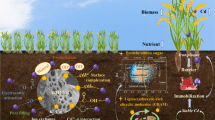
Abstract
Release kinetics governs the detachment and migration of potentially toxic trace elements (PTEs) over time between the solid phase and soil solutions. The assessment of PTEs’ release behavior through time supports more accurate prediction models of toxicity in highly polluted soils. Recently, biochar, a charcoal black carbon ameliorant, was extensively applied for decreasing the release of Zn and Pb to the soil solution. This study was arranged during a short-term (45 days) incubation experiment to estimate the possible effects of walnut leaves (WL) and their biochars produced at three temperatures (200 (B200), 400 (B400), and 600 (B600) °C) on the kinetics of Zn and Pb release in a naturally calcareous and highly polluted soil. Results showed that the biochars could reduce the release of Zn and Pb to the soil solution. The rate of Zn release (“b” in power function) at different biochar levels was 0.35 (2% B600)–0.38 (control) (mg kg−1)−1, respectively; also, the release of Pb was 0.23 (2% B600)–0.33 (control) (mg kg−1)−1, respectively. The results illustrated that a lower concentration of these metals was associated with exchangeable and oxides of Fe-Mn, whereas the higher content of residual fractions of Zn and Pb reduced the release of Zn and Pb in soils treated with biochar. Overall, the more the level and temperature of biochar increased, stabilizing PTEs through changing the distribution of metals in the soil, the more biochars succeeded significantly in lessening the release of PTEs, which ultimately could retard the further toxicity in the soil solution.



Similar content being viewed by others
References
Abdallah MM, Ahmad MN, Walker G, Leahy JJ, Kwapinski W (2019) Batch and continuous systems for Zn, Cu, and Pb metal ions adsorption on spent mushroom compost biochar. Ind Eng Chem Res 58:7296–7307. https://doi.org/10.1021/acs.iecr.9b00749
Ahmad Z, Gao B, Mosa A, Yu H, Yin X, Bashir A, Ghoveisi H, Wang S (2018) Removal of Cu(II), Cd(II) and Pb(II) ions from aqueous solutions by biochars derived from potassium-rich biomass. J Clean Prod 180:437–449. https://doi.org/10.1016/j.jclepro.2018.01.133
Allen ER, Ming DW, Hossner LR, Henninger DL (1995) Modeling transport kinetics in clinoptilolite-phosphate rock systems. Soil Sci Soc Am J 59:248–255. https://doi.org/10.2136/sssaj1995.03615995005900010039x
Bogusz A, Oleszczuk P (2020) Effect of biochar addition to sewage sludge on cadmium, copper and lead speciation in sewage sludge-amended soil. Chemosphere 239:124719. https://doi.org/10.1016/j.chemosphere.2019.124719
Bogusz A, Oleszczuk P, Dobrowolski R (2017) Adsorption and desorption of heavy metals by the sewage sludge and biochar-amended soil. Environ Geochem Health 41(4):1663–1674. https://doi.org/10.1007/s10653-017-0036-1
Boostani HR, Najafi-Ghiri M, Amin H, Mirsoleimani A (2019) Zinc desorption kinetics from some calcareous soils of orange (Citrus sinensis L.) orchards, southern Iran. Soil Sci Plant Nutr 65:20–27. https://doi.org/10.1080/00380768.2018.1554951
Brunori C, Cremisini C, Annibale LD, Massanisso P, Pinto V (2005) A kinetic study of trace element leachability from abandoned minepolluted soil treated with SS-MSW compost and red mud comparison with results from sequential extraction. Anal Bioanal Chem 381:1347–1354. https://doi.org/10.1007/s00216-005-3124-5
Cheng S, Huang CY, Lin YC, Lin SC, Chen KL (2015) Phytoremediation of lead using corn in contaminated agricultural land-an in situ study and benefit assessment. Ecotoxicol Environ Saf 111:72–77. https://doi.org/10.1016/j.ecoenv.2014.09.024
