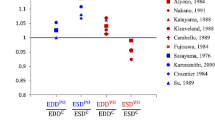
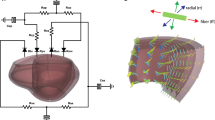
Abstract
Growth and remodeling in the heart is driven by a combination of mechanical and hormonal signals that produce different patterns of growth in response to exercise, pregnancy, and various pathologies. In particular, increases in afterload lead to concentric hypertrophy, a thickening of the walls that increases the contractile ability of the heart while reducing wall stress. In the current study, we constructed a multiscale model of cardiac hypertrophy that connects a finite-element model representing the mechanics of the growing left ventricle to a cell-level network model of hypertrophic signaling pathways that accounts for changes in both mechanics and hormones. We first tuned our model to capture published in vivo growth trends for isoproterenol infusion, which stimulates β-adrenergic signaling pathways without altering mechanics, and for transverse aortic constriction (TAC), which involves both elevated mechanics and altered hormone levels. We then predicted the attenuation of TAC-induced hypertrophy by two distinct genetic interventions (transgenic Gq-coupled receptor inhibitor overexpression and norepinephrine knock-out) and by two pharmacologic interventions (angiotensin receptor blocker losartan and β-blocker propranolol) and compared our predictions to published in vivo data for each intervention. Our multiscale model captured the experimental data trends reasonably well for all conditions simulated. We also found that when prescribing realistic changes in mechanics and hormones associated with TAC, the hormonal inputs were responsible for the majority of the growth predicted by the multiscale model and were necessary in order to capture the effect of the interventions for TAC.







Similar content being viewed by others
Code availability
FEBio Growth Plugin code will be uploaded to the FEBio Plugins page (https://febio.org/plugins/) and to SimTK (https://simtk.org/) when the manuscript is published; Network model can be run independently through Netflux (https://github.com/saucermanlab/Netflux).
References
Akhter SA, Luttrell LM, Rockman HA, Iaccarino G, Lefkowitz RJ, Koch WJ (1998) Targeting the receptor-G(q) interface to inhibit in vivo pressure overload myocardial hypertrophy. Science 280(5363):574–577. https://doi.org/10.1126/science.280.5363.574
Allwood MA, Kinobe RT, Ballantyne L, Romanova N, Melo LG, Ward CA, Brunt KR, Simpson JA (2014) Heme oxygenase-1 overexpression exacerbates heart failure with aging and pressure overload but is protective against isoproterenol-induced cardiomyopathy in mice. Cardiovasc Pathol 23(4):231–237. https://doi.org/10.1016/j.carpath.2014.03.007
Arts T, Delhaas T, Bovendeerd P, Verbeek X, Prinzen FW (2005) Adaptation to mechanical load determines shape and properties of heart and circulation: the CircAdapt model. Am J Physiol Heart Circ Physiol. https://doi.org/10.1152/ajpheart.00444.2004
Benjamin EJ, Virani SS, Callaway CW, Chang AR, Cheng S, Chiuve SE, Cushman M et al (2018) Heart disease and stroke statistics—2018 update: a report from the American Heart Association. Circulation. https://doi.org/10.1161/CIR.0000000000000558
Brooks WW, Conrad CH (2009) Isoproterenol-induced myocardial injury and diastolic dysfunction in mice: structural and functional correlates. Comp Med 59(4):339–343.
