Abstract
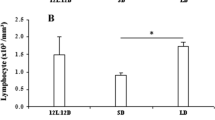
The role of circulatory glucocorticoids along with melatonin in the regulation of lung-associated cell-mediated immunity has never been explored in any tropical bird. This could be interesting because glucocorticoids are immunosuppressive while melatonin is immunostimulatory in nature. In our present study, we report the localization and expression of the glucocorticoid and melatonin receptors and the effect of melatonin on dexamethasone-induced suppression of lung-associated and general immunity in the jungle bush quail Perdicula asiatica. The birds were treated with melatonin and dexamethasone at dose of 25µg/100g B.wt./day and 30µg/100g B.wt./day, respectively, for twenty eight days. At the end of the experiment, the birds were sacrificed and lung tissues and blood samples were collected for histology and morphometric analysis of lymphoid tissue (BALT and non-BALT nodules), total leukocyte count (TLC), lymphocyte count (LC), percent stimulation ratio of isolated lung lymphocytes (% SR), immunohistochemistry, western blot analysis and hormonal assay. Dexamethasone injection reduced the immune status in terms of the size of BALT and non-BALT nodules, TLC, LC and % SR, upregulated expression of the glucocorticoid receptor (GR) and downregulated expression of melatonin receptor types Mel1a and Mel1b. Melatonin administration increased the above immune parameters and upregulated expression of melatonin receptor types Mel1a and Mel1b while downregulating GR. Our results suggest the existence of the trade-off relationship between melatonin and corticosterone, which might be responsible for the regulation of lung-associated cell-mediated immunity under stress conditions via their receptors in the lung of the jungle bush quail Perdicula asiatica.



Similar content being viewed by others
REFERENCES
Kharwar, R.K. and Haldar, C., Anatomical and histological profile of bronchus-associated lymphoid tissue and localization of melatonin receptor types (Mel1a and Mel1b) in the lung-associated immune system of a tropical bird, Perdicula asiatica, Acta Histochem., 2011, vol. 113, pp. 333–339.
Reese, S., Dalamani, G., and Kaspers, B., The avian lung-associated immune system: a review, Vet. Res., 2006, vol. 37, pp. 311–324.
Kharwar, R.K. and Haldar, C., Annual variation in lung associated immunity and season dependent invasion of Alternaria alternata in lungs of Indian jungle bush quail (Perdicula asiatica), Animal Biol., 2012, vol. 62, pp. 301–314.
Bugajski, J., Social stress adapts signaling pathways involved in stimulation of the hypothalamic–pituitary–adrenal axis, J. Physiol. Pharmacol., 1999, vol. 50, pp. 367–379.
Blanchard, R.J., Hebert, M., Sakai, R.R., et al., Chronic social stress: changes in behavioral and physical indices of emotion, Aggress. Behav., 1998, vol. 24, pp. 307–321.
Bohus, B., Koolhaas, J.M., Heijnen, C.J., and de Boer, O., Immunological responses to social stress: dependence on social environment and coping abilities, Neuropsychobiol., 1993, vol. 28, pp. 95–99.
Selye, H., Forty years of stress research: Principal remaining problems and misconceptions, Canad. Med. Assoc. J., 1976, vol. 115, pp. 53–56.
Siegel, H.S., Physiological stress in birds, Bioscience, 1980, vol. 30, pp. 529–533.
Munck, A., Guyre, P.M., and Holbrook, N.J., Physiological functions of glucocorticoids in stress and their relation to pharmacological actions, Endocr. Rev., 1984 vol. 5, pp. 25–44.
Chrousos, G.P. and Gold, P.W., The concepts of stress and stress system disorders. Overview of physical and behavioral homeostasis, JAMA, 1992, vol. 267, pp. 1244–1252.
Hardy, M.P., Gao, H. B., Dong, Q., et al., Stress hormone and male reproductive function, Cell Tiss. Res, 2005, vol. 322, pp. 147–153.
