Abstract
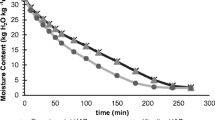
Chlorella sp. are among the highest cultivated species of microalgae. They are considered as an important functional food due to their abundance and valuable health benefits. It would be useful to develop an effective method for phenolic compound extraction from Chlorella sp. using safe technology and green solvents for the applications in food and pharmaceutical fields. Pressurized hot water extraction (PHWE) is an alternative for this purpose. PHWE was employed to extract phenolic compounds from Chlorella sp. microalgae at temperatures between 100 and 250 °C. The data collected is used to create the kinetics of the extraction process. The kinetics studies revealed that the extraction from Chlorella sp. microalgae using pressurized hot water treatment follows first-order kinetics and is strongly dependent on temperature. An increasing trend in the calculated values of mass transfer coefficient, k, was noticed, with increasing temperature. The activation energy, Ea, was calculated as 11.146 kJ/mol for the extraction treatment.






Similar content being viewed by others
References
Joana Gil-Chávez G, Villa JA, Fernando Ayala-Zavala J, Basilio Heredia J, Sepulveda D, Yahia EM, González-Aguilar GA (2013) Technologies for extraction and production of bioactive compounds to be used as nutraceuticals and food ingredients: an overview. Compr Rev Food Sci Food Saf 12(1):5–23
West MB, Zubeck HM (2012) Evaluation of microalgae for use as nutraceuticals and nutritional supplements. J Nutr Food Sci 2(5):1000147. https://doi.org/10.4172/2155-9600.1000147
Lam KS (2007) New aspects of natural products in drug discovery. Trends Microbiol 15(6):279–289
Konishi F, Tanaka K, Himeno K, Taniguchi K, Nomoto K (1985) Antitumor effect induced by a hot water extract of Chlorella vulgaris (CE): resistance to Meth-A tumor growth mediated by CE-induced polymorphonuclear leukocytes. Cancer Immunol Immunother 19(2):73–78
Miranda MS, Sato S, Mancini-Filho J (2001) Antioxidant activity of the microalga Chlorella vulgaris cultered on special conditions. Boll Chim Farm 140(3):165–168
Hasegawa T, Tanaka K, Ueno K, Ueno S, Okuda M, Yoshikai Y, Nomoto K (1989) Augmentation of the resistance against Escherichia coli by oral administration of a hot water extract of Chlorella vulgaris in rats. Int J Immunopharmacol 11(8):971–976
Knekt P, Kumpulainen J, Järvinen R, Rissanen H, Heliövaara M, Reunanen A, Hakulinen T, Aromaa A (2002) Flavonoid intake and risk of chronic diseases. Am J Clin Nutr 76(3):560–568
Klejdus B, Kopecký J, Benešová L, Vacek J (2009) Solid-phase/supercritical-fluid extraction for liquid chromatography of phenolic compounds in freshwater microalgae and selected cyanobacterial species. J Chromatogr A 1216(5):763–771
Suárez B, Álvarez ÁL, García YD, Barrio Gd, Lobo AP, Parra F (2010) Phenolic profiles, antioxidant activity and in vitro antiviral properties of apple pomace. Food Chem 120(1):339–342
Sun C, Wu Z, Wang Z, Zhang H (2015) Effect of ethanol/water solvents on phenolic profiles and antioxidant properties of Beijing propolis extracts. Evid Based Complement Alternat Med 2015:9
Zakaria SM, Kamal SMM (2015) Subcritical water extraction of bioactive compounds from plants and algae: applications in pharmaceutical and food ingredients. Food Eng Rev 8(1):23–34
Wataniyakul P, Pavasant P, Goto M, Shotipruk A (2012) Microwave pretreatment of defatted rice bran for enhanced recovery of total phenolic compounds extracted by subcritical water. Bioresour Technol 124(0):18–22
Khuwijitjaru P, Sayputikasikorn N, Samuhasaneetoo S, Penroj P, Siriwongwilaichat P, Adachi S (2012) Subcritical water extraction of flavoring and phenolic compounds from cinnamon bark (Cinnamomum zeylanicum). J Oleo Sci 61(6):349–355
Jo E-K, Heo D-J, Kim J-H, Lee Y-H, Ju Y-C, Lee S-C (2013) The effects of subcritical water treatment on antioxidant activity of golden oyster mushroom. Food Bioprocess Technol 6(9):2555–2561