Bruno RM, Taddei S (2014) Renal denervation and regression of left ventricular hypertrophy. Eur Heart J 35(33):2205–2207. https://doi.org/10.1093/eurheartj/ehu127
Choi DJ, Koch WJ, Hunter JJ, Rockman HA (1997) Mechanism of β-adrenergic receptor desensitization in cardiac hypertrophy is increased β-adrenergic receptor kinase. J Biol Chem 272(27):17223–17229. https://doi.org/10.1074/jbc.272.27.17223
Clarke SA, Goodman NC, Ailawadi G, Holmes JW (2015) Effect of scar compaction on the therapeutic efficacy of anisotropic reinforcement following myocardial infarction in the dog. J Cardiovasc Transl Res 8(6):353–361. https://doi.org/10.1007/s12265-015-9637-1
Cuspidi C, Vaccarella A, Negri F, Sala C (2010) Resistant hypertension and left ventricular hypertrophy: an overview. J Am Soc Hypertens 4(6):319–324. https://doi.org/10.1016/J.JASH.2010.10.003
De Windt LJ, Lim HW, Bueno OF, Liang Q, Delling U, Braz JC, Glascock BJ et al (2001) Targeted inhibition of calcineurin attenuates cardiac hypertrophy in vivo. Proc Natl Acad Sci 98(6):3322–3327. https://doi.org/10.1073/pnas.031371998
Drews O, Tsukamoto O, Liem D, Streicher J, Wang Y, Ping P (2010) Differential regulation of proteasome function in isoproterenol-induced cardiac hypertrophy. Circ Res 107(9):1094–1101. https://doi.org/10.1161/CIRCRESAHA.110.222364
Estrada AC, Yoshida K, Clarke SA, Holmes JW (2020) Longitudinal reinforcement of acute myocardial infarcts improves function by transmurally redistributing stretch and stress. J Biomech Eng. https://doi.org/10.1115/1.4044030
Frank DU, Sutcliffe MD, Saucerman JJ (2018) Network-based predictions of in vivo cardiac hypertrophy. J Mol Cell Cardiol 121(August):180–189. https://doi.org/10.1016/j.yjmcc.2018.07.243
Galindo CL, Skinner MA, Mounir Errami L, Olson D, Watson DA, Li J, McCormick JF et al (2009) Transcriptional profile of isoproterenol-induced cardiomyopathy and comparison to exercise-induced cardiac hypertrophy and human cardiac failure. BMC Physiol 9(1):23. https://doi.org/10.1186/1472-6793-9-23
Gelsomino S, Frassani R, Morocutti G, Nucifora R, Da Col P, Minen G, Morelli A, Livi U (2001) Time course of left ventricular remodeling after stentless aortic valve replacement. Am Heart J 142(3):556–562. https://doi.org/10.1067/mhj.2001.117777
Göktepe S, Abilez OJ, Parker KK, Kuhl E (2010) A multiscale model for eccentric and concentric cardiac growth through sarcomerogenesis. J Theor Biol 265(3):433–442. https://doi.org/10.1016/j.jtbi.2010.04.023
Grossman W, Jones D, McLaurin LP (1975) Wall stress and patterns of hypertrophy in the human left ventricle. J Clin Investig 56(1):56–64. https://doi.org/10.1172/JCI108079
Humphrey JD, Rajagopal KR (2002) A constrained mixture model for growth and remodeling in soft tissues. Math Models Methods Appl Sci 12(3):407–430. https://doi.org/10.1142/S0218202502001714
Huss JM, Imahashi K-i, Dufour CR, Weinheimer CJ, Courtois M, Kovacs A, Giguère V, Murphy E, Kelly DP (2007) The nuclear receptor ERRα is required for the bioenergetic and functional adaptation to cardiac pressure overload. Cell Metab 6(1):25–37. https://doi.org/10.1016/j.cmet.2007.06.005
Iwanaga Y, Kihara Y, Inagaki K, Onozawa Y, Yoneda T, Kataoka K, Sasayama S (2001) Differential effects of angiotensin II versus endothelin-1 inhibitions in hypertrophic left ventricular myocardium during transition to heart failure. Circulation 104(5):606–612. https://doi.org/10.1161/hc3101.092201
Izumo S, Nadal-Ginard B, Mahdavi V (1988) Protooncogene induction and reprogramming of cardiac gene expression produced by pressure overload. Proc Natl Acad Sci USA 85(2):339–343. https://doi.org/10.1073/pnas.85.2.339
Jaehnig EJ, Heidt AB, Greene SB, Cornelissen I, Black BL (2006) Increased susceptibility to isoproterenol-induced cardiac hypertrophy and impaired weight gain in mice lacking the histidine-rich calcium-binding protein. Mol Cell Biol 26(24):9315–9326. https://doi.org/10.1128/MCB.00482-06
Kamal FA, Mickelsen DM, Wegman KM, Travers JG, Moalem J, Hammes SR, Smrcka AV, Blaxall BC (2014) Simultaneous adrenal and cardiac G-protein-coupled receptor-Gβγ inhibition halts heart failure progression. J Am Coll Cardiol 63(23):2549–2557. https://doi.org/10.1016/j.jacc.2014.02.587