Nakano, K., Suzuki, S., and Oh, C., Significance of increased secretion of glucocorticoids in mice and rats injected with bacterial endotoxin. Brain Behav. Immunol., 1987, vol. 1, pp. 159–172.
Sapolsky, R.M., Romero, M., and Munck, A.U., How do glucocorticoids influence stress responses? Integrating permissive, suppressive, stimulatory, and preparative actions, Endocrine Rev., 2000, vol. 21, pp. 55–89.
Heilman, D.H., Failure of hydrocortisone to inhibit blastogenesis by pokeweed mitogen in human leukocyte cultures, Clin. Exp. Immunol., 1972, vol. 11, pp. 393–403.
Cupps, T.R. and Fauci, A.S., Corticosteroid-mediated immunoregulation in man, Immunol. Rev., 1982, vol. 65, pp. 133–155.
Rupprecht, R., Wodarz, N., Kornhuber, J., et al., In vivo and in vitro effects of glucocorticoids on lymphocyte proliferation in man: relationship to glucocorticoid receptors, Neuropsychobiol., 1991, vol. 24, pp. 61–66.
Gupta, S., and Haldar, C., Physiological crosstalk between melatonin and glucocorticoid receptor modulates T-cell mediated immune responses in a wild tropical rodent, Funambulus pennanti, J. Steroid Biochem. Mol. Biol., 2013, vol. 134, pp. 23–36.
Barnes, P.J. and Adcock, I.M., How do corticosteroids work in asthma? Ann. Intern. Med., 2003, vol. 139, pp. 359–370.
Caldefie-Chezet, F., Poulin, A., Enreille-Leger, A., and Vasson, M.P., Troglitazone reduces leptinemia during experimental dexamethasone-induced stress, Hormone Metab. Res., 2005, vol. 37, pp. 164–171.
Park, S., Jang, J.S., Jun, D.W., and Hong, S.M., Exercise enhances insulin and leptin signaling in the cerebral cortex and hypothalamus during dexamethasone-induced stress in diabetic rats, Neuroendocrinol, 2005, vol. 82, pp. 282–293.
Eid, Y., Ebeid, T., and Younis, H., Vitamin E supplementation reduces dexamethasone-induced oxidative stress in chicken semen, Br. Poultry Sci., 2006, vol. 47, pp. 350–356.
de Vries, A., Holmes, M.C., Heijnis, A., et al., Prenatal dexamethasone exposure induces changes in nonhuman primate offspring cardiometabolic and hypothalamicpituitary–adrenal axis function, J. Clin. Invest., 2007, vol. 117, pp. 1058–1067.
Kemp, M.W., Newnham, J.P., Challis, J.G., Jobe, A.H., and Stock, S.J., The clinical use of corticosteroids in pregnancy, Human Reprod. Update, 2016, vol. 22, pp. 240–259.
Hanafy, A.M., and Khalil, H.A., Influence of chronic dexamethasone administration on reproductive parameters and semen traits in male of Japanese Quail, Asian J. Poultry Sci., 2015, vol. 9(4), pp. 223–232.
Westerhof, I., Van den Brom, W.E., and Mol, J.A., Responsiveness of the glucocorticoid-suppressed pituitary–adrenocortical system of pigeons (Columba livia domestica) to stimulation with arginine vasopressin, Avian Dis., 1996, vol. 40, pp. 312–320.
Aengwanich, W. and Chinrasri, O., Effect of dexamethasone on differential white blood cell counts and heterophil/lymphocyte ratio in Japanese quails (Coturnix coturnix Japonica), Songklanakarin J. Sci. Technol., 2003, vol. 25, pp. 183–189.
Gao, J., Lin, H., Wang, X.J., Song, Z.G., and Jiao, H.C., Vitamin E supplementation alleviates the oxidative stress induced by dexamethasone treatment and improves meat quality in broiler chickens, Poultry Sci., 2010, vol. 89, pp. 318–327.
