Abstract
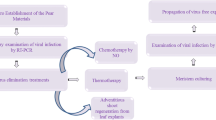
The aim of this study was to establish reliable and efficient detection and elimination methods for five apple viruses and viroids, including Apple chlorotic leaf spot virus (ACLSV), Apple stem grooving virus (ASGV), Apple stem pitting virus (ASPV), Prunus necrotic ringspot virus (PNRSV) and Apple scar skin viroid (ASSVd) from infected apple plants using thermotherapy. Based on multiple alignments of available virus or viroid characteristic genes, compatible degenerate primer pairs were designed and an efficient and sensitive detection method was developed using quantitative real-time PCR (qPCR) and was further verified by short interfering RNA (siRNA) deep sequencing. Coupled with root protecting and shoot growth promoting measures, six cultivars of potted apple plants were subjected to thermotherapy for 30 days, followed by virus detection of new emerged shoots and shoot tip grafting. The results showed that the virus elimination efficiency was over 30% for the five viruses tested for ‘Kudowu’, ‘Fuji 2001’, and ‘Devil Gala’, but only 10% for ‘Red Jonathan’ and ‘Shizuka’, while no virus-free plants were detected for ‘Shinano Gold’. Correlation analysis of the factors influencing the virus elimination showed that the elimination efficiency and the virus free plant number were significantly related to the shoot growth rate, implying a vital role of the shoot growth rate in virus elimination using thermotherapy. This study provides a new qPCR assay for five apple viruses and stresses the importance of cultivar characteristic in virus thermotherapy elimination.


Similar content being viewed by others
References
Al Rwahnih M, Daubert S, Golino D, Rowhani A (2009) Deep sequencing analysis of RNAs from a grapevine showing Syrah decline symptoms reveals a multiple virus infection that includes a novel virus. Virology 387:395–401
Cho I, Igori D, Lim S, Choi G, Hammond J, Lim H, Moon JS (2016) Deep sequencing analysis of apple infecting viruses in Korea. Plant Pathol J 32:441–451
Cieslinska M, Rutkowski KP (2008) Effect of apple chlorotic leaf spot virus on yield and quality of fruits from ’Golden Delicious’ and ’Shampion’ apple trees. Acta Hortic 781:119–124
Cui HG, Liu HZ, Chen J, Zhou JF, Qu LN, Su JM, Wang GP, Hong N (2015) Genetic diversity of Prunus necrotic ringspot virus infecting stone fruit trees grown at seven regions in China and differentiation of three phylogroups by multiplex RT-PCR. Crop Prot 74:30–36
Dar NA (2013) Apple stem grooving virus—a review article. Int J Mod Plant Anim Sci 1:28–42
Eldougdoug KA, Osman ME, Hayam SA, Rehab AD, Reham ME (2010) Elimination of Hop stunt viroid (HSVd) from infected peach and pear plants using cold therapy and chemotherapy. Aust J Basic Appl Sci 4:54–60
Grimova L, Winkowska L, Zíka L, Rysanek P (2016) Distribution of viruses in old commercial and abandoned orchards and wild apple trees. J Plant Pathol 93:549–554
Hao L, Xie JP, Chen SY, Wang SJ, Gong ZQ, Ling KS, Guo LY, Fan ZF, Zhou T (2016) A multiple RT-PCR assay for simultaneous detection and differentiation of latent viruses and apscar viroids in apple trees. J Virol Methods 234:16–21
He Y, Cai L, Zhou LL, Yang ZK, Hong N, Wang GP, Li SF, Xu WX (2017) Deep sequencing reveals the first fabavirus infecting peach. Sci Rep 7:11329
Hu G, Dong Y, Zhang Z, Fan X, Ren F, Zhou J (2015a) Virus elimination from in vitro apple by thermotherapy combined with chemotherapy. Plant Cell Tissue Organ Cult 121(2):435–443
Hu GJ, Zhang ZP, Dong YF, Fan XD, Ren F (2015b) Efficiency of virus elimination from potted apple plants by thermotherapy coupled with shoot-tip grafting. Aust Plant Pathol 44:167–173
Hu GJ, Dong YF, Zhang ZP, Fan XD, Ren F, Li ZN (2017) Efficacy of virus elimination from apple by thermotherapy coupled with in vivo shoot-tip grafting and in vitro meristem culture. J Phytopathol 165:701–706
Hull R (2002) Matthews’ plant virology, 4th edn.
