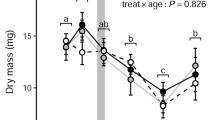
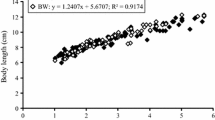
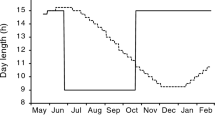
Abstract
Pink salmon hatch in fresh water, but their highly anadromous life history requires them to migrate into the ocean immediately after gravel-emergence, at a very small size. During their down-river migration these larvae undergo rapid smoltification that completely remodels their osmoregulatory physiology. At this time, the larvae reportedly have high whole-body Na+ contents and we hypothesised that the active accumulation of internal Na+ occurs in preparation for ocean entry. Using a comparative approach, the present study characterised the ontogeny of Na+ regulation in larvae of the anadromous pink salmon and the fresh-water rainbow trout. Our results indicate that larvae from both species actively accumulated Na+; however, whole-body Na+ content was higher in rainbow trout larvae compared to pink salmon. The time-course of this response was similar in the two species, with highest Na+-uptake rates (\(J^{{{\text{in}}}}_{{{\text{Na}}^{ + } }}\)) shortly after yolk sac absorption, but the mechanism of Na+ accumulation differed between the species. Rainbow trout larvae greatly increased \(J^{{{\text{in}}}}_{{{\text{Na}}^{ + } }}\) to overcompensate for a large simultaneous increase in Na+-efflux rate (\(J^{{{\text{out}}}}_{{{\text{Na}}^{ + } }}\)), whereas pink salmon mounted a smaller increase in \(J^{{{\text{in}}}}_{{{\text{Na}}^{ + } }}\) while maintaining tight control over \(J^{{{\text{out}}}}_{{{\text{Na}}^{ + } }}\), which is supported by a significantly lower paracellular permeability. Our results indicate that the transient accumulation of internal Na+ is not a unique feature of the highly anadromous life history in pink salmon and may be a common ontogenetic pattern during larval development in salmonids; and perhaps it is associated with the development of the cardiovascular system during the larvae’s transition to a more active lifestyle.




Similar content being viewed by others
Availability of data and material
All relevant data are part of this manuscript and no additional data was published in a repository.
References
Brauner CJ (2008) Acid-base balance. In: Finn RN, Kapoor BG (eds) Fish larval physiology. Science Publishers, New Hampshire, pp 185–201
Brauner CJ, Wood CM (2002a) Effect of long-term silver exposure on survival and ionoregulatory development in rainbow trout (Oncorhynchus mykiss) embryos and larvae, in the presence and absence of added dissolved organic matter. Comp Biochem Physiol C Toxicol Pharmacol 133:161–173
Brauner CJ, Wood CM (2002b) Ionoregulatory development and the effect of chronic silver exposure on growth, survival, and sublethal indicators of toxicity in early life stages of rainbow trout (Oncorhynchus mykiss). J Comp Physiol B 172:153–162
Brauner CJ, Wilson J, Kamunde C, Wood CM (2003) Water chloride provides partial protection during chronic exposure to waterborne silver in rainbow trout (Oncorhynchus mykiss) embryos and larvae. Physiol Biochem Zool 76:803–815. https://doi.org/10.1086/378136
Chasiotis H, Kelly SP (2011) Effect of cortisol on permeability and tight junction protein transcript abundance in primary cultured gill epithelia from stenohaline goldfish and euryhaline trout. Gen Comp Endocrinol 172:494–504. https://doi.org/10.1016/j.ygcen.2011.04.023
Clarke WC (1982) Evaluation of the seawater challenge test as an index of marine survival. Aquaculture 28:177–184
Clarke WC, Shelbourn JE, Ogasawara T, Hirano T (1989) Effect of initial daylength on growth, seawater adaptability and plasma growth hormone levels in underyearling coho, chinook, and chum salmon. Aquaculture 82:51–62. https://doi.org/10.1016/0044-8486(89)90395-5