El-Habbak, M.M., Abou-El-Soud, S.B., and Ebeid, T.A., Effect of induced stress by dexamethasone administration on performance, egg quality and some blood parameters of laying hens, Egypt Poultry Sci., 2005, vol. 25, pp. 89–105.
Eid, Y., Ebeid, T., Moawad, M., and El-Habbak, M., Reduction of dexamethasone-induced oxidative stress and lipid peroxidation in laying hens by dietary vitamin E supplementation, Emir. J. Food Agric., 2008, vol. 20, pp. 28–40.
Singh, S.S., Yadav, S.K., and Haldar, C., Effect of glucocorticoid and melatonin on immune function of an Indian tropical bird, Perdicula asiatica: an in vivo and in vitro study, Eur. J. Inflam., 2010, vol. 8, pp. 89–97.
Duffy, D.L., Bentley, G.E., Drazen, D.L., and Ball, G.F., Effects of testosterone on cell-mediated and humoral immunity in non-breeding adult European starlings, Behav. Ecol., 2000, vol. 11, pp. 654–662.
Peters, A., Testosterone treatment is immunosuppressive in superb fairy-wrens, yet free-living males with high testosterone are more immunocompetent, Proc. Biol. Sci., 2000, vol. 267, pp. 883–889.
Casto, J.M., Nolan, V. Jr., and Ketterson, E.D., Steroid hormones and immune function: experimental studies in wild and captive dark-eyed juncos (Junco hyemalis), Am. Nat., 2001, vol. 157, pp. 408–420.
Buchanan, K.L., Evans, M.R., and Goldsmith, A.R., Testosterone, dominance signalling and immunosuppression in the house sparrow, Passer domesticus, Behav. Ecol. Sociobiol., 2003, vol. 55, pp. 50–59.
Owen-Ashley, N.T., Hasselquist, D., and Wingfield, J.C., Androgens and the immunocompetence handicap hypothesis: unraveling direct and indirect pathways of immunosuppression in song sparrows, Am. Nat., 2004, vol. 164, pp. 490–505.
Deviche, P., and Cortez, L., Androgen control of immunocompetence in the male house finch, Carpodacus mexicanus Muller, J. Exp. Biol., 2005, vol. 208, pp. 1287–1295.
Boughton, R.K., Bridge, E.S., and Schoech, S.J., Energetic trade-offs between immunity and reproduction in male Japanese quail (Coturnix coturnix), J. Exp. Zool., 2007, vol. 307, pp. 479–487.
Fargallo, J.A., Martinez-Padilla, J., Toledano-Diaz, A., et al., Sex and testosterone effect on growth, immunity and melanin colouration of nestling Eurasian kestrels, J. Animal Ecol., 2007, vol. 76, pp. 201–209.
Evans, M.R., Goldsmith, A.R., and Norris, S.R.A., The effects of testosterone on antibody production and plumage colouration in male house sparrows (Passer domesticus), Behav. Ecol. Sociobiol., 2000, vol. 47, pp. 156–163.
Poiani, A., Goldsmith, A.R., and Evan, M.R., Ectoparasites of house sparrows (Passerdomesticus): an experimental test of the immunocompetence handicap hypothesis and a new model, Behav. Ecol. Sociobiol., 2000, vol. 47, pp. 230–242.
Harvey, S., Phillips, J.G., Rees, A., et al., Stress and adrenal function, J. Exp. Zool., 1984, vol. 232, pp. 633–645.
Raberg, L., Stjernman, M., Hasselquist, D., et al., On the adaptive significance of stress-induced immunosupression, Proc. Roy. Soc. Lond. B, 1998, vol. 265, pp. 912–919.
Wingfield, J.C., Breuner, C., and Jacobs, J., Corticosterone and behavioural responses to unpredictable events, Perspectives in Avian Endocrinology, Harvey, S. and Etches, R.J., Eds., Bristol, 1997.
