Abstract
Microglia are resident macrophage-like cells in the central nervous system (CNS). The induction of microglial activation dampens neuroinflammation-related diseases by promoting microglial (re)polarization to the anti-inflammatory (M2) phenotype and can serve as a potential therapeutic approach. Mitochondrial respiration and metabolic reprogramming are required for the anti-inflammatory response of M2 macrophages. However, whether these mitochondrial-dependent pathways are involved in microglial (re)polarization to the anti-inflammatory (M2) phenotype under conditions of lipopolysaccharide (LPS)-induced neuroinflammation remains unclear. Moreover, the mechanisms that coordinate mitochondrial respiration and the functional reprogramming of microglial cells have not been fully elucidated. Rosmarinic acid (RA) possesses antioxidative and anti-inflammatory activities, and we previously reported that RA markedly suppresses LPS-stimulated M1 microglial activation in mice. In this study, we found that RA suppresses M1 microglial polarization and promotes microglial polarization to the M2 phenotype under conditions of neuroinflammation. We identified an increase in mitochondrial respiration and found that metabolic reprogramming is required for the RA-mediated promotion of microglial polarization to the M2 phenotype under LPS-induced neuroinflammation conditions. Hypoxia-inducible factor (HIF) subunits are the key effector molecules responsible for the effects of RA on the restoration of mitochondrial function, metabolic reprogramming, and phenotypic polarization to M2 microglia. The phosphoinositide-dependent protein kinase 1 (PDPK1)/Akt/mTOR pathway is involved in the RA-mediated regulation of HIF expression and increase in M2 marker expression. We propose that the inhibition of PDPK1/Akt/HIFs by RA might be a potential therapeutic approach for inhibiting neuroinflammation through the regulation of microglial M1/M2 polarization.

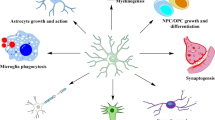
Schematic of the mechanism through which RA suppresses LPS-induced neuroinflammation by promoting microglial polarization to the M2 phenotype via PDPK1/Akt/HIFs. The bold arrows indicate the direction of the effects of RA (i.e., inhibitory or promoting effects on cytokines or mediators).










Similar content being viewed by others
Abbreviations
- CNS:
-
Central nervous system
- LPS:
-
Lipopolysaccharide
- HIF:
-
Hypoxia-inducible factor
- MS:
-
Multiple sclerosis
- AD:
-
Alzheimer’s disease
- PD:
-
Parkinson’s disease
- TNF-α:
-
Tumor necrosis factor-α
- IL:
-
Interleukin
- iNOS:
-
Inducible nitric oxide synthase
- IFN-γ:
-
Interferon-gamma
- Arg1:
-
Arginase-1
- Ym-1:
-
Chitinase 3–like 3
- Fizz-1:
-
Inflammatory zone 1
- TGF-β:
-
Transforming growth factor β
- MRC1:
-
Mannose receptor C type 1
- CD206:
-
Mannose receptor
- CD163:
-
Scavenger receptor
- RA:
-
Rosmarinic acid
- PPP:
-
Pentose phosphate pathway
- ATP:
-
Adenosine triphosphate
- ROS:
-
Reactive oxygen species
- NO:
-
Nitric oxide
- TCA:
-
Tricarboxylic acid
- ACLY:
-
ATP-citrate lyase
- PDPK1:
-
Phosphoinositide-dependent protein kinase 1
- PDK3:
-
Pyruvate dehydrogenase kinase 3
- qRT-PCR:
-
Quantitative real-time PCR
- OM:
-
Oligomycin
- FAO:
-
Glucose and fatty acid oxidation
- 2-DG:
-
2-Deoxy-glucose
- ETO:
-
Etomoxir
- PDK1:
-
Pyruvate dehydrogenase kinase 1
- LDHA:
-
Lactate dehydrogenase A
- PDH:
-
Pyruvate dehydrogenase
- DMOG:
-
Dimethyloxaloylglycine
- TLR4:
-
Toll-like receptor 4
- CD14:
-
Cluster of differentiation 14
- JNK:
-
c-Jun N-terminal kinase
- NF-κB:
-
Nuclear factor-kappa B
- NLRP3:
-
NOD-like receptor family pyrin domain-containing 3
- TPI:
-
Triosephosphate isomerase
- HK2:
-
Hexokinase 2
- PKM:
-
Enolase 1 and pyruvate kinase muscle
- TSC:
-
Tuberous sclerosis complex
- GLUT1:
-
Glycose transporter 1
- OXPHOX:
-
Oxidative phosphorylation
References
Chen, W.W., X. Zhang, and W.J. Huang. 2016. Role of neuroinflammation in neurodegenerative diseases (review). Molecular Medicine Reports 13: 3391–3396.
