Abstract
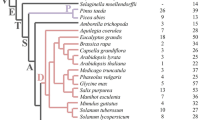
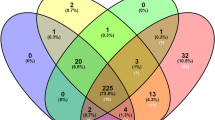
INH2 homologous genes encoding vacuolar invertase inhibitor were identified in 37 Solanaceae species, and the INH2 polymorphism was evaluated. A significant level of interspecific polymorphism of the INH2 coding sequence was found within the Petota and Lycopersicon sections, which corresponds to the high mutation rate during the evolution of the INH2 genes and presumably associated with the modern plant diversity. Structural analysis revealed the presence of conserved motifs and amino acid residues, non-transmembrane signal peptide and the PMEI domain in the INH2 proteins. The possibility of applying the INH2 sequence to assess the inter-genera and inter-section Solanaceae phylogeny was shown.


Similar content being viewed by others
REFERENCES
Bailey, T.L. and Elkan, C., Fitting a mixture model by expectation maximization to discover motifs in biopolymers, in Proc. Second Int. Conf. Intell. Systems Mol. Biol., California: AAAI Press, Menlo Park, 1994, pp. 28–36.
Bate, N.J., Niu, X., Wang, Y., Reimann, K.S., and Helentjaris, T.G., An invertase inhibitor from maize localizes to the embryo surrounding region during early kernel development, Plant Physiol., 2004, vol. 134, no. 1, pp. 246–254.
Brummell, D.A., Chen, R.K., Harris, J.C., Zhang, H., Hamiaux, C., Kralicek, A.V., and McKenzie, M.J., Induction of vacuolar invertase inhibitor mRNA in potato tubers contributes to cold-induced sweetening resistance and includes spliced hybrid mRNA variants, J. Exp. Bot., 2011, vol. 62, no. 10, pp. 3519–3534.
Choi, Y., Sims, G.E., Murphy, S., Miller, J.R., and Chan, A.P., Predicting the functional effect of amino acid substitutions and indels, PLoS One, 2012, vol. 7, no. 10. e46688.
Fukuoka, H., Yamaguchi, H., Nunome, T., Negoro, S., Miyatake, K., and Ohyama, A., Accumulation, functional annotation, and comparative analysis of expressed sequence tags in eggplant (Solanum melongena L.), the third pole of the genus Solanum species after tomato and potato, Gene, 2010, vol. 450, nos. 1–2, pp. 76–84.
Gebhardt, C., The historical role of species from the Solanaceae plant family in genetic research, Theor. Appl. Genet., 2016, vol. 129, no. 12, pp. 2281–2294.
Greiner, S., Rausch, T., Sonnewald, U., and Herbers, K., Ectopic expression of a tobacco invertase inhibitor homolog prevents cold-induced sweetening of potato tubers, Nat. Biotechnol, 1999, vol. 17, no. 7, pp. 708–711.
Hothorn, M., Wolf, S., Aloy, P., Greiner, S., and Scheffzek, K., Structural insights into the target specificity of plant invertase and pectin methylesterase inhibitory proteins, Plant Cell, 2004, vol. 16, no. 12, pp. 3437–3447.
Kelley, L.A., Mezulis, S., Yates, C.M., Wass, M.N., and Sternberg, M.J., The Phyre2 web portal for protein modeling, prediction and analysis, Nat. Protoc., 2015, vol. 10, no. 6, pp. 845–858.
Knapp, S., Bohs, L., Nee, M., and Spooner, D.M., Solanaceae—a model for linking genomics with biodiversity, Comp. Funct. Genom., 2004, vol. 5, pp. 285–291.
Kumar, S., Stecher, G., and Tamura, K., MEGA7: molecular evolutionary genetics analysis version 7.0 for bigger datasets, Mol. Biol. Evol., 2016, vol. 33, no. 7, pp. 1870–1874.
Link, M., Rausch, T., and Greiner, S., In Arabidopsis thaliana, the invertase inhibitors AtC/VIF1 and 2 exhibit distinct target enzyme specificities and expression profiles, FEBS Lett., 2004, vol. 573, nos. 1–3, pp. 105–110.
Machida-Hirano, R., Diversity of potato genetic resources, Breed. Sci., 2015, vol. 65, no. 1, pp. 26–40.
Di Matteo, A., Giovane, A., Raiola, A., Camardella, L., Bonivento, D., De Lorenzo, G., Cervone, F., Bellincampi, D., and Tsernoglou, D., Structural basis for the interaction between pectin methylesterase and a specific inhibitor protein, Plant Cell, 2005, vol. 17, no. 3, pp. 849–858.