Baykara, B., Tekmen, I., Pekcetin, C., Ulukus, C., Tuncel, P., Sagol, O., Ormen, M., and Ozogul, C., The protective effects of carnosine and melatonin in ischemia-reperfusion injury in the rat liver, Acta Histochem., 2009, vol. 111, pp. 42–51.
Hardeland, R., Cardinali, D.P., Srinivasan, V., Spence, D.W., Brown, G.M., and Pandi-Perumal, S.R., Melatonin-a pleiotropic, orchestrating regulator molecule, Progr. Neurobiol., 2011, vol. 93, pp. 350–384.
Maldonado, M.D., Mora-Santos, M., Naji, L., Carrascosa-Salmoral, M.P., Naranjo, M.C., and Calvo, J.R., Evidence of melatonin synthesis and release by mast cells. Possible modulatory role on inflammation, Pharmacol. Res., 2010, vol. 62, pp. 282–287.
Nava, M., Quiroz, Y., Vaziri, N., and Rodriguez-Iturbe, B., Melatonin reduces renal interstitial inflammation and improves hypertension in spontaneously hypertensive rats, Am. J. Physiol. Renal Physiol., 2003, vol. 284, pp. 447–454.
Radogna, F., Diederich, M., and Ghibelli, L., Melatonin: a pleiotropic molecule regulating inflammation, Biochem. Pharmacol., 2010, vol. 80, pp. 1844–1852.
Maestroni, G.J.M., and Pierpaoli, W., Pharmacological control of the hormonally mediated immune response, Psychoneuroimmunology, Ader, R., ed., Academic Press, New York, 1981, pp. 405–425.
Ahmad, R., and Haldar, C., Photoperiodic regulation of melatonin receptor MT1 and MT2 expression dynamics in spleen and thymus of a tropical rodent Funambulus pennanti during reproductively active and inactive phase, Chronobiol. Int., 2010, vol. 27, pp. 446–462.
Maestroni, G.J.M., Conti, A., and Pierpaoli, W., The pineal gland and the circadian opiatergic immunoregulatory role of melatonin, Ann. NY Acad. Sci., 1987, vol. 496, pp. 67–77.
Maestroni, G.J.M., Conti, A., and Pierpaoli, W., Role of the pineal gland in immunity. III. Melatonin antagonizes the immunosuppressive effect of acute stress via an opiatergic mechanism, Immunology, 1988, vol. 63, pp. 465–469.
Maestroni, G.J.M., Conti, A., and Pierpaoli, W., Melatonin, stress and the immune system, Pineal Research Reviews, vol. 7, Reiter, R.J., Ed., New York, 1989, pp. 203–233.
Ahmad, R., Haldar, C., and Gupta, S., Melatonin membrane receptor type MT1 modulates cell-mediated immunity in the seasonally breeding tropical rodent Funambulus pennanti, Neuroimmunomodul., 2012, vol. 19, pp. 50–59.
Ebisawa, T., Karne, S., Lerner, M.R., and Reppert, S.M., Expression cloning of a high-affinity melatonin receptor from Xenopus dermal melanophores, Proc. Natl. Acad. Sci. USA, 1994, vol. 91(13), pp. 6133–6137.
Reppert, S.M., Weaver, D.R., Cassone, V.M., Godson, C., and Kolakowski, L.F. Jr., Melatonin receptors are for the birds: molecular analysis of two receptor subtypes differentially expressed in chick brain, Neuron, 1995, vol. 15(5), pp. 1003–1015.
Kharwar, R.K. and Haldar, C., Reproductive phase dependent variation in Lung associated immune system (LAIS) and expression of melatonin receptors (Mel1a & Mel1b) in lung of a tropical bird, Perdicula asiatica, Canad. J. Zool., 2011, vol. 89, pp. 10–18.