Benakis, C., L. Garcia-Bonilla, C. Iadecola, and J. Anrather. 2014. The role of microglia and myeloid immune cells in acute cerebral ischemia. Frontiers in Cellular Neuroscience 8: 461.
Orihuela, R., C.A. McPherson, and G.J. Harry. 2016. Microglial M1/M2 polarization and metabolic states. British Journal of Pharmacology 173: 649–665.
Du, L., Y. Zhang, Y. Chen, J. Zhu, Y. Yang, and H.L. Zhang. 2017. Role of microglia in neurological disorders and their potentials as a therapeutic target. Molecular Neurobiology 54: 7567–7584.
Moehle, M.S., and A.B. West. 2015. M1 and M2 immune activation in Parkinson's disease: Foe and ally? Neuroscience 302: 59–73.
Mosser, D.M., and J.P. Edwards. 2008. Exploring the full spectrum of macrophage activation. Nature Reviews Immunology 8: 958–969.
Ransohoff, R.M. 2016. A polarizing question: Do M1 and M2 microglia exist? Nature Neuroscience 19: 987–991.
Glass, C.K., K. Saijo, B. Winner, M.C. Marchetto, and F.H. Gage. 2010. Mechanisms underlying inflammation in neurodegeneration. Cell 140: 918–934.
Tao, Y., L. Li, B. Jiang, Z. Feng, L. Yang, J. Tang, Q. Chen, J. Zhang, Q. Tan, H. Feng, Z. Chen, and G. Zhu. 2016. Cannabinoid receptor-2 stimulation suppresses neuroinflammation by regulating microglial M1/M2 polarization through the cAMP/PKA pathway in an experimental GMH rat model. Brain Behavior and Immunity 58: 118–129.
Yang, X., S. Xu, Y. Qian, and Q. Xiao. 2017. Resveratrol regulates microglia M1/M2 polarization via PGC-1alpha in conditions of neuroinflammatory injury. Brain Behavior and Immunity 64: 162–172.
Porro, C., A. Cianciulli, R. Calvello, and M.A. Panaro. 2015. Reviewing the role of resveratrol as a natural modulator of microglial activities. Current Pharmaceutical Design 21: 5277–5291.
Calvello, R., A. Cianciulli, G. Nicolardi, F. De Nuccio, L. Giannotti, R. Salvatore, C. Porro, T. Trotta, M.A. Panaro, and D.D. Lofrumento. 2017. Vitamin D treatment attenuates neuroinflammation and dopaminergic neurodegeneration in an animal model of Parkinson's disease, shifting M1 to M2 microglia responses. Journal of Neuroimmune Pharmacology 12: 327–339.
Liu, J., X. Li, J. Lin, Y. Li, T. Wang, Q. Jiang, and D. Chen. 2016. Sarcandra glabra (Caoshanhu) protects mesenchymal stem cells from oxidative stress: A bioevaluation and mechanistic chemistry. BMC Complementary and Alternative Medicine 16: 423.
Zhou, H., J. Liang, D. Lv, Y. Hu, Y. Zhu, J. Si, and S. Wu. 2013. Characterization of phenolics of Sarcandra glabra by non-targeted high-performance liquid chromatography fingerprinting and following targeted electrospray ionisation tandem mass spectrometry/time-of-flight mass spectrometry analyses. Food Chemistry 138: 2390–2398.
Ghaffari, H., M. Venkataramana, G.B. Jalali, N.S. Chandra, A. Nataraju, N.P. Geetha, and H.S. Prakash. 2014. Rosmarinic acid mediated neuroprotective effects against H2O2-induced neuronal cell damage in N2A cells. Life Sciences 113: 7–13.