Chibuike GU, Obiora SC (2014) Heavy metal polluted soils: effect on plants and bioremediation methods. Appl Environ Soil Sci 2014:1–12. https://doi.org/10.1155/2014/752708
Cui L, Noerpel MR, Scheckel KG, Ippolito JA (2019) Wheat straw biochar reduces environmental cadmium bioavailability. Environ Int 126:69–75. https://doi.org/10.1016/j.envint.2019.02.022
Degryse F, Smolders E, Parker DR (2009) Partitioning of metals (Cd, Co, Cu, Ni, Pb, Zn) in soils: concepts, methodologies, prediction and applications - a review. Eur J Soil Sci 60:590–612. https://doi.org/10.1111/j.1365-2389.2009.01142.x
Ding W, Dong X, Ime IM, Gao B, Ma LQ (2014) Pyrolytic temperatures impact lead sorption mechanisms by bagasse biochars. Chemosphere 105:68–74. https://doi.org/10.1016/j.chemosphere.2013.12.042
Dokht HF, Dordipour E, Alireza S, Naeini M (2017) Adsorption and desorption of lead in Iranian acid and alkaline soils amended with sewage sludge-derived biochar. J Adv Environ Health Res 5:59–69. https://doi.org/10.22102/jaehr.2017.71682
Fangueiro D, Bermond A, Santos E, Carapuca H, Duarte A (2005) Kinetic approach to heavy metal mobilization assessment in sediments: choose of kinetic equations and models to achieve maximum information. Talanta 66:844–857. https://doi.org/10.1016/j.talanta.2004.12.036
Farshadirad A, Hosseinpur A, Motaghian H, Ghorbani S (2017) Release kinetics and distribution of lead in various size fraction of aggregates. J Soils and Sediments 18(1):179–188. https://doi.org/10.1007/s11368-017-1722-4
Feng C, Chen Y, Zhang S, Wang G, Zhong Q, Zhou W, Xu X, Li T (2020) Removal of lead, zinc and cadmium from contaminated soils with two plant extracts: mechanism and potential risks. Ecotoxicol Environ Saf:109829. https://doi.org/10.1016/j.ecoenv.2019.109829
Gismera MJ, Lacal J, Da Silva P, Garcia R, Teresa Sevilla M, Procopio JR (2004) Study of metal fractionation in river sediments. A comparison between kinetic and sequential extraction procedures. Environ Pollut 127:175–182. https://doi.org/10.1016/j.envpol.2003.08.004
Han FX, Banin A (2000) Long-term transformations of cadmium, cobalt, copper, nickel, zinc, vanadium, manganese, and iron in arid-zone soils under saturated condition. Commun Soil Sci Plant Anal 31:943–957. https://doi.org/10.1080/00103620009370489
Havlin JL, Westfall DG, Olsen SR (1985) Mathematical models for potassium release kinetics in calcareous soils. Soil Sci Soc Am J 49:371–376. https://doi.org/10.2136/sssaj1985.03615995004900020020x
Houben D, Evrard L, Sonnet P (2013) Mobility, bioavailability and pH-dependent leaching of cadmium, zinc and lead in a contaminated soil amended with biochar. Chemosphere 92:1450–1457. https://doi.org/10.1016/j.chemosphere.2013.03.055
Hussain S, Maqsood MA, Rahmatullah (2011) Zinc release characteristics from calcareous soils using diethylenetriaminepentaacetic acid and other organic acids. Commun Soil Sci Plant Anal 42:1870–1881. https://doi.org/10.1080/00103624.2011.587571
Inyang M, Gao B, Ding W, Pullammanappallil P, Zimmerman AR, Cao X (2011) Enhanced lead sorption by biochar derived from anaerobically digested sugarcane bagasse. Sep Sci Technol 46:1950–1956. https://doi.org/10.1080/01496395.2011.584604
Inyang M, Gao B, Yao Y, Xue Y, Zimmerman AR, Pullammanappallil P, Cao X (2012) Removal of heavy metals from aqueous solution by biochars derived from anaerobically digested biomass. Bioresour Technol 110:50–56. https://doi.org/10.1016/j.biortech.2012.01.072
Jalali M, Majeri M, Najafi S (2019) Kinetic release and fractionation of cobalt in some calcareous soils. J Geochem Explor 204:131–141. https://doi.org/10.1016/j.gexplo.2019.02.006