Kharwar, R.K. and Haldar, C., Photoperiod regulates lung associated immunological parameters and melatonin receptor (Mel1a and Mel1b) in lungs of a tropical bird, Perdicula asiatica, Photochem.Photobiol., 2011, vol. 87, pp. 427–432.
Kharwar, R.K., Haldar, C., and Singh, S., Me11b receptor mediated action of melatonin in regulation of lung associated immune system (LAIS) of Perdicula asiatica: an in vitro study, Biologia, 2015, vol. 70(11), pp. 1537–1544.
Kharwar, R.K. and Haldar, C., Reproductive phase dependent daily variation in melatonin receptors (Mel1a and Mel1b), androgen receptor (AR) and lung associated immunity of Perdicula asiatica, Comp. Biochem. Physiol. Pt. A, 2011, vol. 159, pp. 119–124.
Pauly, J.L., and Sokal, J.E., A simplified technique for in vitro studies of lymphocytes reactivity, Proc. Soc. Exp. Biol. Med., 1972, vol. 140, pp. 40–44.
Savaskan, E., Wirz-Justice, A., Olivieri, G., et al., Distribution of melatonin receptors immunoreactivity in human retina, J. Histochem. Cytochem., 2002, vol. 50, pp. 519–526.
Roy, D., Angelini, N.L., Fujieda, H., et al., Cyclical regulation of GnRH gene expression in GT1-7 GnRH-secreting neurons by melatonin, Endocrinol., 2001, vol. 142, pp. 4711–4720.
Treeck, O., Haldar, C., and Ortmann, O., Antiestrogens modulate MT1 melatonin receptor expression in breast and ovarian cancer cell lines, Oncol. Rep., 2006, vol. 15, pp. 231–235.
Rollag, M.D., and Niswender, G.D., Radioimmunoassay of melatonin in sheep exposed to different light regim, Endocrinol., 1976, vol. 98, pp. 482–488.
Singh, S.S., and Haldar, C., Peripheral melatonin modulates seasonal immunity and reproduction of Indian tropical male bird (Perdicula asiatica), Comp. Biochem. Physiol. A, 2007, vol. 146, pp. 446–450.
Sudhakumari, C.C., Haldar, C., and Senthilkumaran, B., Seasonal changes in adrenal and gonadal activity in the quail, Perdicula asiatica: involvement of the pineal gland, Comp. Biochem. Physiol. B, 2001, vol. 128, pp. 793–804.
Bruning, J.L. and Knitz, B.L., Computational Handbook of Statistics, Scott Foresman and Company, Illinois, 1977.
Nelson, R.J., and Drazen, D.L., Melatonin mediates seasonal adjustments in immune function, Reprod. Nut. Dev., 1999, vol. 39, pp. 383–398.
Nelson, R.J., and Demas, G.E., Role of Melatonin in Mediating Seasonal Energetic and Immunologic Adaptations, Brain Res. Bull., 1997, vol. 44(4), pp. 423–430.
Bonier, F., Martin, P.R., Moore, I.T., and Wingfield, J.C., Do baseline glucocorticoids predict fitness? Trends Ecol. Evol., 2009, vol. 24(11), pp. 634–642.
Montiel, M., Bonilla, E., Valero, N., et al., Melatonin decreases brain apoptosis, oxidative stress, and CD200 expression and increased survival rate in mice infected by Venezuelan equine encephalitis virus, Antivir. Chem. Chemother., 2015, vol. 24(3–4), pp. 99–108.
Kaiser, P., Wu, Z., Rothwell, L., et al., Prospects for understanding immune-endocrine interactions in the chicken, Gen. Comp. Endocrinol., 2009, vol. 163, pp. 83–91.
Oppong, E., Cato, A.C., Effects of glucocorticoids in the immune system, Adv. Exp. Med. Biol., 2015, vol. 872, pp. 217–233.