Yang, E.J., S.K. Ku, W. Lee, S. Lee, T. Lee, K.S. Song, and J.S. Bae. 2013. Barrier protective effects of rosmarinic acid on HMGB1-induced inflammatory responses in vitro and in vivo. Journal of Cellular Physiology 228: 975–982.
Rocha, J., M. Eduardo-Figueira, A. Barateiro, A. Fernandes, D. Brites, R. Bronze, C.M. Duarte, A.T. Serra, R. Pinto, M. Freitas, E. Fernandes, B. Silva-Lima, H. Mota-Filipe, and B. Sepodes. 2015. Anti-inflammatory effect of rosmarinic acid and an extract of Rosmarinus officinalis in rat models of local and systemic inflammation. Basic & Clinical Pharmacology & Toxicology 116: 398–413.
Luan, H., Z. Kan, Y. Xu, C. Lv, and W. Jiang. 2013. Rosmarinic acid protects against experimental diabetes with cerebral ischemia: Relation to inflammation response. Journal of Neuroinflammation 10: 28.
Wei, Y., J. Chen, Y. Hu, W. Lu, X. Zhang, R. Wang, and K. Chu. 2018. Rosmarinic acid mitigates lipopolysaccharide-induced neuroinflammatory responses through the inhibition of TLR4 and CD14 expression and NF-κB and NLRP3 inflammasome activation. Inflammation 41: 732–740.
Van den Bossche, J., L.A. O'Neill, and D. Menon. 2017. Macrophage immunometabolism: Where are we (going)? Trends in Immunology 38: 395–406.
Van den Bossche, J., J. Baardman, N.A. Otto, S. van der Velden, A.E. Neele, S.M. van den Berg, R. Luque-Martin, H.J. Chen, M.C. Boshuizen, M. Ahmed, M.A. Hoeksema, A.F. de Vos, and M.P. de Winther. 2016. Mitochondrial dysfunction prevents repolarization of inflammatory macrophages. Cell Reports 17: 684–696.
Huang, S.C., A.M. Smith, B. Everts, M. Colonna, E.L. Pearce, J.D. Schilling, and E.J. Pearce. 2016. Metabolic reprogramming mediated by the mTORC2-IRF4 signaling axis is essential for macrophage alternative activation. Immunity 45: 817–830.
Everts, B., E. Amiel, S.C. Huang, A.M. Smith, C.H. Chang, W.Y. Lam, V. Redmann, T.C. Freitas, J. Blagih, G.J. van der Windt, M.N. Artyomov, R.G. Jones, E.L. Pearce, and E.J. Pearce. 2014. TLR-driven early glycolytic reprogramming via the kinases TBK1-IKKvarepsilon supports the anabolic demands of dendritic cell activation. Nature Immunology 15: 323–332.
Byles, V., A.J. Covarrubias, I. Ben-Sahra, D.W. Lamming, D.M. Sabatini, B.D. Manning, and T. Horng. 2013. The TSC-mTOR pathway regulates macrophage polarization. Nature Communications 4: 2834.
Cheng, S.C., J. Quintin, R.A. Cramer, K.M. Shepardson, S. Saeed, V. Kumar, E.J. Giamarellos-Bourboulis, J.H. Martens, N.A. Rao, A. Aghajanirefah, G.R. Manjeri, Y. Li, D.C. Ifrim, R.J. Arts, B.M. van der Veer, P.M. Deen, C. Logie, L.A. O'Neill, P. Willems, F.L. van de Veerdonk, J.W. van der Meer, A. Ng, L.A. Joosten, C. Wijmenga, H.G. Stunnenberg, R.J. Xavier, and M.G. Netea. 2014. MTOR- and HIF-1alpha-mediated aerobic glycolysis as metabolic basis for trained immunity. Science 345: 1250684.
Covarrubias, A.J., H.I. Aksoylar, J. Yu, N.W. Snyder, A.J. Worth, S.S. Iyer, J. Wang, I. Ben-Sahra, V. Byles, T. Polynne-Stapornkul, E.C. Espinosa, D. Lamming, B.D. Manning, Y. Zhang, I.A. Blair, and T. Horng. 2016. Akt-mTORC1 signaling regulates Acly to integrate metabolic input to control of macrophage activation. eLife 5.