Conte FP, Wagner HH (1965) Development of osmotic and ionic regulation in juvenile steelhead trout, (Salmo gairdneri). Comp Biochem Physiol 14:603–620
Eddy FB, Talbot C (1985) Sodium balance in eggs and dechorionated embryos of the Atlantic salmon Salmo salar L. exposed to zinc, aluminium and acid waters. Comp Biochem Physiol C Comp Pharmacol 81:259–266. https://doi.org/10.1016/0742-8413(85)90003-9
Evans DH, Piermarini PM, Choe KP (2005) The multifunctional fish gill: dominant site of gas exchange, osmoregulation, acid–base regulation, and excretion of nitrogenous waste. Physiol Rev 85:97–177
Fu C, Wilson JM, Rombough PJ, Brauner CJ (2010) Ions first: Na+ uptake shifts from the skin to the gills before O2 uptake in developing rainbow trout, Oncorhynchus mykiss. Proc R Soc Lond Ser B Biol Sci 277:1553–1560. https://doi.org/10.1098/rspb.2009.1545
Gallagher ZS, Bystriansky JS, Farrell AP, Brauner CJ (2013) A novel pattern of smoltification in the most anadromous salmonid: pink salmon (Oncorhynchus gorbuscha). Can J Fish Aquat Sci 70:349–357. https://doi.org/10.1139/cjfas-2012-0390
Gallagher EJ, Harter TS, Wilson JM, Brauner CJ (2020) The ontogeny of Na+ uptake in larval rainbow trout reared in waters of different Na+ content. J Comp Physiol B. https://doi.org/10.1007/s00360-020-01311-3
Grant A, Gardner M, Nendick L et al (2009) Growth and ionoregulatory ontogeny of wild and hatchery-raised juvenile pink salmon (Oncorhynchus gorbuscha). Can J Zool 87:221–228. https://doi.org/10.1139/Z08-149
Gross MR, Coleman RM, McDowall RM (1988) Aquatic productivity and the evolution of diadromous fish migration. Science 239:1291–1293
Hansen LP, Quinn TP (1998) The marine phase of the Atlantic salmon (Salmo salar) life cycle, with comparisons to Pacific salmon. Can J Fish Aquat Sci 55:104–118
Hargreaves NB, LeBrasseur RJ (1986) Size selectivity of coho (Oncorhynchus kisutch) preying on Juvenile Chum Salmon (O. keta). Can J Fish Aquat Sci 43:581–586. https://doi.org/10.1139/f86-069
Harter TS, Verreth JA, Heinsbroek LT, Schrama JW (2013) Isoenergetic replacement of fat by starch in diets for African Catfish (Clarias gariepinus): effect on water fluxes in the gastro intestinal tract. PLoS ONE 8:e55245
Heard WR (1991) Life history of pink salmon (Oncorhynchus gorbuscha). In: Groot C, Margolis L (eds) Pacific salmon life histories. UBC Press, Vancouver, pp 119–230
Hoar WS (1976) Smolt transformation: evolution, behavior, and physiology. J Fish Res Bd Can 33:1233–1252. https://doi.org/10.1139/f76-158
Holmes WN, Donaldson EM (1969) The body compartments and the distribution of electrolytes. In: Hoar WS, Randall DJ (eds) Fish physiology. Academic Press, New York, pp 1–90
Holmes WN, Stainer IM (1966) Studies on the renal excretion of electrolytes by the trout. J Exp Biol 44:33–46
Killen SS, Costa I, Brown JA, Gamperl AK (2006) Little left in the tank: metabolic scaling in marine teleosts and its implications for aerobic scope. Proc R Soc Lond Ser B Biol Sci 274:431–438
Kristiansen HR, Rankin JC (2001) Discrimination between endogeneous and exogeneus water sources in juvenile rainbow trout fed extruded dry feed. Aquat Liv Resour 14:359–366
Kumai Y, Bahubeshi A, Steele S, Perry SF (2011) Strategies for maintaining Na+ balance in zebrafish (Danio rerio) during prolonged exposure to acidic water. Comp Biochem Physiol A Mol Integr Physiol 160:52–62. https://doi.org/10.1016/j.cbpa.2011.05.001