Kabiri P, Motaghian H, Hosseinpur A (2019) Effects of walnut leaves biochars on lead and zinc fractionation and phytotoxicity in a naturally calcareous highly contaminated soil. Water, Air, Soil Pollut:230. https://doi.org/10.1007/s11270-019-4316-5
Karimi A, Moezzi A, Chorom M, Enayatizamir N (2019) Chemical fractions and availability of Zn in a calcareous soil in response to biochar amendments. J Soil Sci Plant Nutr 19:851–864. https://doi.org/10.1007/s42729-019-00084-1
Karimi A, Moezzi A, Chorom M, Enayatizamir N (2020) Application of biochar changed the status of nutrients and biological activity in a calcareous soil. J Soil Sci Plant Nutr 20:450–459. https://doi.org/10.1007/s42729-019-00129-5
Kołodyńska D, Krukowsk JA, Thomas P (2017) Comparison of sorption and desorption studies of heavy metal ions from biochar and commercial active carbon. Chem Eng J 307:353–363. https://doi.org/10.1016/j.cej.2016.08.088
Kotoky P, Bora BJ, Baruah NK, Baruah J, Baruah P, Borah GC (2003) Chemical fractionation of heavy metals in soils around oil installations, Assam. Chem Spec Bioavailab 15:115–126. https://doi.org/10.3184/095422903782775181
Kouassi NLB, Yao KM, Sangare N, Trokourey A, Soro Metongo B (2019) The mobility of the trace metals copper, zinc, lead, cobalt, and nickel in tropical estuarine sediments, Ebrie Lagoon, Côte d’Ivoire. J Soils and Sediments 19:929–944. https://doi.org/10.1007/s11368-018-2098-9
Lahori AH, Mierzwa-Hersztek M, Rashid M, Ali Kalhoro S (2020) Residual effects of tobacco biochar along with different fixing agents on stabilization of trace elements in multi-metal contaminated soils. J Environ Sci 87:299–309. https://doi.org/10.1016/j.jes.2019.07.003
Li LY (2006) Removal of multiple-metals from contaminated clay minerals. Environ Tech 27(7):811–822. https://doi.org/10.1080/09593332708618694
Lu HP, Li ZA, Gascó G, Méndez A, Shen Y, Paz-Ferreiro J (2018) Use of magnetic biochars for the immobilization of heavy metals in a multi-contaminated soil. Sci Total Environ 622:892–899. https://doi.org/10.1016/j.scitotenv.2017.12.056
Ma L, Xu R, Jiang J (2010) Adsorption and desorption of Cu(II) and Pb(II) in paddy soils cultivated for various years in the subtropical China. J Environ Sci 22:689–695
Marschner H (1995) Mineral nutrition of higher plants, 2nd edn. Academic Press, London https://doi.org/10.1016/C2009-0-63043-9
McLaren RG, Ritchie GSP (1993) The long-term fate of copper fertilizer applied to a lateritic sandy soil in Western Australia. Aust J Soil Res 31:39–50. https://doi.org/10.1071/SR9930039
Mohseni A, Reyhanitabar A, Najafi N, Oustan S, Bazargan K (2018) Kinetics of DTPA extraction of Zn, Pb, and Cd from contaminated calcareous soils amended with sewage sludge. Arab J Geosci 11:384. https://doi.org/10.1007/s12517-018-3735-8
Moore F, Javad M, Nematollahi N, Keshavarzi B (2015) Heavy metals fractionation in surface sediments of Gowatr bay-Iran. Environ Monit Asses 187:4117. https://doi.org/10.1007/s10661-014-4117-7
Moral R, Gilkes RJ, Jordán MM (2005) Distribution of heavy metals in calcareous and non-calcareous soils in Spain. Water Air Soil Pollut 162:127–142. https://doi.org/10.1007/s11270-005-5997-5
Motaghian HR, Hosseinpur AR (2013) Zinc desorption kinetics in wheat (Triticum Aestivum L.) rhizosphere in some sewage sludge amended soils. J Soil Sci Plant Nutr 13:664–678. https://doi.org/10.4067/S0718-95162013005000053
Motaghian H, Hosseinpur A, Safian M (2020) The effects of sugarcane-derived biochar on phosphorus release characteristics in a calcareous soil. J Soil Sci Plant Nutr 20:66–74. https://doi.org/10.1007/s42729-019-00101-3
