Abstract
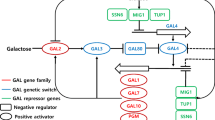
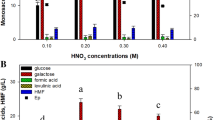
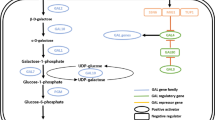
A total 42.68 g/L monosaccharide with 0.10 g/L HMF was obtained from 10% (w/v) Kappaphycus alvarezii with thermal acid hydrolysis using 350 mM HNO3 at 121 °C for 60 min and enzymatic saccharification with a 1:1 mixture of Viscozyme L and Celluclast 1.5 L for 72 h. To enhance the galactose utilization rate, fermentation was performed with overexpression of GAL1 (galactokinase), GAL7 (galactose-1-phosphate uridyltransferase), GAL10 (UDP-glucose-4-epimerase), and PGM2 (phosphoglucomutase 2) in Saccharomyces cerevisiae CEN.PK2 using CCW12 as a strong promoter. Among the strains, the overexpression of PGM2 showed twofold high galactose utilization rate (URgal) and produced ethanol 1.4-fold more than that of the control. Transcriptional analysis revealed the increase of PGM2 transcription level leading to enhance glucose-6-phosphate and fructose-6-phosphate and plays a key role in ensuring a higher glycolytic flux in the PGM2 strain. This finding shows particular importance in biofuel production from seaweed because galactose is one of the major monosaccharides in seaweeds such as K. alvarezii.





Similar content being viewed by others
References
Vassilev, S. V., & Vassileva, C. G. (2016). Composition, properties and challenges of algae biomass for biofuel application: an overview. Fuel, 181, 1–33.
Sudhakar, K., Mamat, R., Samykano, M., Azmi, W. H., Ishak, W. F. W., & Yusaf, T. (2018). An overview of marine macroalgae as bioresource. Renewable and Sustainable Energy Reviews, 91, 165–179.
Ra, C. H., Nguyen, T. H., Jeong, G. T., & Kim, S. K. (2016). Evaluation of hyper thermal acid hydrolysis of Kappaphycus alvarezii for enhanced bioethanol production. Bioresource Technology, 209, 66–72.
Jinek, M., Chylinski, K., Fonfara, I., Hauer, M., Doudna, J. A., & Charpentier, E. (2012). A programmable dual-RNA-guided DNA endonuclease in adaptive bacterial immunity. Science (New York, N.Y.), 337(6096), 816–821.
DiCarlo, J. E., Norville, J. E., Mali, P., Rios, X., Aach, J., & Church, G. M. (2013). Genome engineering in Saccharomyces cerevisiae using CRISPR-Cas systems. Nucleic Acids Research, 41(7), 4336–4343.
Zhang, G.-C., Kong, I. I., Kim, H., Liu, J.-J., Cate, J. H. D., & Jin, Y.-S. (2014). Construction of a quadruple auxotrophic mutant of an industrial polyploid Saccharomyces cerevisiae strain by using RNA-guided Cas9 nuclease. Applied and Environmental Microbiology, 80(24), 7694–7701.
Tsai, C.-S., Kong, I. I., Lesmana, A., Million, G., Zhang, G.-C., Kim, S. R., & Jin, Y.-S. (2015). Rapid and marker-free refactoring of xylose-fermenting yeast strains with Cas9/CRISPR. Biotechnology and Bioengineering, 112(11), 2406–2411.
Ostergaard, S., Roca, C., Rønnow, B., Nielsen, J., & Olsson, L. (2000). Physiological studies in aerobic batch cultivations of Saccharomyces cerevisiae strains harboring the MEL1 gene. Biotechnology and Bioengineering, 68(3), 252–259.
Lee, K.-S., Hong, M.-E., Jung, S.-C., Ha, S.-J., Yu, B. J., Koo, H. M., Park, S. M., Kweon, D. H., Park, J. C., & Jin, Y.-S. (2011). Improved galactose fermentation of Saccharomyces cerevisiae through inverse metabolic engineering. Biotechnology and Bioengineering, 108(3), 621–631.
Frey, P. A. (1996). The Leloir pathway: a mechanistic imperative for three enzymes to change the stereochemical configuration of a single carbon in galactose. FASEB Journal, 10(4), 461–470.
Howard, S. M., & Heinrich, M. R. (1965). The anomeric specificity of yeast galactokinase. Archives of Biochemistry and Biophysics, 110(2), 395–400.
Segawa, T., & Fukasawa, T. (1979). The enzymes of the galactose cluster in Saccharomyces cerevisiae. Purification and characterization of galactose-1-phosphate uridylyltransferase. The Journal of Biological Cheimistry, 254(21), 10707–10709.
Nogi, Y., & Fukasawa, T. (1980). A novel mutation that affects utilization of galactose in Saccharomyces cerevisiae. Current Genetics, 2(2), 115–120.
