Abstract
Objective
To explore the RecET-Cre/loxP system for chromosomal replacement of promoter and its application on enhancement l-leucine production in Corynebacterium glutamicum (C. glutamicum) ATCC14067.
Results
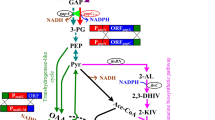
The RecET-Cre/loxP system was used to achieve the chromosomal replacement of promoter in C. glutamicum ATCC14067 to adjust the metabolic flux involving the l-leucine synthetic pathway. First, leuAr_13032 from C. glutamicum ATCC13032 which carried two mutations was overexpressed to release enzyme feedback inhibition. Then, comparing different mutations in ilvBNC gene clusters, the results indicated that ilvBNC_CP was most effective to enhance the metabolic flux of pyruvate towards l-leucine synthesis. The promoters of pck, odx and pyk2 were overexpressed under the strong promoter Peftu or Psod to improve the supply of pyruvate. Besides, the promoter PilvBNC was employed to dynamically control the transcription level of icd due to its attenuation mechanism by responding to the concentration of l-leucine. The final engineered strain produced 14.05 g l-leucine/L in flask cultivation.
Conclusion
The RecET-Cre/loxP system is effective for gene manipulation in C. glutamicum ATCC14067. Besides, the results demonstrate the potential of C. glutamicum ATCC14067 for l-leucine production and provide new targets and strategies for strain development.





Similar content being viewed by others
References
Buchholz J, Schwentner A, Brunnenkan B, Gabris C, Grimm S, Gerstmeir R, Blombach B (2013) Platform engineering of Corynebacterium glutamicum with reduced pyruvate dehydrogenase complex activity for improved production of L-lysine, L-valine, and 2-ketoisovalerate. Appl Environ Microbiol 79(18):5566–5575. doi:https://doi.org/10.1128/AEM.01741-13
Chen C, Li YY, Hu JY, Dong XY, Wang XY (2015) Metabolic engineering of Corynebacterium glutamicum ATCC13869 for L-valine production. Metab Eng 29:66–75. doi:https://doi.org/10.1016/j.ymben.2015.03.004
Eikmanns BJ, Blombach B (2014) The pyruvate dehydrogenase complex of Corynebacterium glutamicum: an attractive target for metabolic engineering. J Biotechnol 192 Pt B:339–345. https://doi.org/10.1016/j.jbiotec.2013.12.019
Elisakova V, Patek M, Holatko J, Nesvera J, Leyval D, Goergen JL, Delaunay S (2005) Feedback-resistant acetohydroxy acid synthase increases valine production in Corynebacterium glutamicum. Appl Environ Microbiol 71(1):207–213. doi:https://doi.org/10.1128/AEM.71.1.207-213.2005
Feng LY, Xu JZ, Zhang WG (2018) Improved L-leucine production in corynebacterium glutamicum by optimizing the aminotransferases. Molecules. https://doi.org/10.3390/molecules23092102
Gui YL, Ma YC, Xu QY, Zhang CL, Xie XX, Chen N (2016) Complete genome sequence of Corynebacterium glutamicum CP, a Chinese L-leucine producing strain. J Biotechnol 220:64–65. https://doi.org/10.1016/j.jbiotec.2016.01.010
Hasegawa S, Suda M, Uematsu K, Natsuma Y, Hiraga K, Jojima T, Inui M, Yukawa H (2013) Engineering of Corynebacterium glutamicum for high-yield L-valine production under oxygen deprivation conditions. Appl Environ Microbiol 79(4):1250–1257. doi:https://doi.org/10.1128/AEM.02806-12
Huang Q, Liang L, Wu W, Wu S, Huang J (2017a) Metabolic engineering of Corynebacterium glutamicum to enhance L-leucine production. Afr J Biotech 16(18):1048–1060. doi:10.5897/ajb2017a.15911
Huang YY, Li L, Xie S, Zhao NN, Han SY, Lin Y, Zheng SP (2017b) Recombineering using RecET in Corynebacterium glutamicum ATCC14067 via a self-excisable cassette. Sci Rep 7(1):7916. doi:10.1038/s41598-017-08352-9
Jäger W, Schäfer A, Pühler A, Labes G, Wohlleben W (1992) Expression of the Bacillus subtilis sacB Gene Leads to Sucrose Sensitivity in the Gram-Positive Bacterium Corynebactenium glutamicum but Not in Streptomyces lividans. J Bacteriol 174:5462–5465. doi:https://doi.org/10.1128/jb.174.16.5462-5465.1992