Kawel-Boehm N, Kronmal R, Eng J, Folsom A, Gregory Burke J, Carr J, Shea S, Lima JAC, Bluemke DA (2019) Left ventricular mass at MRI and long-term risk of cardiovascular events: the multi-ethnic study of atherosclerosis (MESA). Radiology 293(1):107–114. https://doi.org/10.1148/radiol.2019182871
Kerckhoffs RCP, Omens JH, McCulloch AD (2012) A single strain-based growth law predicts concentric and eccentric cardiac growth during pressure and volume overload. Mech Res Commun 42:40–50. https://doi.org/10.1016/j.mechrescom.2011.11.004
Li L, Zhou N, Gong H, Jian W, Lin L, Komuro I, Ge J, Zou Y (2010) Comparison of angiotensin II type 1-receptor blockers to regress pressure overload-induced cardiac hypertrophy in mice. Hypertens Res 33(12):1289–1297. https://doi.org/10.1038/hr.2010.182
Liao Y, Takashima S, Asano Y, Asakura M, Ogai A, Shintani Y, Minamino T et al (2003) Activation of adenosine A1 receptor attenuates cardiac hypertrophy and prevents heart failure in murine left ventricular pressure-overload model. Circ Res 93(8):759–766. https://doi.org/10.1161/01.RES.0000094744.88220.62
Lin IE, Taber LA (1995) A model for stress-induced growth in the developing heart. J Biomech Eng 117(3):343–349. https://doi.org/10.1115/1.2794190
Lindpaintner K, Lund DD, Schmid PG (1987) Effects of chronic progressive myocardial hypertrophy on indexes of cardiac autonomic innervation. Circ Res 61(1):55–62. https://doi.org/10.1161/01.RES.61.1.55
Ling H, Zhang T, Pereira L, Means CK, Cheng H, Yusu G, Dalton ND et al (2009) Requirement for Ca2+/calmodulin-dependent kinase II in the transition from pressure overload-induced cardiac hypertrophy to heart failure in mice. J Clin Investig 119(5):1230–1240. https://doi.org/10.1172/JCI38022
Lorell BH, Carabello BA (2000) Left ventricular hypertrophy. Circulation 102(4):470–479. https://doi.org/10.1161/01.CIR.102.4.470
Monrad ES, Hess OM, Murakami T, Nonogi H, Corin WJ, Krayenbuehl HP (1988) Time course of regression of left ventricular hypertrophy after aortic valve replacement. Circulation 77(6):1345–1355. https://doi.org/10.1161/01.cir.77.6.1345
Nishimura RA, Otto CM, Bonow RO, Carabello BA, Erwin JP, Guyton RA, O’Gara PT et al (2014) 2014 AHA/ACC guideline for the management of patients with valvular heart disease: executive summary: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol 63(22):2438–2488. https://doi.org/10.1016/j.jacc.2014.02.537
Otto CM (2006) Valvular aortic stenosis. disease severity and timing of intervention. J Am Coll Cardiol 47(11):2141–2151. https://doi.org/10.1016/j.jacc.2006.03.002
Patrizio M, Musumeci M, Stati T, Fasanaro P, Palazzesi S, Catalano L, Marano G (2007) Propranolol causes a paradoxical enhancement of cardiomyocyte foetal gene response to hypertrophic stimuli. Br J Pharmacol 152(2):216–222. https://doi.org/10.1038/sj.bjp.0707350
Rapacciuolo A, Esposito G, Caron K, Mao L, Thomas SA, Rockman HA (2001) Important role of endogenous norepinephrine and epinephrine in the development of in vivo pressure-overload cardiac hypertrophy. J Am Coll Cardiol 38(3):876–882. https://doi.org/10.1016/S0735-1097(01)01433-4
Richards DA, Aronovitz MJ, Calamaras TD, Tam K, Martin GL, Liu P, Bowditch HK, Zhang P, Huggins GS, Blanton RM (2019) Distinct phenotypes induced by three degrees of transverse aortic constriction in mice. Sci Rep 9(1):1–15. https://doi.org/10.1038/s41598-019-42209-7
Rockman HA, Wachhorst SP, Mao L, Ross J (1994) ANG II receptor blockade prevents ventricular hypertrophy and ANF gene expression with pressure overload in mice. Am J Physiol 266(6 Pt 2):H2468–H2475. https://doi.org/10.1152/ajpheart.1994.266.6.H2468
Rodriguez EK, Hoger A, McCulloch AD (1994) Stress-dependent finite-growth in soft elastic tissues. J Biomech 27(4):455–467
Ryall KA, Holland DO, Delaney KA, Kraeutler MJ, Parker AJ, Saucerman JJ (2012) Network reconstruction and systems analysis of cardiac myocyte hypertrophy signaling. J Biol Chem 287(50):42259–42268. https://doi.org/10.1074/jbc.M112.382937
Ryu Y, Jin L, Kee HJ, Piao ZH, Cho JY, Kim GR, Choi SY, Lin MQ, Jeong MH (2016) Gallic acid prevents isoproterenol-induced cardiac hypertrophy and fibrosis through regulation of JNK2 signaling and Smad3 binding activity. Sci Rep 6(1):34790. https://doi.org/10.1038/srep34790