Moscone, E.A., Scaldaferro, M.A., Grabiele, M., Cecchini, N.M., Garcia, Y.S., Jarret, R., Davina, J.R., Ducasse, D.A., Barboza, G.E., and Ehrendorfer, F., The evolution of chili peppers (Capsicum—Solanaceae): a cytogenetic perspective, Acta Hort. ISHS, 2007, vol. 745, pp. 137–170.
Peralta, I.E., Spooner, D.M., and Knapp, S., Taxonomy of wild tomatoes and their relatives (Solanum sect. Lycopersicoides, Sect. Juglandifolia, Sect. Lycopersicon; Solanaceae), Syst. Bot. Monogr., 2008, vol. 84, pp. 1–186.
Pressey, R., Invertase inhibitor from potatoes: purification, characterization, and reactivity with plant invertases, Plant Physiol., 1967, vol. 42, no. 12, pp. 1780–1786.
Qin, G., Zhu, Z., Wang, W., Cai, J., Chen, Y., Li, L., and Tian, S., A tomato vacuolar invertase inhibitor mediates sucrose metabolism and influences fruit ripening, Plant Physiol., 2016, vol. 172, no. 3, pp. 1596–1611.
Rausch, T. and Greiner, S., Plant protein inhibitors of invertases, Biochim. Biophys. Acta, 2004, vol. 16962. 253e261.
Scognamiglio, M.A., Ciardiello, M.A., Tamburrini, M., Carratore, V., Rausch, T., and Camardella, L., The plant invertase inhibitor shares structural properties and disulfide bridges arrangement with the pectin methylesterase inhibitor, J. Protein Chem., 2003, vol. 22, pp. 363–369.
Sherson, S.M., Alford, H.L., Forbes, S.M., Wallace, G., and Smith, S.M., Roles of cell-wall invertases and monosaccharide transporters in the growth and development of Arabidopsis,J. Exp. Bot., 2003, vol. 54, pp. 525–531.
Slugina, M.A., Shchennikova, A.V., and Kochieva, E.Z., TAI vacuolar invertase orthologs: the interspecific variability in tomato plants (Solanum section Lycopersicon), Mol. Genet. Genomics, 2017, vol. 292, no. 5, pp. 1123–1138.
Spooner, D.M., Fajardo, D., and Salas, A., Revision of the Solanum medians complex (Solanum section Petota), Syst. Bot., 2008, vol. 33, no. 3, pp. 579–588.
Spooner, D.M., Ghislain, M., Simon, R., Jansky, S.H., and Gavrilenko, T., Systematics, diversity, genetics, and evolution of wild and cultivated potatoes, Bot. Rev., 2014, vol. 80, pp. 283–383.
Spooner, D.M., Ruess, H., Arbizu, C.I., Rodriguez, F., and Solis-Lemus, C., Greatly reduced phylogenetic structure in the cultivated potato clade (Solanum section Petota pro parte), Am. J. Bot., 2018, vol. 105, no. 1, pp. 60–70.
Tymowska-Lalanne, Z. and Kreis, M., Expression of the Arabidopsis thaliana invertase gene family, Planta, 1998, vol. 207, no. 2, pp. 259–265.
Wan, H., Wu, L., Yang, Y., Zhou, G., and Ruan, Y.L., Evolution of sucrose metabolism: the dichotomy of invertases and beyond, Trends Plant Sci, 2018, vol. 23, no. 2, pp. 163–177.
ACKNOWLEDGMENTS
The authors are grateful to S.R. Mayorov for providing plant material, as well as E.O. Shmelkova for significant technical assistance in conducting the study.
Funding
This work was supported by the Russian Foundation for Basic Research (grant no. 18-016-00108), the Ministry of Science and Higher Education of the Russian Federation, and the Federal Targeted Program for Agricultural Development of the Russian Federation for 2017–2025 (subprogram Development of Selection and Seed Production of Potatoes in the Russian Federation).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflict of interest. This article does not contain any studies involving animals or human participants performed by any of the authors.
Rights and permissions
About this article
Cite this article
Slugina, M.A., Kochieva, E.Z. & Shchennikova, A.V. Polymorphism and Phylogeny of the Vacuolar Invertase Inhibitor Gene INH2 Homologs in Solanaceae Species. Biol Bull Russ Acad Sci 47, 474–481 (2020). https://doi.org/10.1134/S1062359020040111
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1062359020040111