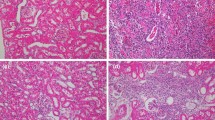
In male rats, acute renal failure was simulated by clamping the vascular pedicle of the left kidney for 60 or 90 min and right-sided nephrectomy. In the control series, no therapy was performed. In the experimental series, the animals were daily injected subcutaneously with Cellex, a protein-peptide complex (PPC) chromatographically isolated from the brain tissue of pig embryos with a molecular weight of its components from 10 to 250 kDa. PPC was administered 5 times a week (10 injections) in a dose of 0.1 ml/kg (0.1 mg active substance per 1 kg body weight). Ischemia of a single kidney led to the development of acute renal failure, more severe after 90-min ischemia. PPC therapy reduced the severity of functional disorders mainly at the early stages (3 and 7 days) with normalization of blood concentrations of urea and creatinine, creatinine clearance, tubular reabsorption of sodium and calcium, including the cases with 90-min ischemia, which did not occur in the control series. PPC therapy also contributed to hypertrophy of many glomeruli, prevented the development of glomerulosclerosis, and reduced damage to the epithelium of the renal tubules. At the same time, neither pronounced lymphohistiocytic infiltration, nor focal nephrosclerosis typical of control series were observed.
Similar content being viewed by others
References
Kamchatnov PR, Izmailov IA, Sokolov MA. Results of Cerebrovascular Diseases Treatment with Cellex. Nervn. Bol. 2018;(1):26-31. Russian.
Khasanova DP, Danilova TV, Demin TV, Knni KS, Gaifutdinova LV. The effect of Cellex on the recovery of motor and speech functions during early neurorehabilitation of patients with ischemic stroke. Med. Sovet. 2018;(9):14-19. Russian.
Beer L, Mildner M, Ankersmit HJ. Cell secretome based drug substances in regenerative medicine: when regulatory affairs meet basic science. Ann. Transl. Med. 2017;5(7):170. https://doi.org/10.21037/atm.2017.03.50
Bi B, Schmitt R, Israilova M, Nishio H, Cantley LG. Stromal cells protect against acute tubular injury via an endocrine effect. J. Am. Soc. Nephrol. 2007;18(9):2486-2496.
Dittmer J, Leyh B. Paracrine effects of stem cells in wound healing and cancer progression (Review). Int. J. Oncol. 2014; 44(6):1789-1798.
Fan M, Zhang J, Xin H, He X, Zhang X. Current perspectives on role of MSC in renal pathophysiology. Front. Physiol. 2018;9. ID 1323. https://doi.org/10.3389/fphys.2018.01323
Golle L, Gerth HU, Beul K, Heitplatz B, Barth P, Fobker M, Pavenstädt H, Di Marco GS, Brand M. Bone marrow-derived cells and their conditioned medium induce microvascular repair in uremic rats by stimulation of endogenous repair mechanisms. Sci. Rep. 2017;7(1):9444. https://doi.org/10.1038/s41598-017-09883-x
Humphreys BD, Czerniak S, DiRocco DP, Hasnain W, Cheema R, Bonventre JV. Repair of injured proximal tubule does not involve specialized progenitors. Proc. Natl Acad. Sci. USA. 2011;108(22):9226-9231.
Meyer-Schwesinger C. The role of renal progenitors in renal regeneration. Nephron. 2016;132(2):101-109.
Perico L, Morigi M, Rota C, Breno M, Mele C, Noris M, Introna M, Capelli C, Longaretti L, Rottoli D, Conti S, Corna D, Remuzzi G, Benigni A. Human mesenchymal stromal cells transplanted into mice stimulate renal tubular cells and enhance mitochondrial function. Nat. Commun. 2017;8(1):983. https://doi.org/10.1038/s41467-017-00937-2
Phelps J, Sanati-Nezhad A, Ungrin M, Duncan NA, Sen A. Bioprocessing of mesenchymal stem cells and their derivatives: toward cell-free therapeutics. Stem Cells Int. 2018;2018: 9415367. https://doi.org/10.1155/2018/9415367
Rota C, Morigi M, Imberti B. Stem cell therapies in kidney diseases: progress and challenges. Int. J. Mol. Sci. 2019;20(11). pii: E2790. https://doi.org/10.3390/ijms20112790
Rota C, Morigi M, Cerullo D, Introna M, Colpani O, Corna D, Capelli C, Rabelink T.J, Leuning D.G, Rottoli D, Benigni A, Zoja C, Remuzzi G. Therapeutic potential of stromal cells of non-renal or renal origin in experimental chronic kidney disease. Stem Cell Res. Ther. 2018;9(1):220. https://doi.org/10.1186/s13287-018-0960-8
Tögel F, Hu Z, Weiss K, Isaac J, Lange C, Westenfelder C. Administered mesenchymal stem cells protect against ischemic acute renal failure through differentiation-independent mechanisms. Am. J. Physiol. Renal Physiol. 2005;289(1):F31-F42.
Vizoso FJ, Eiro N, Cid S, Schneider J, Perez-Fernandez R. Mesenchymal stem cell secretome: toward cell-free therapeutic strategies in regenerative medicine. Int. J. Mol. Sci. 2017;18(9). pii: E1852. https://doi.org/10.3390/ijms18091852
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Byulleten’ Eksperimental’noi Biologii i Meditsiny, Vol. 169, No. 5, pp. 551-558, May, 2020
Rights and permissions
About this article
Cite this article
Kirpatovskii, V.I., Sivkov, A.V., Golovanov, S.A. et al. Stimulation of Structural and Functional Recovery of the Kidney in Rats with Postischemic Acute Renal Failure of Different Severity by Embryonic Protein-Peptide Complex Therapy. Bull Exp Biol Med 169, 623–629 (2020). https://doi.org/10.1007/s10517-020-04941-3
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10517-020-04941-3