Liberman, A.C., Budzinski, M.L., Sokn, C., Gobbini, R.P., Steininger, A., and Arzt, E., Regulatory and mechanistic actions of glucocorticoids on T and inflammatory cells, Front. Endocrinol., 2018, vol. 9, p. 235.
Varga, G., Ehrchen J., Brockhausen, A., et al., Immune suppression via glucocorticoid-stimulated monocytes: a novel mechanism to cope with inflammation, The J. Immunol., 2014, vol. 193, pp. 1090–1099.
Trout, J.M., Mashaly, M.M., and Siegel, H.S., Changes in the profiles of circulating white blood cells, corticosterone, T3 and T4 during the initiation of humoral immunity in immature male chickens, Dev. Comp. Immunol., 1988, vol. 12, pp. 331–346.
Haldar, C., Rai, S., and Singh, R., Melatonin blocks dexamethasone induced immunosuppression in a seasonally breeding rodent Indian palm squirrel, Funambulus pennanti, Steroids, 2004, vol. 69, pp. 367–377.
Maestroni, G.J., Conti, A., and Pierpaoli, W., Role of pineal gland in immunity: circadian synthesis and release of melatonin modulates the antibody response and antagonizes the immune suppressive effect of corticosterone, J. Neuroimmunol., 1986, vol. 13, pp. 19–30.
Vishwas, D.K., Mukherjee, A., and Haldar, C., Melatonin improves humoral and cell-mediated immune responses of male golden hamster following stress induced by dexamethasone, J. Neuroimmunol., 2013, vol. 259, pp. 17–25.
Nelson, R.J., Demas, G.E., and Klein S.L., Photoperiodic mediation of seasonal breeding and immune function in rodents: a multi-factorial approach, Amer.Zool., 1998, vol. 38, pp. 226–237.
Rafii, El., Idrissi, M., Calvo, J.R., Pozo, D., et al., Specific binding of 2-[125I] iodomelatonin by rat splenocytes: characterization and its role on regulation of cyclic AMP production, J. Neuroimmunol., 1995, vol. 57, pp. 171–178.
Calvo, J.R., Rafii, El, Idrissi, M., Pozo, D., and Guerrero, G.M., Immunomodulatory role of melatonin: specific binding sites in human and rodent lymphoid cells, J. Pineal Res., 1995, vol. 18, pp. 119–126.
Guerrero, J.M., and Reiter, R.J., A brief survey of pineal gland–immune system interrelationships, Endocr. Res., 1992, vol. 18, pp. 91–113.
Maestroni, G.J., The immunoneuroendocrine role of melatonin, J. Pineal Res., 1993, vol. 14, pp. 1–10.
Maestroni, G.J., The photoperiod transducer melatonin and the immune–hematopoietic system, J. Photochem. Photobiol., 1998, vol. 43, pp. 186–192.
Lissoni, P., The pineal gland as a central regulator of cytokine network, Neuroendocrinol. Letters, 1999, vol. 20, pp. 343–349.
Skwarlo-Sonta, K., Reciprocal interdependence between pineal gland and avian immune system, Neuroendocrinol. Letters, 1999, vol. 20, pp. 151–156.
Reiter, R.J., Tan, D.X., Osuna, C., et al., Actions of melatonin in the reduction of oxidative stress: a review, J. Biomed. Sci., 2000, vol. 7, pp. 444–458.
Moore, C.B., and Siopes, T.D., Effect of lighting conditions and melatonin supplementation of the cellular and humoral immune responses in Japanese quail (Coturnix coturnix japonica), Gen. Comp. Endocrinol., 2000, vol. 119, pp. 95–104.
Haldar, C., Singh, R., and Guchait, P., Relationship between the annual rhythm in melatonin and immune system in the tropical palm squirrel (Funambulus pennanti), Chronobiol. Int., 2001, vol. 18, pp. 61–69.
Sainz, R.M., Mayo, J.C., Uria, H., et al., The pineal neurohormone melatonin prevents in vivo and in vitro apoptosis in thymocytes, J. Pineal. Res., 1995, vol. 19, pp. 179–188.