Tan, Z., N. Xie, H. Cui, D.R. Moellering, E. Abraham, V.J. Thannickal, and G. Liu. 2015. Pyruvate dehydrogenase kinase 1 participates in macrophage polarization via regulating glucose metabolism. Journal of Immunology 194: 6082–6089.
Feldhoff, L.M., C.M. Rueda, M.E. Moreno-Fernandez, J. Sauer, C.M. Jackson, C.A. Chougnet, and J. Rupp. 2017. IL-1beta induced HIF-1alpha inhibits the differentiation of human FOXP3(+) T cells. Scientific Reports 7: 465.
Sun, L., M. Zhao, X.J. Yu, H. Wang, X. He, J.K. Liu, and W.J. Zang. 2013. Cardioprotection by acetylcholine: A novel mechanism via mitochondrial biogenesis and function involving the PGC-1alpha pathway. Journal of Cellular Physiology 228: 1238–1248.
Wu, F., Q. Zou, X. Ding, D. Shi, X. Zhu, W. Hu, L. Liu, and H. Zhou. 2016. Complement component C3a plays a critical role in endothelial activation and leukocyte recruitment into the brain. Journal of Neuroinflammation 13: 23.
Wei, Y., H. Hong, X. Zhang, W. Lai, Y. Wang, K. Chu, J. Brown, G. Hong, and L. Chen. 2017. Salidroside inhibits inflammation through PI3K/Akt/HIF signaling after focal cerebral ischemia in rats. Inflammation 40: 1297–1309.
Huang, S.C., B. Everts, Y. Ivanova, D. O'Sullivan, M. Nascimento, A.M. Smith, W. Beatty, L. Love-Gregory, W.Y. Lam, C.M. O'Neill, C. Yan, H. Du, N.A. Abumrad, J.J. Urban, M.N. Artyomov, E.L. Pearce, and E.J. Pearce. 2014. Cell-intrinsic lysosomal lipolysis is essential for alternative activation of macrophages. Nature Immunology 15: 846–855.
Kim, J.W., I. Tchernyshyov, G.L. Semenza, and C.V. Dang. 2006. HIF-1-mediated expression of pyruvate dehydrogenase kinase: A metabolic switch required for cellular adaptation to hypoxia. Cell Metabolism 3: 177–185.
Jantsch, J., D. Chakravortty, N. Turza, A.T. Prechtel, B. Buchholz, R.G. Gerlach, M. Volke, J. Glasner, C. Warnecke, M.S. Wiesener, K.U. Eckardt, A. Steinkasserer, M. Hensel, and C. Willam. 2008. Hypoxia and hypoxia-inducible factor-1 alpha modulate lipopolysaccharide-induced dendritic cell activation and function. Journal of Immunology 180: 4697–4705.
Nakamura, H., Y. Makino, K. Okamoto, L. Poellinger, K. Ohnuma, C. Morimoto, and H. Tanaka. 2005. TCR engagement increases hypoxia-inducible factor-1 alpha protein synthesis via rapamycin-sensitive pathway under hypoxic conditions in human peripheral T cells. Journal of Immunology 174: 7592–7599.
Lv, R., L. Du, X. Liu, F. Zhou, Z. Zhang, and L. Zhang. 2019. Rosmarinic acid attenuates inflammatory responses through inhibiting HMGB1/TLR4/NF-κB signaling pathway in a mouse model of Parkinson's disease. Life Sciences 223: 158–165.
Coelho, V.R., C.M. Viau, R.B. Staub, M.S. De Souza, P. Pfluger, G.G. Regner, P. Pereira, and J. Saffi. 2017. Rosmarinic acid attenuates the activation of murine microglial n9 cells through the downregulation of inflammatory cytokines and cleaved caspase-3. Neuroimmunomodulation 24: 171–181.
Song, G.J., and K. Suk. 2017. Pharmacological modulation of functional phenotypes of microglia in neurodegenerative diseases. Frontiers in Aging Neuroscience 9: 139.
Wang, L., S. Pavlou, X. Du, M. Bhuckory, H. Xu, and M. Chen. 2019. Glucose transporter 1 critically controls microglial activation through facilitating glycolysis. Molecular Neurodegeneration 14: 2.
