Abstract
Objectives
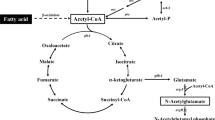
To establish a technique for efficient fatty acid production through enhancement of coenzyme A (CoA) biosynthesis and malonyl-CoA supply by introducing exogenous pantothenate kinase (coaA) and acetyl-CoA carboxylase (acc) in Escherichia coli.
Results
The expression of acc, obtained from Corynebacterium glutamicum, accumulated 2.2-fold more fatty acids in E. coli. The addition of coaA from Pseudomonas putaida or fatty acid synthase (fasA) from C. glutamicum resulted in a 3.1- and 3.6-fold increase in fatty acid synthesis in E. coli cells, which expressed acc and coaA, or acc and fasA, respectively. The transformants, simultaneously possessing all three genes, produced 5.6-fold more fatty acids. The strain possessing acc, coaA, and fasA stored 691 mg/L of fatty acids, primarily as phospholipids, inside the inner membrane after 72-h cultivation. In addition, 19% of the total CoA pool was occupied by malonyl-CoA.
Conclusions
Increased malonyl-CoA significantly contributed to fatty acid production, and the effect was boosted by the expanded total CoA pool. Manipulation of the intracellular CoA species is effective for fatty acid production in E. coli.



Similar content being viewed by others
References
Ariga N, Maruyama K, Kawaguchi A (1984) Comparative studies of fatty acid synthases of corynebacteria. J Gen Appl Microbiol 30:87–95. https://doi.org/10.2323/jgam.30.87
Bligh EG, Dyer WJ (1959) A rapid method of total lipid extraction and purification. Can J Biochem Physiol 37:911–917. https://doi.org/10.1139/y59-099
Brand LA, Strauss E (2005) Characterization of a new pantothenate kinase isoform from Hricobacter pylori. J Biol Chem 280:20185–20188. https://doi.org/10.1074/jbc.C500044200
Chalut C, Botella L, de Sousa-D’Auria C, Houssin C, Gilhot C (2006) The nonredundant roles of two 4′-phosphopantetheinyl transferases in vital processes of Mycobacteria. Proc Natl Acad Sci 103:8511–8516. https://doi.org/10.1073/pnas.0511129103
Cheng Z, Jiang J, Wu H, Li Z, Ye Q (2016) Enhanced production of 3-hydroxypropionic acid from glucose via malonyl-CoA pathway by engineered Escherichia coli. Bioresour Technol 200:897–904. https://doi.org/10.1016/j.biortech.2015.10.107
Chohnan S, Furukawa H, Fujio T, Nishihara H, Takamura Y (1997) Changes in the size and composition of intracellular pools of nonesterified coenzyme A and coenzyme A thioesters in aerobic and facultatively anaerobic bacteria. Appl Environ Microbiol 63:553–560
Chohnan S, Izawa H, Nishihara H, Takamura Y (1998) Changes in size of intracellular pools of coenzyme A and its thioesters in Escherichia coli K-12 cells to various carbon sources and stresses. Biosci Biotechnol Biochem 62:1122–1128. https://doi.org/10.1271/bbb.62.1122
Chohnan S, Matsuno S, Shimizu K, Tokutake Y, Kohari D, Toyoda A (2020) Coenzyme A and its thioester pools in obese Zucker and Zucker diabetic fatty rats. Nutrients 12:417. https://doi.org/10.3390/nu12020417
Choudhry AE, Mandichak TL, Broskey JP, Egolf RW, Kinsland C, Begley TP, Seefeld MA, Ku TW, Brown JR, Zalacain M, Ratnam K (2003) Inhibitors of pantothenate kinase: novel antibiotics for Staphylococcal infections. Antimicrob Agents Chemother 47:2051–2055. https://doi.org/10.1128/AAC.47.6.2051-2055.2003
Cronan JE Jr, Rock CO (1996) Biosynthesis of membrane lipids. In: Neidhardt FC, Curtiss R III, Gross CA, Ingraham JL, Lin ECC, Low KB, Magasanik B, Reznikoff WS, Riley M, Schaechter M, Umbarger HE (eds) Escherichia coli and Salmonella typhimurium: cellular and molecular biology, 2nd edn. American Society for Microbiology, Washington, DC, pp 612–636