Zawilska, J.B., and Sadowska, M., Prolonged treatment with glucocorticoid dexamethasone suppresses melatonin production by the chick pineal gland and retina, Pol. J. Pharmacol., 2002, vol. 54, pp. 61–66.
Barriga, C., Marchena J.M., Lea, R.W., Harvey, S., and Rodríguez, A.B., Effect of stress and dexamethasone treatment on circadian rhythms of melatonin and corticosterone in ring dove (Streptopelia risoria), Mol. Cell. Biochem., 2002, vol. 232, pp. 27–31.
Martin, L.B., Gilliam, J., Han, P., et al., Corticosterone suppresses cutaneous immune function in temperate but not tropical house sparrows, Passer domesticus, Gen. Comp. Endocrinol., 2005, vol. 140, pp. 126–135.
Loiseau, C., Sorci, G., Dano, S., et al., Effects of experimental increase of corticosterone levels on begging behavior, immunity and parental provisioning rate in house sparrows, Gen. Comp. Endocrinol., 2008, vol. 155, pp. 101–108.
Shini, S., Kaiser, P., Shini, A., et al., Biological response of chickens (Gallus gallus domesticus) induced by corticosterone and a bacterial endotoxin, Comp. Biochem. Phys. B, 2008, vol. 149, pp. 324–333.
Merrill, L., Angelier, F., O’Loghlen, A.L., et al., Sex-specific variation in brown-headed cowbird immunity following acute stress: a mechanistic approach, Oecologia, 2012, vol. 170, pp. 25–38.
Konakchieva, R., Mitev, Y., Almeida, O.F.X., and Patchev, V.K., Chronic melatonin treatment counteracts glucocorticoid-induced dysregulation of the hypothalamic–pituitary–adrenal axis in the rat, Neuroendocrinol., 1998, vol. 67, pp. 171–180.
Chang, H., and Tain, Y., Postnatal dexamethasone-induced programmed hypertension is related to the regulation of melatonin and its receptors, Steroids, 2016, vol. 108, pp. 1–6.
Poon, A.M.S., Liu, Z.M., Tang, F., et al., Cortisol decreases 2[125I] iodomelatonin binding sites in the duck thymus, Eur. J. Endocrinol., 1994, vol. 130, pp. 320–324.
Lopez-Gonzalez, M.A., Calvo, J.R., Osuna, C., et al., Interaction of melatonin with human lymphocytes: evidence for binding sites coupled to potentiation of cyclic AMP stimulated by vasoactive intestinal peptide and activation of cyclic GMP production, J. Pineal Res., 1992, vol. 12, pp. 97–104.
Lopez-Gonzalez, M.A., Calvo, J.R., Osuna, C., et al., Synergistic action of melatonin and vasoactive intestinal peptide in stimulating cyclic AMP production in human lymphocytes, J Pineal Res., 1992, vol. 12, pp. 174–180.
Lopez-Gonzalez, M.A., Calvo, J.R., Segura, J.J., et al., Characterization of melatonin binding sites in human peripheral blood neutrophils, Biotechnol. Ther., 1993, vol. 4, pp. 253–262.
ACKNOWLEDGMENTS
The possibility of using the equipment (CO2 incubator and high-speed refrigerated centrifuge) provided by Alexander von Humboldt Foundation (Germany) and financial support from the Council for Scientific and Industrial Research (New Delhi) to Rajesh Kumar Kharwar are greatly acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kharwar, R.K., Singh, V. & Haldar, C. Expression of Melatonin and Glucocorticoid Receptor Varies along with Lung-Associated Cell-Mediated Immunity in the Jungle Bush Quail Perdicula asiatica: a Trade-off between Melatonin and Dexamethasone. J Evol Biochem Phys 56, 364–380 (2020). https://doi.org/10.1134/S0022093020040079
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0022093020040079