Li YJ, Wei HB, Wang T, Xu QY, Zhang CL, Fan XG, Ma Q, Chen N, Xie XX (2017) Current status on metabolic engineering for the production of L-aspartate family amino acids and derivatives. Bioresour Technol 245(Pt B):1588–1602. doi:https://doi.org/10.1016/j.biortech.2017.05.145
Liu YD, Wang XY, Zhan J, Hu JY (2019) The 138(th) residue of acetohydroxyacid synthase in Corynebacterium glutamicum is important for the substrate binding specificity. Enzyme Microb Technol 129:109357. doi:https://doi.org/10.1016/j.enzmictec.2019.06.001
Morbach S, Junger C, Sahm H, Eggeling L (2000) Attenuation control of ilvBNC in Corynebacterium glutamicum: evidence of leader peptide formation without the presence of a ribosome binding site. J Biosci Bioeng 90(5):501–507. doi:https://doi.org/10.1016/s1389-1723(01)80030-x
Oldiges M, Eikmanns BJ, Blombach B (2014) Application of metabolic engineering for the biotechnological production of L-valine. Appl Microbiol Biotechnol 98(13):5859–5870. doi:https://doi.org/10.1007/s00253-014-5782-8
Petersen S, Mack C, de Graaf AA, Riedel C, Eikmanns BJ, Sahm H (2001) Metabolic consequences of altered phosphoenolpyruvate carboxykinase activity in Corynebacterium glutamicum reveal anaplerotic regulation mechanisms in vivo. Metab Eng 3(4):344–361. doi:https://doi.org/10.1006/mben.2001.0198
Tittmann K, Vyazmensky M, Hubner G, Barak Z, Chipman DM (2005) The carboligation reaction of acetohydroxyacid synthase II: steady-state intermediate distributions in wild type and mutants by NMR. Proc Natl Acad Sci USA 102(3):553–558. https://doi.org/10.1073/pnas.0408210101
Vogt M, Haas S, Klaffl S, Polen T, Eggeling L, van Ooyen J, Bott M (2014) Pushing product formation to its limit: metabolic engineering of Corynebacterium glutamicum for L-leucine overproduction. Metab Eng 22:40–52. doi:https://doi.org/10.1016/j.ymben.2013.12.001
Wang YY, Xu JZ, Zhang WG (2019) Metabolic engineering of l-leucine production in Escherichia coli and Corynebacterium glutamicum: a review. Crit Rev Biotechnol 39(5):633–647. doi:https://doi.org/10.1080/07388551.2019.1577214
Yamamoto K, Tsuchisaka A, Yukawa H (2017) Branched-chain amino acids. Adv Biochem Eng Biotechnol 159:103–128. https://doi.org/10.1007/10_2016_28
Zhang CL, Li YJ, Ma J, Liu Y, He JL, Li YZ, Chen N (2018) High production of 4-hydroxyisoleucine in Corynebacterium glutamicum by multistep metabolic engineering. Metab Eng 49:287–298. https://doi.org/10.1016/j.ymben.2018.09.008
Acknowledgements
All the authors are thankful for the financial support of the National Natural Science Foundation of China (31671840) and National Key R&D Program of China (2018YFA0901700).
Supporting information
Supplementary Table 1—Bacterial strains used in this study.
Supplementary Table 2—Plasmids used in this study.
Supplementary Table 3—Primers used in this study.
Supplementary Table 4—Concentration of l-glutamate in strains.
Supplementary Figure 1—Chromosomal replacement of promoter in C. glutamicum ATCC14067 via the RecET-Cre/loxP system.
Supplementary Figure 2—Flask fermentation of WT, WT-pEC-T18-Peftu-leuA, WT-pEC-T18-Peftu-leuA_13032 and WT-pEC-T18-Peftu-leuAr_13032, and production of l-leucine and the major by-products l-valine and l-isoleucine.
Supplementary Figure 3. Flask fermentation of WT, WT-pEC-XK99E-Peftu-ilvBNC, WT-pEC-XK99E-Peftu-ilvBNrC, WT-pEC-XK99E-Peftu-ilvBNrC (E251K) and WT-pEC-XK99E-Peftu-ilvBNC_CP, and production of l-leucine and the major by-products l-valine and l-isoleucine.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Luo, G., Zhao, N., Jiang, S. et al. Application of RecET-Cre/loxP system in Corynebacterium glutamicum ATCC14067 for l-leucine production. Biotechnol Lett 43, 297–306 (2021). https://doi.org/10.1007/s10529-020-03000-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-020-03000-1