Bro, C., Knudsen, S., Regenberg, B., Olsson, L., & Nielsen, J. (2005). Improvement of galactose uptake in Saccharomyces cerevisiae through overexpression of phosphoglucomutase: example of transcript analysis as a tool in inverse metabolic engineering. Applied and Environmental Microbiology, 71(11), 6465–6472.
Marinho-Soriano, E., Fonseca, P. C., Carneiro, M. A. A., & Moreira, W. S. C. (2006). Seasonal variation in the chemical composition of two tropical seaweeds. Bioresource Technology, 97(18), 2402–2406.
Ra, C. H., Kim, Y. J., Lee, S. Y., Jeong, G. T., & Kim, S. K. (2015). Effects of galactose adaptation in yeast for ethanol fermentation from red seaweed, Gracilaria verrucosa. Bioprocess and Biosystems Engineering, 38(9), 1715–1722.
Bae, Y.-H., Kweon, D.-H., Park, Y.-C., & Seo, J.-H. (2014). Deletion of the HXK2 gene in Saccharomyces cerevisiae enables mixed sugar fermentation of glucose and galactose in oxygen-limited conditions. Process Biochemistry, 49(4), 547–553.
Livak, K. J., & Schmittgen, T. D. (2001). Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods, 25(4), 402–408.
Hong, I. K., Jeon, H., & Lee, S. B. (2014). Comparison of red, brown and green seaweeds on enzymatic saccharification process. Journal of Industrial and Engineering Chemistry, 20(5), 2687–2691.
Kim, I., Lee, B., Park, J. Y., Choi, S. A., & Han, J. I. (2014). Effect of nitric acid on pretreatment and fermentation for enhancing ethanol production of rice straw. Carbohydrate Polymers, 99, 563–567.
Tutt, M., Kikas, T., & Olt, J. (2012). Influence of different pretreatment methods on bioethanol production from wheat straw. Agonomy Research, Biosystems Engineering Special Issue, 1, 209–276.
Antonetti, C., Licursi, D., Fulignati, S., Valentini, G., & Raspolli Galletti, A. (2016). New frontiers in the catalytic synthesis of levulinic acid: from sugars to raw and waste biomass as starting feedstock. Catalysts, 6(12), 196.
Rodríguez, A., De La Cera, T., Herrero, P., & Moreno, F. (2001). The hexokinase 2 protein regulates the expression of the GLK1, HXK1 and HXK2 genes of Saccharomyces cerevisiae. The Biochemical Journal, 355(3), 625–631.
Li, C., Wang, Q., & Zhao, Z. K. (2008). Acid in ionic liquid: an efficient system for hydrolysis of lignocellulose. Green Chemistry, 10(2), 177–182.
Rodríguez-Chong, A., Ramírez, J. A., Garrote, G., & Vázquez, M. (2004). Hydrolysis of sugar cane bagasse using nitric acid: a kinetic assessment. Journal of Food Engineering, 61(2), 143–152.
Ahn, D. J., Kim, S. K., & Yun, H. S. (2012). Optimization of pretreatment and saccharification for the production of bioethanol from water hyacinth by Saccharomyces cerevisiae. Bioprocess and Biosystems Engineering, 35(1–2), 35–41.
Garcia Sanchez, R., Hahn-Hägerdal, B., & Gorwa-Grauslund, M. F. (2010). PGM2 overexpression improves anaerobic galactose fermentation in Saccharomyces cerevisiae. Microbial Cell Factories, 9, 1–8.
De Jongh, W. A., Bro, C., Ostergaard, S., Regenberg, B., Olsson, L., & Nielsen, J. (2008). The roles of galactitol, galactose-1-phosphate, and phosphoglucomutase in galactose-induced toxicity in Saccharomyces cerevisiae. Biotechnology and Bioengineering, 101(2), 317–326.
Gonçalves, P. M., Griffioen, G., Paul Bebelman, J., & Planta, R. J. (1997). Signalling pathways leading to transcriptional regulation of genes involved in the activation of glycolysis in yeast. Molecular Microbiology, 25(03), 483–493.
Timson, D. J. (2007). Galactose metabolism in Saccharomyces cerevisiae. Dynamic Biochemistry, Process Biotechnology and Molecular Biology, 1(1), 63–73.
Ostergaard, S., Olsson, L., & Nielsen, J. (2001). In vivo dynamics of galactose metabolism in Saccharomyces cerevisiae: metabolic fluxes and metabolite levels. Biotechnology and Bioengineering, 73(5), 412–425.
Lowry, O. H., & Passonneau, J. V. (1969). Phosphoglucomutase kinetics with the phosphates of fructose, glucose, mannose, ribose, and galactose. Journal of Biological Chemistry, 244(4), 910–916.
Funding
This research was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (2019R1F1A1041288).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sunwoo, I.Y., Sukwong, P., Park, Y.R. et al. Enhancement of Galactose Uptake from Kappaphycus alvarezii Hydrolysate Using Saccharomyces cerevisiae Through Overexpression of Leloir Pathway Genes. Appl Biochem Biotechnol 193, 335–348 (2021). https://doi.org/10.1007/s12010-020-03422-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-020-03422-7