Kwong RWM, Kumai Y, Perry SF (2013) Evidence for a role of tight junctions in regulating sodium permeability in zebrafish (Danio rerio) acclimated to ion-poor water. J Comp Physiol B 183:203–213. https://doi.org/10.1007/s00360-012-0700-9
Lauren DJ, McDonald DG (1987) Acclimation to copper by rainbow trout, Salmo gairdneri. Physiol Can J Fish Aquat Sci 44:99–104
McCormick SD, Saunders RL (1987) Preparatory physiological adaptations for marine life of salmonids: osmoregulation, growth, and metabolism. Am Fish Soc Symp I:211–229
McCormick SD, Shrimpton JM, Zydlewski JD (1996) Temperature effects on osmoregulatory physiology of juvenile anadromous fish. In: Wood CM, McDonald DG (eds) Global warming: implications for freshwater and marine fish. Cambridge University Press, Cambridge, pp 279–301
McCormick SD, Hansen LP, Quinn TP, Saunders RL (1998) Movement, migration, and smolting of Atlantic salmon (Salmo salar). Can J Fish Aquat Sci 55:77–92
McCormick SD, Regish AM, Christensen AK, Björnsson BT (2013) Differential regulation of sodium–potassium pump isoforms during smolt development and seawater exposure of Atlantic salmon. J Exp Biol 216:1142–1151. https://doi.org/10.1242/jeb.080440
Mitrovic D, Perry SF (2009) The effects of thermally induced gill remodeling on ionocyte distribution and branchial chloride fluxes in goldfish (Carassius auratus). J Exp Biol 212:843–852
Nendick L, Grant A, Gardner M et al (2009) Swimming performance and associated ionic disturbance of juvenile pink salmon Oncorhynchus gorbuscha determined using different acceleration profiles. J Fish Biol 75:1626–1638
Nendick L, Sackville M, Tang S et al (2011) Sea lice infection of juvenile pink salmon (Oncorhynchus gorbuscha): effects on swimming performance and postexercise ion balance. Can J Fish Aquat Sci 68:241–249
Nilsen TO, Ebbesson LOE, Madsen SS et al (2007) Differential expression of gill Na+/K+ α- and β-subunits, Na+, K+, 2Cl− cotransporter and CFTR anion channel in juvenile anadromous and landlocked Atlantic salmon Salmo salar. J Exp Biol 210:2885–2896. https://doi.org/10.1242/jeb.002873
Ou M, Hamilton TJ, Eom J et al (2015) Responses of pink salmon to CO2-induced aquatic acidification. Nat Clim Change 5:950
Pappenheimer JR, Reiss KZ (1987) Contribution of solvent drag through intercellular junctions to absorption of nutrients by the small intestine of the rat. J Membr Biol 100:123–136. https://doi.org/10.1007/BF02209145
Prodocimo V, Galvez F, Freire CA, Wood CM (2007) Unidirectional Na+ and Ca2+ fluxes in two euryhaline teleost fishes, Fundulus heteroclitus and Oncorhynchus mykiss, acutely submitted to a progressive salinity increase. J Comp Physiol B 177:519–528. https://doi.org/10.1007/s00360-007-0150-y
Quinn TP, Myers KW (2004) Anadromy and the marine migrations of Pacific salmon and trout: Rounsefell revisited. Rev Fish Biol Fisheries 14:421–442. https://doi.org/10.1007/s11160-005-0802-5
Rombough PJ (1999) The gill of larvae. Is it primarily a respiratory or an ionoregulatory structure? J Fish Biol 55:186–204
Rombough PJ, Garside ET (1984) Disturbed ion balance in alevins of Atlantic salmon Salmo salar chronically exposed to sublethal concentrations of cadmium. Can J Zool 62:1443–1450. https://doi.org/10.1139/z84-207
RStudioTeam (2016) RStudio: integrated development environment for R. RStudio Inc, Boston
Rudy PP, Potts WTW (1969) Sodium balance in the eggs of the Atlantic salmon, Salmo salar. J Exp Biol 50:239–246
Sackville M, Tang S, Nendick L et al (2011) Pink salmon (Oncorhynchus gorbuscha) osmoregulatory development plays a key role in sea louse (Lepeophtheirus salmonis) tolerance. Can J Fish Aquat Sci 68:1087–1096
Sackville M, Wilson JM, Farrell AP, Brauner CJ (2012) Water balance trumps ion balance for early marine survival of juvenile pink salmon (Oncorhynchus gorbuscha). J Comp Physiol B 182:781–792