Xu H, Wang W, Jiang J, Yuan F, Gao Y (2014) Subcritical water extraction and antioxidant activity evaluation with on-line HPLC-ABTS·+ assay of phenolic compounds from marigold (Tagetes erecta L.) flower residues. J Food Sci Technol 52(6):3803–3811
Azmir J, Zaidul I, Rahman M, Sharif K, Mohamed A, Sahena F, Jahurul M, Ghafoor K, Norulaini N, Omar A (2013) Techniques for extraction of bioactive compounds from plant materials: a review. J Food Eng 117(4):426–436
Rakotondramasy-Rabesiaka L, Havet J-L, Porte C, Fauduet H (2007) Solid–liquid extraction of protopine from Fumaria officinalis L.—analysis determination, kinetic reaction and model building. Sep Purif Technol 54(2):253–261. https://doi.org/10.1016/j.seppur.2006.09.015
PureBulk (2018). Chlorella (Broken Cell Wall). Retrieved from https://purebulk.com/products/chlorella-broken-cell-wall
Cliffe S, Fawer MS, Maier G, Takata K, Ritter G (1994) Enzyme assays for the phenolic content of natural juices. J Agric Food Chem 42(8):1824–1828
Sulaiman S, Abdul Aziz AR, Kheireddine Aroua M (2013) Optimization and modeling of extraction of solid coconut waste oil. J Food Eng 114(2):228–234
Srinivas K, King JW, Howard LR, Monrad JK (2010) Solubility of gallic acid, catechin, and protocatechuic acid in subcritical water from (298.75 to 415.85) K. J Chem Eng Data 55(9):3101–3108
Kalinichev AG, Bass JD (1997) Hydrogen bonding in supercritical water. 2. Computer simulations. J Phys Chem A 101(50):9720–9727
Silva EM, Rogez H, Larondelle Y (2007) Optimization of extraction of phenolics from Inga edulis leaves using response surface methodology. Sep Purif Technol 55(3):381–387
Singh PP, Saldaña MDA (2011) Subcritical water extraction of phenolic compounds from potato peel. Food Res Int 44(8):2452–2458
Teo CC, Tan SN, Yong JWH, Hew CS, Ong ES (2010) Pressurized hot water extraction (PHWE). J Chromatogr A 1217(16):2484–2494
Plaza M, Turner C (2015) Pressurized hot water extraction of bioactives. TrAC Trends Anal Chem 71:39–54
Meziane S, Kadi H, Lamrous O (2006) Kinetic study of oil extraction from olive foot cake. Grasas Aceites 57(2):175–179. https://doi.org/10.3989/gya.2006.v57.i2.34
Kashif A, Muhammad SH, Joong-Ho K (2017) Green extraction methods for polyphenols from plant matrices and their byproducts: a review. Compr Rev Food Sci Food Saf 16(2):295–315
Ahmad A, Yasin NM, Derek C, Lim J (2014) Kinetic studies and thermodynamics of oil extraction and transesterification of Chlorella sp. for biodiesel production. Environ Technol 35(7):891–897
Paunović DĐ, Mitić SS, Kostić DA, Mitić MN, Stojanović BT, Pavlović JL (2014) Kinetics and thermodynamics of the solid-liquid extraction process of total polyphenols from barley. Adv Technol 3(2):58–63. https://doi.org/10.5937/savteh1402058P
Amin SK, Hawash S, El Diwani G, El Rafei S (2010) Kinetics and thermodynamics of oil extraction from Jatropha curcas in aqueous acidic hexane solutions. J Am Sci 6(11):293–300
El-Hefny NE (2017) Chemical kinetics and reaction mechanisms in solvent extraction: new trends and applications. J Phys Sci 28(1):129–156. https://doi.org/10.21315/jps2017.28.1.10
Santos SB d, Martins MA, Caneschi AL, Aguilar PRM, Coimbra JS d R (2015) Kinetics and thermodynamics of oil extraction from Jatropha curcas L. using ethanol as a solvent. Int J Chem Eng:Article ID 871236. https://doi.org/10.1155/2015/871236
Bucić-Kojić A, Planinić M, Tomas S, Bilić M, Velić D (2007) Study of solid–liquid extraction kinetics of total polyphenols from grape seeds. J Food Eng 81(1):236–242
Funding
The authors acknowledge the Universiti Putra Malaysia for the study and financial support from the Fundamental Research Grant Scheme (FRGS: 03-01-15-1627FR) provided by the Ministry of Higher Education Malaysia.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zakaria, S.M., Mustapa Kamal, S.M., Harun, M.R. et al. Extraction of phenolic compounds from Chlorella sp. microalgae using pressurized hot water: kinetics study. Biomass Conv. Bioref. 12, 2081–2089 (2022). https://doi.org/10.1007/s13399-020-01027-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13399-020-01027-y