Jung, Y.J., J.S. Isaacs, S. Lee, J. Trepel, and L. Neckers. 2003. IL-1beta-mediated up-regulation of HIF-1alpha via an NF κB/COX-2 pathway identifies HIF-1 as a critical link between inflammation and oncogenesis. FASEB Journal 17: 2115–2117.
Dang, E.V., J. Barbi, H.Y. Yang, D. Jinasena, H. Yu, Y. Zheng, Z. Bordman, J. Fu, Y. Kim, H.R. Yen, W. Luo, K. Zeller, L. Shimoda, S.L. Topalian, G.L. Semenza, C.V. Dang, D.M. Pardoll, and F. Pan. 2011. Control of T(H)17/T(reg) balance by hypoxia-inducible factor 1. CELL 146: 772–784.
Bollinger, T., S. Gies, J. Naujoks, L. Feldhoff, A. Bollinger, W. Solbach, and J. Rupp. 2014. HIF-1alpha- and hypoxia-dependent immune responses in human CD4+CD25 high T cells and T helper 17 cells. Journal of Leukocyte Biology 96: 305–312.
Palazon, A., A.W. Goldrath, V. Nizet, and R.S. Johnson. 2014. HIF transcription factors, inflammation, and immunity. Immunity 41: 518–528.
Eltzschig, H.K., D.L. Bratton, and S.P. Colgan. 2014. Targeting hypoxia signalling for the treatment of ischaemic and inflammatory diseases. Nature Reviews Drug Discovery 13: 852–869.
Shi, L.Z., R. Wang, G. Huang, P. Vogel, G. Neale, D.R. Green, and H. Chi. 2011. HIF1alpha-dependent glycolytic pathway orchestrates a metabolic checkpoint for the differentiation of TH17 and Treg cells. Journal of Experimental Medicine 208: 1367–1376.
Wang, Y., Y. Bi, X. Chen, C. Li, Y. Li, Z. Zhang, J. Wang, Y. Lu, Q. Yu, H. Su, H. Yang, and G. Liu. 2016. Histone deacetylase SIRT1 negatively regulates the differentiation of Interleukin-9-producing CD4(+) T cells. Immunity 44: 1337–1349.
Imtiyaz, H.Z., E.P. Williams, M.M. Hickey, S.A. Patel, A.C. Durham, L.J. Yuan, R. Hammond, P.A. Gimotty, B. Keith, and M.C. Simon. 2010. Hypoxia-inducible factor 2 alpha regulates macrophage function in mouse models of acute and tumor inflammation. Journal of Clinical Investigation 120: 2699–2714.
Takeda, N., E.L. O'Dea, A. Doedens, J.W. Kim, A. Weidemann, C. Stockmann, M. Asagiri, M.C. Simon, A. Hoffmann, and R.S. Johnson. 2010. Differential activation and antagonistic function of HIF-{alpha} isoforms in macrophages are essential for NO homeostasis. Genes & Development 24: 491–501.
Arranz, A., C. Doxaki, E. Vergadi, D.L.T.Y. Martinez, K. Vaporidi, E.D. Lagoudaki, E. Ieronymaki, A. Androulidaki, M. Venihaki, A.N. Margioris, E.N. Stathopoulos, P.N. Tsichlis, and C. Tsatsanis. 2012. Akt1 and Akt2 protein kinases differentially contribute to macrophage polarization. Proceedings of the National Academy of Sciences of the United States of America 109: 9517–9522.
Vergadi, E., K. Vaporidi, E.E. Theodorakis, C. Doxaki, E. Lagoudaki, E. Ieronymaki, V.I. Alexaki, M. Helms, E. Kondili, B. Soennichsen, E.N. Stathopoulos, A.N. Margioris, D. Georgopoulos, and C. Tsatsanis. 2014. Akt2 deficiency protects from acute lung injury via alternative macrophage activation and miR-146a induction in mice. Journal of Immunology 192: 394–406.
Lopez-Pelaez, M., I. Soria-Castro, L. Bosca, M. Fernandez, and S. Alemany. 2011. Cot/tpl2 activity is required for TLR-induced activation of the Akt p70 S6k pathway in macrophages: Implications for NO synthase 2 expression. European Journal of Immunology 41: 1733–1741.