Schmidt-Nielsen K (1972) Locomotion: energy cost of swimming, flying, and running. Science 177:222–228
Segar JL, Grobe CC, Grobe JL (2020) Fetal storage of osmotically inactive sodium. Am J Physiol Regul Integr Comp Physiol 318:R512–R514. https://doi.org/10.1152/ajpregu.00336.2019
Smith GR, Stearley RF (1989) The classification and scientific names of rainbow and cutthroat trouts. Fisheries 14(1):4–10
Takahashi K, Hatta N, Sugawara Y, Ryuhei S (1979) Organogenesis and functional revelation of alimentary tract and kidney of chum salmon. Tohoku J Agric Res 29:98–109
Tytler P, Tatner M, Findlay C (1990) The ontogeny of drinking in the rainbow trout, Oncorhynchus mykiss (Walbaum). J Fish Biol 36:867–875
Varnavsky VS, Kalinin SV, Kinas NM, Rostomova SS (1992) The early sea life of coho, Oncorhynchus kisutch, and pink salmon, O. gorbuscha, as a period of completion of smoltification. Environ Biol Fish 34:401–408
Varnavsky VS, Kinas NM, Rostomova SA (1993) Development of seawater adaptation in pink salmon, Oncorhynchus gorbuscha, during downstream migration: relationships to temperature and residual yolk. Environ Biol Fishes 36:373–379. https://doi.org/10.1007/BF00012415
Varsamos S, Nebel C, Charmantier G (2005) Ontogeny of osmoregulation in postembryonic fish: a review. Comp Biochem Physiol A Mol Integr Physiol 141:401–429
Zimmer AM, Wright PA, Wood CM (2014) What is the primary function of the early teleost gill? Evidence for Na+/NH4+ exchange in developing rainbow trout (Oncorhynchus mykiss). Proc R Soc Lond Ser B Biol Sci 281:20141422
Acknowledgements
Dedicated to the memory of Emily Gallagher who sadly passed away on June 22nd, 2015, at the young age of 27. Emily conducted all experiments, analysed and interpreted the data and wrote a first version of the manuscript as her MSc. thesis. Thanks are due to Sara Northrup and the Fraser Valley Trout Hatchery for providing the rainbow trout embryos and the Quinsam River Hatchery for the generous donation of pink salmon embryos. We thank Patrick Tamkee and Victor Chan for their assistance with fish care.
Funding
This study was supported by a Natural Sciences and Engineering Research Council of Canada (NSERC) Discovery Grant (261924–13) and an Accelerator Supplement (446005–13) to CJB.
Author information
Authors and Affiliations
Contributions
EJG and CJB conceptualized the study; EJB carried out all experiments, analysed the data and wrote a first version of this manuscript as her MSc thesis under the supervision of CJB; TSH analysed data, produced the figures and wrote this manuscript with editorial input from CJB.
Corresponding author
Ethics declarations
Conflicts of interest/Competing interests
We declare no conflict of interest or competing interests.
Ethical approval
Animal housing and all procedures were in strict compliance with the guidelines of the Canadian Council on Animal Care (CCAC) and were approved by The University of British Columbia (Protocol No. A11-0235).
Consent to participate
All authors approve this work and consented to participate.
Consent for publication
All authors consent to the publication of this work.
Code availability
Not applicable.
Additional information
Communicated by Bernd Pelster.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gallagher, E.J., Harter, T.S. & Brauner, C.J. The ontogeny of Na+ balance during rapid smoltification in pink salmon (Oncorhynchus gorbuscha). J Comp Physiol B 191, 17–28 (2021). https://doi.org/10.1007/s00360-020-01309-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00360-020-01309-x