;2
Luczkovich JJ, Ward GP, Johnson JC, Christian RR, Baird D, Neckles H, Rizzo WM (2002) Determining the trophic guilds of fishes and macroinvertebrates in a seagrass food web. Estuaries 25:1143–1163. https://doi.org/10.1007/BF02692212
Martins I, Ligeiro R, Hughes RM, Macedo DR, Callisto M (2018) Regionalisation is key to establishing reference conditions for neotropical savanna streams. Mar Freshw Res 69:82. https://doi.org/10.1071/MF16381
Mazzoni R, Rezende C, Manna L (2010) Feeding ecology of Hypostomus punctatus Valenciennes, 1840 (Osteichthyes, Loricariidae) in a costal stream from Southeast Brazil. Braz J Biol 70:569–574. https://doi.org/10.1590/S1519-69842010000300013
McCutchan JH, Lewis WM, Kendall C, McGrath CC (2003) Variation in trophic shift for stable isotope ratios of carbon, nitrogen, and sulfur. Oikos 102:378–390. https://doi.org/10.1034/j.1600-0706.2003.12098.x
McMeans BC, Kadoya T, Pool TK et al (2019) Consumer trophic positions respond variably to seasonally fluctuating environments. Ecology 100:e02570. https://doi.org/10.1002/ecy.2570
Mill AC, Pinnegar JK, Polunin NVC (2007) Explaining isotope trophic-step fractionation: why herbivorous fish are different. Funct Ecol 21:1137–1145. https://doi.org/10.1111/j.1365-2435.2007.01330.x
Minas Gerais (1992) LEI No 10.629 de 16/01/1992. Brazil
MMA (2014) Portaria MMA No 445, de 17 de dezembro de 2014. Lista Nacional Oficial de Espécies da Fauna Ameaçadas de Extinção - Peixes e Invertebrados Aquáticos Mma 126–144
Moore JW, Semmens BX (2008) Incorporating uncertainty and prior information into stable isotope mixing models. Ecol Lett 11:470–480. https://doi.org/10.1111/j.1461-0248.2008.01163.x
Moreno P, França JS, Ferreira WR, Paz AD, Monteiro IM, Callisto M (2009) Use of the BEAST model for biomonitoring water quality in a neotropical basin. Hydrobiologia 630:231–242. https://doi.org/10.1007/s10750-009-9796-7
Morrissey CA, Boldt A, Mapstone A, Newton J, Ormerod SJ (2013) Stable isotopes as indicators of wastewater effects on the macroinvertebrates of urban rivers. Hydrobiologia 700:231–244. https://doi.org/10.1007/s10750-012-1233-7
Nagata R, Moreira M, Pimentel C, Morandini A (2015) Food web characterization based on δ15N and δ13C reveals isotopic niche partitioning between fish and jellyfish in a relatively pristine ecosystem. Mar Ecol Prog Ser 519:13–27. https://doi.org/10.3354/meps11071
Newsome SD, Rio CM del, Bearhop S, Phillips DL (2007) A niche for isotopic ecology. Front Ecol Environ 5:429–436. https://doi.org/10.1890/060150.01 T
Newsome SD, Yeakel JD, Wheatley PV, Tinker MT (2012) Tools for quantifying isotopic niche space and dietary variation at the individual and population level. J Mammal 93:329–341. https://doi.org/10.1644/11-MAMM-S-187.1
Nikolenko O, Jurado A, Borges AV, Knӧller K, Brouyѐre S (2018) Isotopic composition of nitrogen species in groundwater under agricultural areas: a review. Sci Total Environ 621:1415–1432. https://doi.org/10.1016/j.scitotenv.2017.10.086
Noble RAA, Cowx IG, Goffaux D, Kestemont P (2007) Assessing the health of European rivers using functional ecological guilds of fish communities: standardising species classification and approaches to metric selection. Fish Manag Ecol 14:381–392. https://doi.org/10.1111/j.1365-2400.2007.00575.x
Odum EP (1980) The status of three ecosystem-level hypotheses regarding salt marsh estuaries: tidal subsidy, Outwelling, and detritus-based food chains. Academic Press, INC.