Schunkert H, Dzau VJ, Tang SS, Hirsch AT, Apstein CS, Lorell BH (1990) Increased rat cardiac angiotensin converting enzyme activity and mRNA expression in pressure overload left ventricular hypertrophy: effects on coronary resistance, contractility, and relaxation. J Clin Investig 86(6):1913–1920. https://doi.org/10.1172/JCI114924
Sergeeva IA, Christoffels VM (2013) Regulation of expression of atrial and brain natriuretic peptide, biomarkers for heart development and disease. Biochim Biophys Acta Mol Basis Dis. https://doi.org/10.1016/j.bbadis.2013.07.003
Sucharov CC, Hijmans JG, Sobus RD, Melhado WFA, Miyamoto SD, Stauffer BL (2013) β-Adrenergic receptor antagonism in mice: a model for pediatric heart disease. J Appl Physiol (Bethesda, MD.: 1985) 115(7):979–987. https://doi.org/10.1152/japplphysiol.00627.2013
Sugden PH (1999) Signaling in myocardial hypertrophy: life after calcineurin? Circ Res 84(6):633–646. https://doi.org/10.1161/01.RES.84.6.633
Swynghedauw B (1999) Molecular mechanisms of myocardial remodeling. Physiol Rev 79(1):215–262. https://doi.org/10.1152/physrev.1999.79.1.215
Szymanski MW, Singh DP (2019) Isoproterenol. StatPearls Publishing, Treasure Island
Taber LA (1998) Biomechanical growth laws for muscle tissue. J Theor Biol. https://doi.org/10.1006/jtbi.1997.0618
Tshori S, Gilon D, Beeri R, Nechushtan H, Kaluzhny D, Pikarsky E, Razin E (2006) Transcription factor MITF regulates cardiac growth and hypertrophy. J Clin Investig 116(10):2673–2681. https://doi.org/10.1172/JCI27643
Tuerdi N, Lu X, Zhu B, Chen C, Cao Y, Wang Y, Zhang Q, Li Z, Qi R (2016) Preventive effects of simvastatin nanoliposome on isoproterenol-induced cardiac remodeling in mice. Nanomed Nanotechnol Biol Med 12(7):1899–1907. https://doi.org/10.1016/j.nano.2016.05.002
Wang X, Ye Y, Gong H, Jian W, Yuan J, Wang S, Yin P et al (2016) The effects of different angiotensin II type 1 receptor blockers on the regulation of the ACE-AngII-AT1 and ACE2-Ang(1–7)-Mas axes in pressure overload-induced cardiac remodeling in male mice. J Mol Cell Cardiol 97(August):180–190. https://doi.org/10.1016/j.yjmcc.2016.05.012
Waters SB, Diak DM, Zuckermann M, Goldspink PH, Leoni L, Roman BB (2013) Genetic background influences adaptation to cardiac hypertrophy and Ca2+ handling gene expression. Front Physiol 4:11. https://doi.org/10.3389/fphys.2013.00011
Witzenburg CM, Holmes JW (2018) Predicting the time course of ventricular dilation and thickening using a rapid compartmental model. J Cardiovasc Transl Res 11(2):109–122. https://doi.org/10.1007/s12265-018-9793-1
Xiang YK (2011) Compartmentalization of β-adrenergic signals in cardiomyocytes. Circ Res 109(2):231–244. https://doi.org/10.1161/CIRCRESAHA.110.231340
Yamazaki T, Komuro I, Kudoh S, Zou Y, Shiojima I, Mizuno T, Takano H et al (1995) Angiotensin II partly mediates mechanical stress-induced cardiac hypertrophy. Circ Res 77(2):258–265. https://doi.org/10.1161/01.RES.77.2.258
Yamazaki T, Komuro I, Kudoh S, Zou Y, Shiojima I, Hiroi Y, Mizuno T et al (1996) Endothelin-1 is involved in mechanical stress-induced cardiomyocyte hypertrophy. J Biol Chem 271(6):3221–3228. https://doi.org/10.1074/jbc.271.6.3221
Yoshida Y, Shimizu I, Katsuumi G, Jiao S, Suda M, Hayashi Y, Minamino T (2015) P53-induced inflammation exacerbates cardiac dysfunction during pressure overload. J Mol Cell Cardiol 85:183–198
Zhang Y, Jingting X, Long Z, Wang C, Wang L, Sun P, Li P, Wang T (2016) Hydrogen (H2) inhibits isoproterenol-induced cardiac hypertrophy via antioxidative pathways. Front Pharmacol 7(October):392. https://doi.org/10.3389/fphar.2016.00392
Acknowledgements
This study was funded by the National Institutes of Health (NIH) Grant U01 HL127654 and American Heart Association (AHA) Grant 16PRE30250007.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Estrada, A.C., Yoshida, K., Saucerman, J.J. et al. A multiscale model of cardiac concentric hypertrophy incorporating both mechanical and hormonal drivers of growth. Biomech Model Mechanobiol 20, 293–307 (2021). https://doi.org/10.1007/s10237-020-01385-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10237-020-01385-6