Luyendyk, J.P., G.A. Schabbauer, M. Tencati, T. Holscher, R. Pawlinski, and N. Mackman. 2008. Genetic analysis of the role of the PI3K-Akt pathway in lipopolysaccharide-induced cytokine and tissue factor gene expression in monocytes/macrophages. Journal of Immunology 180: 4218–4226.
Polumuri, S.K., V.Y. Toshchakov, and S.N. Vogel. 2007. Role of phosphatidylinositol-3 kinase in transcriptional regulation of TLR-induced IL-12 and IL-10 by Fc gamma receptor ligation in murine macrophages. Journal of Immunology 179: 236–246.
Pengal, R.A., L.P. Ganesan, G. Wei, H. Fang, M.C. Ostrowski, and S. Tridandapani. 2006. Lipopolysaccharide-induced production of interleukin-10 is promoted by the serine/threonine kinase Akt. Molecular Immunology 43: 1557–1564.
Fang, C., J. Yu, Y. Luo, S. Chen, W. Wang, C. Zhao, Z. Sun, W. Wu, W. Guo, Z. Han, X. Hu, F. Liao, and X. Feng. 2015. Tsc1 is a critical regulator of macrophage survival and function. Cellular Physiology and Biochemistry 36: 1406–1418.
Brown, J., H. Wang, J. Suttles, D.T. Graves, and M. Martin. 2011. Mammalian target of rapamycin complex 2 (mTORC2) negatively regulates toll-like receptor 4-mediated inflammatory response via FoxO1. Journal of Biological Chemistry 286: 44295–44305.
Festuccia, W.T., P. Pouliot, I. Bakan, D.M. Sabatini, and M. Laplante. 2014. Myeloid-specific Rictor deletion induces M1 macrophage polarization and potentiates in vivo pro-inflammatory response to lipopolysaccharide. PLoS One 9: e95432.
Mercalli, A., I. Calavita, E. Dugnani, A. Citro, E. Cantarelli, R. Nano, R. Melzi, P. Maffi, A. Secchi, V. Sordi, and L. Piemonti. 2013. Rapamycin unbalances the polarization of human macrophages to M1. Immunology 140: 179–190.
Lobo-Silva, D., G.M. Carriche, A.G. Castro, S. Roque, and M. Saraiva. 2016. Balancing the immune response in the brain: IL-10 and its regulation. Journal of Neuroinflammation 13: 297.
Zhou, S., X. Guo, S. Chen, Z. Xu, W. Duan, and B. Zeng. 2019. Apelin-13 regulates LPS-induced N9 microglia polarization involving STAT3 signaling pathway. NEUROPEPTIDES 76: 101938.
Subedi, L., J.H. Lee, S. Yumnam, E. Ji, S.Y. Kim. 2019. Anti-inflammatory effect of sulforaphane on LPS-activated microglia potentially through JNK/AP-1/NF-κB inhibition and Nrf2/HO-1 activation. Cells 8.
Gunzl, P., and G. Schabbauer. 2008. Recent advances in the genetic analysis of PTEN and PI3K innate immune properties. Immunobiology 213: 759–765.
Acknowledgments
The authors would like to acknowledge the staff of the Animal Centre of the Fujian University of Traditional Chinese Medicine for their excellent technical support.
Availability of Supporting Data
Not applicable
Funding
This work was financially supported by grants from the Fujian Provincial Health and Family Planning Commission (Grant No. 2018-ZQN-66) and the Department of Technology and Science of the Fujian Provincial Government (Grant No. 2018 J01869).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Ethical Approval and Consent to Participate
The study does not involve human participants, human data, or human tissue. All the animal experiments were performed in accordance with the guidelines of the National Institutes of Health Guide concerning the care and use of laboratory animals. The procedures for this study were approved by the Animal Care and Use Committee of Fujian University of Traditional Chinese Medicine.
Consent for Publication
Not applicable.
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 508 kb)
Rights and permissions
About this article
Cite this article
Wei, Y., Chen, J., Cai, GE. et al. Rosmarinic Acid Regulates Microglial M1/M2 Polarization via the PDPK1/Akt/HIF Pathway Under Conditions of Neuroinflammation. Inflammation 44, 129–147 (2021). https://doi.org/10.1007/s10753-020-01314-w
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-020-01314-w