Dall’Aglio P, Arthur CJ, Williams C, Vasilakis K, Maple HJ, Crosby J, Crump MP, Hadfield AT (2011) Analysis of Streptomyces coelicolor phosphopantetheinyl transferase, AcpS, reveals the basis for relaxed substrate specificity. Biochemistry 50:5704–5717. https://doi.org/10.1021/bi2003668
Davis MS, Solbiati J, Cronan JE Jr (2000) Overproduction of acetyl-CoA carboxylase activity increases the rate of fatty acid biosynthesis in Escherichia coli. J Biol Chem 275:28593–28698. https://doi.org/10.1074/jbc.M004756200
Gande R, Dover LG, Krumbach K, Besra GS, Sahm H, Oikawa T, Eggeling L (2007) The two carboxylases of Corynebacterium glutamicum essential for fatty acid and mycolic acid synthesis. J Bacteriol 189:5257–5264. https://doi.org/10.1128/JB.00254-07
Haushalter RW, Groff D, Deutsch S, The L, Chavkin TA, Brunner SF, Katz L, Keasling JD (2015) Development of an orthogonal fatty acid biosynthesis system in E. coli for oleochemical production. Metab Eng 30:1–6. https://doi.org/10.1016/j.ymben.2015.04.003
Hong BS, Yun MK, Zhang Y-M, Chohnan S, Rock CO, White SW, Jackowski S, Park H-W, Leonardi R (2006) Prokaryotic type II and type III pantothenate kinases: the same monomer fold creates dimers with distinct catalytic properties. Structure 14:1251–1261. https://doi.org/10.1016/j.str.2006.06.008
Ikeda M, Nagashima T, Nakamura E, Kato R, Ohshita M, Hayashi M, Takeno S (2017) In vivo roles of fatty acid biosynthesis enzymes in biosynthesis of biotin and α-lipoic acid in Corynebacterium glutamicum. Appl Environ Microbiol 83:e01322–e01317. https://doi.org/10.1128/AEM.01322-17
Jeon E, Lee S, Won J-I, Han SO, Kim J, Lee J (2011) Development of Escherichia coli MG1655 strains to produce long chain fatty acids by engineering fatty acid synthesis (FAS) metabolism. Enzyme Microb Technol 49:44–51. https://doi.org/10.1016/j.enzmictec.2011.04.001
Leonard E, Lim K-H, Saw P-N, Koffas MA (2007) Engineering central metabolic pathways for high-level flavonoid production in Escherichia coli. Appl Environ Microbiol 73:3877–3886. https://doi.org/10.1128/AEM.00200-07
Leonardi R, Chohnan S, Zhang Y-M, Virga KG, Lee RE, Rock CO, Jackowski S (2005a) A pantothenate kinase from Staphylococcus aureus refractory to feedback regulation by coenzyme A. J Biol Chem 280:3314–3322. https://doi.org/10.1074/jbc.M411608200
Leonardi R, Zhang Y-M, Rock CO, Jackowski S (2005b) Coenzyme A: back in action. Prog Lipid Res 44:125–153. https://doi.org/10.1016/j.plipres.2005.04.001
Li S-J, Cronan JE Jr (1992a) The gene encoding the biotin carboxylase subunit of Escherichia coli acetyl-CoA carboxylase. J Biol Chem 267:855–8636. https://www.jbc.org/content/267/2/855.abstract
Li S-J, Cronan JE Jr (1992b) The genes encoding the two carboxyltransferase subunits of Escherichia coli acetyl-CoA carboxylase. J Biol Chem 267:16841–16847. https://www.jbc.org/content/267/24/16841.abstract
Liu L, Chen J (2011) Cofactor engineering enhances the physiological function of an industrial strain. In: Carpi A (ed) Progress in molecular and environmental bioengineering-from analysis and modeling to technology applications. IntechOpen, London. https://doi.org/10.5772/18249
Lu X, Vora H, Khosla C (2008) Overproduction of free fatty acids in E. coli: implications for biodiesel production. Metab Eng 10:333–339. https://doi.org/10.1016/j.ymben.2008.08.006
Meng X, Yang J, Cao Y, Li L, Jiang X, Xu X, Liu W, Xian M, Zhang Y (2011) Increasing fatty acid production in E. coli by simulating the lipid accumulation of oleaginous microorganisms. J Ind Microbial Biotechnol 38:919–925. https://doi.org/10.1007/s10295-010-0861-z