Orlandi L, Calizza E, Careddu G, Carlino P, Costantini ML, Rossi L (2017) The effects of nitrogen pollutants on the isotopic signal (δ15N) of Ulva lactuca : microcosm experiments. Mar Pollut Bull 115:429–435. https://doi.org/10.1016/j.marpolbul.2016.12.051
Pardo I, Gómez-Rodríguez C, Wasson J-G, Owen R, van de Bund W, Kelly M, Bennett C, Birk S, Buffagni A, Erba S, Mengin N, Murray-Bligh J, Ofenböeck G (2012) The European reference condition concept: a scientific and technical approach to identify minimally-impacted river ecosystems. Sci Total Environ 420:33–42. https://doi.org/10.1016/j.scitotenv.2012.01.026
Parnell AC, Inger R, Bearhop S, Jackson AL (2010) Source partitioning using stable isotopes: coping with too much variation. PLoS One 5:e9672. https://doi.org/10.1371/journal.pone.0009672
Peipoch M, Martí E, Gacia E (2012) Variability in δ15N natural abundance of basal resources in fluvial ecosystems: a meta-analysis. Freshw Sci 31:1003–1015. https://doi.org/10.1899/11-157.1
Peterson BJ, Fry B (2012) Stable isotopes in ecosystem studies. Annu Rev Ecol Syst 18:293–320
Pompeu PS, Alves CBM, Callisto M (2005) The effects of urbanization on biodiversity and water quality in the Rio das Velhas Basin, Brazil. Am Fish Soc Symp 47:11–22
Post DM (2002) Using stable isotopes to estimate trophic position: models, methods, and assumptions. Ecology 83:703. https://doi.org/10.2307/3071875
Prejs A, Prejs K (1987) Feeding of tropical freshwater fishes: seasonality in resource availability and resource use. Oecologia 71:397–404. https://doi.org/10.1007/BF00378713
R Core team (2018) R: A Language and Environment for Statistical Computing
Rodrigues D (2013) Ecologia trófica das principais espécies de peixes do Rio Paraíba do Sul na área de influência do Aproveitamento Hidrelétrico Simplício durante o período pré- represamento. Universidade Federal de Juiz de Fora
Rybczynski SM, Walters DM, Fritz KM, Johnson BR (2008) Comparing trophic position of stream fishes using stable isotope and gut contents analyses. Ecol Freshw Fish 17:199–206. https://doi.org/10.1111/j.1600-0633.2007.00289.x
Sacramento PA, Manetta GI, Benedito E (2016) Diet-tissue discrimination factors ( Δ 13 C and Δ 15 N) and turnover rate in somatic tissues of a neotropical detritivorous fish on C 3 and C 4 diets. J Fish Biol 89:213–219. https://doi.org/10.1111/jfb.12859
Simberloff D, Dayan T (1991) The guild concept and the structure of ecological communities. Annu Rev Ecol Syst 22:115–143. https://doi.org/10.1146/annurev.es.22.110191.000555
Smith BN, Epstein S (1971) Two categories of 13 C/ 12 C ratios for higher plants. Plant Physiol 47:380–384. https://doi.org/10.1104/pp.47.3.380
Stefani PM (2010) Ecologia trófica e ecomorfologia de peixes em um trecho do Alto Rio São Francisco impactado pela transposição do Rio Piumhi, com ênfase nas espécies Pimelodus fur Lütken, 1874 e Leporinus reinhardti Lütken, 1875. UFSCar
Stock B, Semmens B (2016a) MixSIAR GUI user manual. Version 31:1–59. https://doi.org/10.5281/zenodo.47719
Stock BC, Semmens BX (2016b) Unifying error strucctures in commonly used biotracer mixing models. Ecology 97:576–582. https://doi.org/10.1002/ecy.1517
Teresa FB, De Souza LS, da Silva DMA et al (2016) Environmental constraints structuring fish assemblages in riffles: evidences from a tropical stream. Neotrop Ichthyol 14:1–6. https://doi.org/10.1590/1982-0224-20150185
Trajano E, Secutti S, Mattox GMT (2010) Epigean and subterranean ichthyofauna in Cordisburgo karst area, eastern Brazil. Biota Neotrop 9:277–281. https://doi.org/10.1590/s1676-06032009000300026
Uieda VS, Fujihara CY (2018) Structure of the ichthyofauna of adventitious streams in the south Pantanal. Iheringia Série Zool 108:1–8. https://doi.org/10.1590/1678-4766e2018019
Vander Zanden MJ, Rasmussen JB (1999) Primary consumer d13C and d15N and the trophic position of aquatic consumers. Ecology 80:1395–1404. https://doi.org/10.1890/0012-9658(1999)080[1395:pccana]2.0.co;2
Vander Zanden MJ, Rasmussen JB (2001) Variation in δ15N and δ13C trophic fractionation: implications for aquatic food web studies. Limnol Oceanogr 46:2061–2066. https://doi.org/10.4319/lo.2001.46.8.2061