Komorowska B, Siedlecki P, Kaczanowski S, Hasiowjaroszewska B, Malinowski T (2011) Sequence diversity and potential recombination events in the coat protein gene of apple stem pitting virus. Virus Res 158:263–267
Krizan B, Ondrusikova E, Holleinova V, Moravcova K, Blahova L (2009) Elimination of grapevine fanleaf virus in grapevine by in vivo and in vitro thermotherapy. Hort Sci (Prague) 36:105–108
Li BQ, Feng CH, Hu LY, Wang MR, Wang QC (2016) Shoot tip culture and cryopreservation for elimination of apple stem pitting virus (ASPV) and Apple stem grooving virus (ASGV) from apple rootstocks ’M9’ and ’M26’. Ann Appl Biol 168:142–150
Lizarraga A, Ascasíbar J, Gonzalez ML (2017) Fast and effective thermotherapy treatment for in vitro virus elimination in apple and pear trees. AJPS 08:2474–2482
Maxim A, Zagrai L, Zagrai I, Isac M (2004) Studies on the influence of apple stem grooving virus on tree growth of various apple cultivars in the nursery. Acta Hortic 657:41–44
Menzel W, Zahn V, Maiss E (2003) Multiplex RT-PCR-ELISA compared with bioassay for the detection of four apple viruses. J Virol Methods 110:153–157
Paprstein F, Sedlak J, Polak J, Svobodova L, Hassan M, Bryxiova M (2008) Results of in vitro thermotherapy of apple cultivars. Plant Cell Tissue Organ Cult 94:347–352
Payungporn S, Chutinimitkul S, Chaisingh A, Damrongwantanapokin S, Buranathai C, Amonsin A, Theamboonlers A, Poovorawan Y (2006) Single step multiplex real-time RT-PCR for H5N1 influenza A virus detection. J Virol Methods 131:143–147
Postman JD, DeNoma JS, Reed BM (2005) Detection and elimination of viruses in USDA hop (Humulus lupulus) germplasm collection. Acta Hortic 668:143–148
Sedlak J, Paprstein F, Talacko L (2011) Elimination of apple stem pitting virus from pear cultivars by in vitro chemotherapy. Acta Hortic 923:111–116
Sharma S, Dasgupta I (2012) Development of SYBR green I based real-time PCR assays for quantitative detection of rice tungro bacilliform virus and rice tungro spherical virus. J Virol Methods 181:86–92
Vivek M, Modgil M (2018) Elimination of viruses through thermotherapy and meristem culture in apple cultivar ‘Oregon Spur-II’. VirusDis 6:1–8
Walia Y, Dhir S, Ram R, Zaidi AA, Hallan V (2014) Identification of the herbaceous host range of apple scar skin viroid and analysis of its progeny variants. Plant Pathol 63:684–690
Wang MR, Li BQ, Feng CH, Wang QC (2016) Culture of shoot tips from adventitious shoots can eradicate apple stem pitting virus but fails in apple stem grooving virus. Plant Cell Tissue Organ Cult 125:283–291
Yoshikawa N, Yamagishi N, Yaegashi H, Ito T (2012) Deep sequence analysis of viral small RNAs from a green crinkle-diseased apple tree. Petria 22:292–297
Funding
This study was funded by the Beijing Agricultural Science and Technology Project [grant number 20170117], the Ministry of Agriculture of the People’s Republic of China [grant number CARS-27], and the Beijing Municipal Education Commission [grant number CEFF-PXM2017014207000043].
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
X.L. Li, M.J. Li, J. Zhou, Q.P. Wei and J.K. Zhang declare that they have no competing interests.
Caption Electronic Supplementary Material
Table S1
siRNA deep sequencing analysis for five viruses in ‘M9-T337’
Rights and permissions
About this article
Cite this article
Li, X.L., Li, M.J., Zhou, J. et al. Acquisition of Virus Eliminated Apple Plants by Thermotherapy and the Factors Influenced the Eliminating Efficiency. Erwerbs-Obstbau 62, 257–264 (2020). https://doi.org/10.1007/s10341-020-00480-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10341-020-00480-3