Olama V, Ronaghi A, Karimian N, Ghasemi-Fasaei R, Yasrebi J, Tavajjoh M (2010) Copper release behavior in two calcareous soils amended with three organic materials. Commun Soil Sci Plant Anal 41:2448–2458. https://doi.org/10.1080/00103624.2010.511376
Petrov N, Budinova T, Khavesov I (1992) Adsorption of the ions of zinc, cadmium, copper, and lead on oxidized anthracite. Carbon 30:135–139. https://doi.org/10.1016/0008-6223(92)90072-5
Reyhanitabar A, Gilkes RJ (2010) Kinetics of DTPA extraction of zinc from calcareous soils. Geoderma 154:289–293. https://doi.org/10.1016/j.geoderma.2009.10.016
Sadegh L, Fekri M, Gorgin N (2012) Effects of poultry manure and pistachio compost on the kinetics of copper desorption from two calcareous soils. Arab J Geosci 5:571–578. https://doi.org/10.1007/s12517-010-0211-5
Siva Subramanian S, Talibudeen O (1972) Potassium aluminium exchange in acid soils I. Kinetics J Soil Sci 23:163–176. https://doi.org/10.1111/j.1365-2389.1972.tb01651.x
Sposito G, Chang LJ, Lund AC (1982) Trace metal chemistry in arid-zone field soils amended with sewage sludge: I. fractionation of Ni, Cu, Zn, Cd, and Pb in solid phases. Soil Sci Soc Am J 46(2):260–264. https://doi.org/10.2136/sssaj1982.03615995004600020009x
Staff SS (2014) Keys to soil taxonomy, 12th edn. USDA-Natural Resources, DC, Washington
Sun J, Cui L, Quan G, Yan J, Wang H, Wu L (2020) Effects of biochar on heavy metals migration and fractions changes with different soil types in column experiments. BioResources 15(2):4388–4406. https://doi.org/10.15376/biores.15.2.4388-4406
Taghdis S, Hejazi Mehrizi M, Jalali V (2016) Effect of oxalic and citric acids on zinc release kinetic in two calcareous soils. Commun Soil Sci Plant Anal 47:2479–2489. https://doi.org/10.1080/00103624.2016.1254789
Tessier A, Campbell PGC, Bisson M (1979) Sequential extraction procedure for the speciation of particulate trace metals. Anal Chem 51:844–851. https://doi.org/10.1021/ac50043a017
Wang S, Zimmerman AR, Li Y, Zimmerman R, Li Y, Ma L, Harris WG, Migliaccio KW (2015) Physicochemical and sorptive properties of biochars derived from woody and herbaceous biomass. Chemosphere 134:257–262. https://doi.org/10.1016/j.chemosphere.2015.04.062
Xu X, Cao X, Zhao L (2013) Comparison of rice husk-and dairy manure-derived biochars for simultaneously removing heavy metals from aqueous solutions: role of mineral components in biochars. Chemosphere 92(8):955–961. https://doi.org/10.1016/j.chemosphere.2013.03.009
Yildiztugay A, Ozfidan-Konakci C, Yildiztugay E, Kucukoduk M (2019) Biochar triggers systemic tolerance against cobalt stress in wheat leaves through regulation of water status and antioxidant metabolism. J Soil Sci Plant Nutr 19:935–947. https://doi.org/10.1007/s42729-019-00091-2
Zahedifar M, Moosavi AA (2017) Modeling desorption kinetics of the native and applied zinc in biochar-amended calcareous soils of different land uses. Environ Earth Sci 76:567. https://doi.org/10.1007/s12665-017-6895-z
Zahedifar M, Karimian N, Yasrebi J (2012) Influence of applied zinc and organic matter on zinc desorption kinetics in calcareous soils. Arch Agron Soil Sci 58:169–178. https://doi.org/10.1080/03650340.2010.507195
Funding
This study is supported by funds allocated by the Vice President for research of Shahrekord University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
ESM 1
(DOCX 24 kb)
Rights and permissions
About this article
Cite this article
Kabiri, P., Motaghian, H. & Hosseinpur, A. Impact of Biochar on Release Kinetics of Pb (II) and Zn (II) in a Calcareous Soil Polluted with Mining Activities. J Soil Sci Plant Nutr 21, 22–34 (2021). https://doi.org/10.1007/s42729-020-00336-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42729-020-00336-5