Vander Zanden MJ, Cabana G, Rasmussen JB (1997) Comparing trophic position of freshwater fish calculated using stable nitrogen isotope ratios (δ15N) and literature dietary data. Can J Fish Aquat Sci 54:1142–1158. https://doi.org/10.1139/cjfas-54-5-1142
Vanderklift MA, Ponsard S (2003) Sources of variation in consumer-diet 15N enrichment: a meta-analysis. Oecologia 136:169–182. https://doi.org/10.1007/s00442-003-1270-z
Vieira F, Santos GB, Alves CBM (2005) A ictiofauna do Parque Nacional da Serra do Cipó (Minas Gerais) e áreas adjacentes. Lundiana 6:77–87
Villares-Junior GA, Cardone IB, Goitein R (2016) Comparative feeding ecology of four syntopic Hypostomus species in a Brazilian southeastern river. Braz J Biol 76:692–699. https://doi.org/10.1590/1519-6984.00915
Winemiller KO (1989) Ontogenetic diet shifts and resource partitioning among piscivorous fishes in the Venezuelan ilanos. Environ Biol Fish 26:177–199. https://doi.org/10.1007/BF00004815
Winemiller KO, Jepsen DB (1998) Effects of seasonality and fish movement on tropical river food webs. J Fish Biol 53:267–296. https://doi.org/10.1111/j.1095-8649.1998.tb01032.x
Zellhuber A, Siqueira R (2016) Rio São Francisco em descaminho: degradação e revitalização. In: Cadernos do CEAS: Revista crítica de humanidades. pp 3–24
Acknowledgments
We thank the Agência Peixe Vivo and the Comitê de Bacia Hidrográfica do Rio das Velhas CBH - Rio das Velhas for project financial support and the Projeto Manuelzão for the logistical support. This study was also financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior – Brasil (CAPES) – Finance code 001 and D.R.C. sandwich doctorate scholarship granted (88881.190508/2018-01). Thanks to Aline Junqueira Grossi (Federal University of Lavras, UFLA) and Luiza Hoehne (Federal University of Minas Gerais, UFMG) for the support on processing of samples. Thanks to the Benthos Ecology Laboratory (UFMG) and the Laboratory of Fish Ecology (UFLA) who assisted in the processing of samples and infrastructure, and to the Centre for Nuclear Energy in Agriculture (CENA) for their support and partnership in the isotopic analysis. P.S.P. received a research grant and a research fellowship from the Conselho Nacional de Desenvolvimento Científico e Tecnológico (303548/2017-7) and from the Fundação de Amparo à Pesquisa do Estado de Minas Gerais (PPM-00237/13).
Authors’ contributions conceptualization
Conceptualization: Mirella Bisso Alonso, Débora Reis de Carvalho, Carlos Bernardo Mascarenhas Alves, Paulo Santos Pompeu; Data collection: Carlos Bernardo Mascarenhas Alves; Material preparation: Mirella Bisso Alonso, Débora Reis de Carvalho; Formal analyses and investigation: Mirella Bisso Alonso, Débora Reis de Carvalho, Paulo Santos Pompeu; Writing - original draft preparation: Mirella Bisso Alonso, Débora Reis de Carvalho; Writing - review and editing: Carlos Bernardo Mascarenhas Alves, Paulo Santos Pompeu; Funding acquisition: Carlos Bernardo Mascarenhas Alves, Paulo Santos Pompeu; Supervision: Paulo Santos Pompeu.
Availability of data and material
The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
Funding
Agência Peixe Vivo; Comitê de Bacia Hidrográfica do Rio das Velhas CBH - Rio das Velhas; Coordenação de Aperfeiçoamento de Pessoal de Nível Superior – Brasil (88881.190508/2018–01); Conselho Nacional de Desenvolvimento Científico e Tecnológico (303548/2017–7); Fundação de Amparo à Pesquisa do Estado de Minas Gerais (PPM-00237/13).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflict of interest.
Ethics approval
The methodology for this study was approved by the Ethics committee of the Universidade Federal de Lavras (Federal University of Lavras). Ethics approval number: 070/15.
Consent to participate
(include appropriate statements) Not applicable.
Consent for publication
(include appropriate statements) Not applicable.
Code availability
(software application or custom code) Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 35 kb)
Rights and permissions
About this article
Cite this article
Alonso, M.B., de Carvalho, D.R., Alves, C.B.M. et al. Trophic structure of a fish assemblage in a reference condition river located in a polluted watershed. Environ Biol Fish 103, 1437–1452 (2020). https://doi.org/10.1007/s10641-020-01033-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10641-020-01033-z