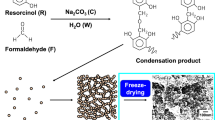
Abstract
We evaluated the performance of a resorcinol–formaldehyde (RF) hydrogel as an adsorbent for removing aqueous cesium ions (Cs+), by synthesizing cylindrical pellets of RF gels under different molar ratios (R/C) between resorcinol (R) and sodium carbonate (C) as the catalyst for the sol–gel polycondensation in the range from 2.5 to 400. The RF gel prepared at R/C = 2.5, containing the greatest amount of sodium ion (Na+), had the greatest Cs+ adsorption amount (0.660 mmol g−1), because Cs+ could be adsorbed on the network structure in the gel through exchange with Na+ at an equal molar ratio, but with the smallest effective diffusion coefficient of Cs+ (1.45 × 10–12 m2 s−1). Kinetic analyses based on the Dryden–Kay and intra-particle diffusion models revealed that the diffusivity of Cs+ in the RF gel depended strongly on the density of the network structure, which can be controlled by varying the R/C ratio.
Graphic abstract











Similar content being viewed by others
References
Arnal, J.M., Sancho, M., Verdú, G., Campayo, J.M., Villaescusa, J.I.: Treatment of 137Cs liquid wastes by reverse osmosis Part I. Preliminary tests. Desalination 154(1), 27–33 (2003)
Blanco, M.C., Leisner, D., Vázquez, C., López-Quintela, A.M.: Dynamic light scattering in transient reversible gel. Langmuir 16(23), 8585–8594 (2000)
Brotin, T., Montserret, R., Bouchet, A., Cavagnat, D., Linares, M., Buffeteau, T.: High affinity of water–soluble cryptophanes for cesium cations. J. Org. Chem. 77(2), 1198–1201 (2012)
Cabaud, C., Barré, Y., Windt, L.D., Gill, S., Dooryhée, E., Moloney, M.P., Massoni, N., Grandjean, A.: Removing Cs within a continuous flow set–up by an ionic exchanger material transformable into a final waste form. Adsorption 25, 765–771 (2019)
Dryden, C.E., Kay, W.B.: Kinetics of batch adsorption and desorption. Ind. Eng. Chem. 46(11), 2294–2300 (1954)
Dwivedi, C., Kumar, A., Ajish, J.K., Singh, K.K., Kumar, M., Wattal, P.K., Bajaj, P.N.: Resorcinol–formaldehyde coated XAD resin beads for removal of cesium ions from radioactive waste: synthesis, sorption and kinetic studies. RSC Adv. 2, 5557–5564 (2012)
Dwivedi, C., Pathak, S.K., Kumar, M., Tripathi, S.C., Bajaj, P.N.: Removal of cesium by spherical resorcinol–formaldehyde resin beads: sorption and kinetic studies. J. Radioanal. Nucl. Chem. 297(1), 1–8 (2013)
Fujita, H., Sasano, H., Miyajima, R., Sakoda, A.: Adsorption equilibrium and kinetics of cesium onto insoluble prussian blue synthesized by an immediate precipitation reaction between Fe3+ and [Fe(CN)6]4−. Adsorption 20, 905–915 (2014)
Goto, S., Goto, M., Teshima, H.: Simplified evaluations of mass transfer resistances from batch–wise adsorption and ion-exchange data. 1. Linear isotherms. Ind. Eng. Chem. Fundam. 20, 368–371 (1981a)
Goto, S., Goto, M., Teshima, H.: Simplified evaluations of mass transfer resistances from batch–wise adsorption and ion-exchange data. 2. Nonlinear isotherms. Ind. Eng. Chem. Fundam. 20, 371–375 (1981b)
Hashimoto, K., Miura, K., Nagata, S.: Intraparticle diffusivities in liquid–phase adsorption with nonlinear isotherms. J. Chem. Eng. Jpn. 8(5), 367–373 (1975)
Hui, C.-W., Chen, B., McKay, G.: Pore–surface diffusion model for batch adsorption processes. Langmuir 19, 4188–4196 (2003)
Jagasia, P., Mohapatra, P.K., Dhami, P.S., Patil, A.B., Adya, V.C., Sengupta, A., Gandhi, P.M., Wattal, P.K.: Studies on the radiolytic stability of newly developed solvent systems containing four calix-crown-6 ligands for radio–cesium recovery. J. Radioanal. Nucl. Chem. 302(3), 1087–1093 (2014)
James, A.M., Harding, S., Robshaw, T., Bramall, N., Ogden, M.D., Dawson, R.: Selective environmental remediation of strontium and cesium using sulfonated hyper-cross-linked polymers (SHCPs). ACS Appl. Mater. Interfaces 11(25), 22464–22473 (2019)
Kobayashi, T., Ohshiro, M., Nakamoto, K., Uchida, S.: Decontamination of extra–diluted radioactive cesium in Fukushima water using zeolite–polymer composite fibers. Ind. Eng. Chem. Res. 55(25), 6996–7002 (2016)