Miyahisa I, Kaneko M, Funa N, Kawasaki H, Kojima H, Ohnishi Y, Horinouchi S (2005) Efficient production of (2S)-flavanones by Escherichia coli containing an artificial biosynthetic gene cluster. Appl Microbiol Biotechnol 68:498–504. https://doi.org/10.1007/s00253-005-1916-3
Ogata Y, Chohnan S (2015) Prokaryotic typeIII pantothenate kinase enhances coenzyme A biosynthesis in Escherichi coli. J Gen Appl Microbiol 61:266–269. https://doi.org/10.2323/jgam.61.266
Radmacher E, Alderwick LJ, Besra GS, Brown AK, Gibson KJC, Sahm H, Eggeling L (2005) Two functional FAS-I type fatty acid synthases in Corynebacterium glutamicum. Microbiology 151:2421–2427. https://doi.org/10.1099/mic.0.28012-0
Rathnasingh C, Raj SM, Lee Y, Catherine C, Ashok S, ParK S (2012) Production of 3-hydroxypropionic acid via malonyl-CoA pathway using recombinant Escherichia coli strains. J Biotechnol 157:633–640. https://doi.org/10.1016/j.jbiotec.2011.06.008
Stuible HP, Wagner C, Andreou I, Huter G, Haselmann J, Schweizer E (1996) Identification and functional differentiation of two type I fatty acid synthases in Brevibacterium ammoniagenes. J Bacteriol 178:4787–4793. https://doi.org/10.1128/jb.178.16.4787-4793.1996
Takamura Y, Nomura G (1988) Changes in the intracellular concentration of acetyl-CoA and malonyl-CoA in relation to the carbon and energy metabolism of Escherichia coli K12. J Gen Microbiol 134:2249–2253. https://doi.org/10.1099/00221287-134-8-2249
Takeno S, Takasaki M, Urabayashi A, Miura A, Muramatsu T, Mitsuhashi S, Ikeda M (2013) Development of fatty acid-producing Corynebacterium glutamicum strains. Appl Environ Microbiol 79:6776–6783. https://doi.org/10.1128/AEM.02003-13
Vadali RV, Bennett GN, San KY (2004) Cofactor engineering of intracellular CoA/acetyl-CoA and its effect on metabolic flux redistribution in Escherichia coli. Metab Eng 6:133–139. https://doi.org/10.1016/j.ymben.2004.02.001
Vallari DS, Jackowski S, Rock CO (1987) Regulation of pantothenate kinase by coenzyme A and its thioesters. J Biol Chem 262:2468–2471. https://www.jbc.org/content/262/6/2468.abstract
Wang Y, San K-Y, Bennett GN (2013) Cofactor engineering for advancing chemical biotechnology. Curr Opin Biotechnol 24:994–999. https://doi.org/10.1016/j.copbio.2013.03.022
Zha W, Rubin-Pitel SB, Shao Z, Zhao H (2009) Improving cellular malonyl-CoA level in Escherichia coli via metabolic engineering. Metab Eng 11:192–198. https://doi.org/10.1016/j.ymben.2009.01.005
Zhu B, Cai G, Hall EO, Freeman GJ (2007) In-fusion assembly: seamless engineering of multidomain fusion proteins, modular vectors, and mutations. Biotechniques 43:354–359. https://doi.org/10.2144/000112536
Acknowledgements
This work was supported by a Grant-in-Aid for Scientific Research (C) 16K07655 from The Japan Society for the Promotion of Science. We would like to thank Editage (www. editage.com) for English language editing.
Supporting information
Supplementary Table 1. Oligonucleotide primers used in this work.
Supplementary Table 2. Glucose consumption, pH of media, and pantothenate kinase (CoaA) activities of the E. coli transformants in time-course experiments (See Fig. 3).
Supplementary Fig. 1. Plasmids used in this work. fasA, Corynebacterium glutamicum fatty acid synthase; acpS, C. glutamicum acyl carrier protein synthase; dtsR1, β-subunit of C. glutamicum acetyl-CoA carboxylase; accBC, α-subunit; accE, ε-subunit; coaA, Pseudomonas putida pantothenate kinase.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Satoh, S., Ozaki, M., Matsumoto, S. et al. Enhancement of fatty acid biosynthesis by exogenous acetyl-CoA carboxylase and pantothenate kinase in Escherichia coli. Biotechnol Lett 42, 2595–2605 (2020). https://doi.org/10.1007/s10529-020-02996-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-020-02996-w