Lee, H.Y., Kim, H.S., Jeong, H.-K., Park, M., Chung, D.-Y., Lee, K.-Y., Lee, E.-H., Lim, W.T.: Selective removal of radioactive cesium from nuclear waste by zeolites: on the origin of cesium selectivity revealed by systematic crystallographic studies. J. Phys. Chem. C 121(19), 10594–10608 (2017)
Li, T., Cao, M., Liang, J., Xie, X., Du, G.: Mechanism of base–catalyzed resorcinol–formaldehyde and phenol–resorcinol–formaldehyde condensation reactions: a theoretical study. Polymers (Basel) 9, 426 (2017)
Liao, Z., Fang, X., Li, Q., Xie, J., Ni, L., Wang, D., Sun, X., Wang, L., Li, J.: Resorcinol–formaldehyde nanobowls modified thin film nanocomposite membrane with enhanced nanofiltration performance. J. Membr. Sci. 594, 117468 (2020)
Nightingale Jr., E.R.: Phenomenological theory of ion solvation. Effective radii of hydrated ions. J. Phys. Chem. 63(9), 1381–1387 (1959)
Pekala, R.W.: Organic aerogels from the polycondensation of resorcinol with formaldehyde. J. Mater. Sci. 24(9), 3221–3227 (1989)
Saxena, S., Prasad, M., D'Souza, S.F.: Radionuclide sorption onto low–cost mineral adsorbent. Ind. Eng. Chem. Res. 45(26), 9122–9128 (2006)
Tranter, T.J., Herbst, R.S., Todd, T.A.: Determination of a solid phase mass transfer coefficient for modeling an adsorption bed system using ammonium molybdophosphate-polyacrylonitrile (AMP-PAN) as a sorbent for the removal of 137Cs from acidic nuclear waste solutions. Adsorption 8, 291–299 (2002)
Valsala, T.P., Sonavane, M.S., Kore, S.G., Sonar, N.L., De, V., Raghavendra, Y., Chattopadyaya, S., Dani, U., Kulkarni, Y., Changrani, R.D.: Treatment of low level radioactive liquid waste containing appreciable concentration of TBP degraded products. J. Hazard. Mater. 196(30), 22–28 (2011)
Walton, H.F.: Ion exchange in analytical chemistry. J. Chem. Educ. 42(2), 111–115 (1965)
Weber, W.J., Morris, J.C.: Kinetics of adsorption on carbon from solutions. J. Sanit. Eng. Div. Am. Soc. Civ. Eng. 89, 31–60 (1963)
Wu, F.-C., Tseng, R.-L., Juang, R.-S.: Initial behavior of intraparticle diffusion model used in the description of adsorption kinetics. Chem. Eng. J. 153, 1–8 (2009)
Yang, H., Luo, M., Luo, L., Wang, H., Hu, D., Lin, J., Wang, X., Wang, Y., Wang, S., Bu, X., Feng, P., Wu, T.: Highly selective and rapid uptake of radionuclide cesium based on robust zeolitic chalcogenide via stepwise ion–exchange strategy. Chem. Mater. 28, 8774–8780 (2016)
Yin, X., Wang, X., Wu, H., Takahashi, H., Inaba, Y., Ohnuki, T., Takeshita, K.: Effects of NH4+, K+, Mg2+, and Ca2+ on the cesium adsorption/desorption in binding sites of vermiculitized biotite. Environ. Sci. Technol. 51, 13886–13894 (2017)
Yoshida, S., Iwamura, S., Ogino, I., Mukai, S.R.: Continuous–flow separation of cesium ion by ammonium molybdophosphate immobilized in a silica microhoneycomb (AMP-SMH). Adsorption 25, 1089–1098 (2019)
Zakaria, E.S., Ali, I.M., Aly, H.F.: Adsorption behaviour of 134Cs and 22Na ions on Tin and Titanium ferrocyanides. Adsorption 10, 237–244 (2004)
Zhou, X.-X., Li, Y.-J., Liu, J.-F.: Highly efficient removal of silver-containing nanoparticles in waters by aged iron oxide magnetic particles. ACS Sustain. Chem. Eng. 5(6), 5468–5476 (2017)
Acknowledgements
This work was supported by the Hyogo Science and Technology Association.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
The manuscript was written through contributions of all authors. All authors have given approval to the final version of the manuscript. These authors contributed equally.
Corresponding author
Ethics declarations
Conflict of interest
All the authors declared that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Taguchi, S., Nakatani, T., Saeki, H. et al. Characterization of resorcinol–formaldehyde hydrogel as adsorbent for cesium ion. Adsorption 27, 81–90 (2021). https://doi.org/10.1007/s10450-020-00271-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10450-020-00271-y