Abstract
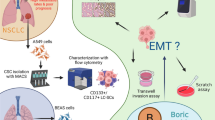
Colorectal cancer is highly metastatic. In a quarter of patients with tumors of this type, liver metastases are detected already at the time of diagnosis. Therefore, the search for drugs that can reduce the metastatic activity of cells is an important task. A tumor experiences constant stress, resulting in an increased activity of the transcription factor HSF1, which is able to trigger the synthesis of heat shock proteins. This increases the resistance of cancer cells to both stress and antitumor therapy. It has been shown recently that HSF1 plays a critical role in the process of the epithelial–mesenchymal transition (EMT) underling the formation of metastases. Here, we show that a new inhibitor of HSF1 activity, CL-43, is capable of suppressing EMT that is induced by TGFβ1. CL-43 significantly reduced the migration and proliferative potency of DLD1 cells treated with TGFβ1. Analysis of vimentin level showed that cell treatment with CL-43 cells reduced this EMT marker and returned the expression and localization of E-cadherin in the cells to the original pattern. These findings suggest that CL-43 has therapeutic potential for treatment of colorectal tumors.



Similar content being viewed by others
REFERENCES
Bessen, H.A., Therapeutic and toxic effects of Digitalis: William Withering, 1785, J. Emerg. Med., 1986, vol. 4, pp. 243–248.
Bradford, M.M., A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein–dye binding, Anal. Biochem., 1976, vol. 72, pp. 248–254.
Carpenter, R.L., Paw, I., Dewhirst, M.W., and Lo, H.W., Akt phosphorylates and activates HSF-1 independent of heat shock, leading to slug overexpression and epithelial-mesenchymal transition (EMT) of HER2-overexpressing breast cancer cells, Oncogene, 2015, vol. 34, pp. 546–557.
Chen, K., Qian, W., Li, J., Jiang, Z., Cheng, L., Yan, B., Cao, J., Sun, L., Zhou, C., Lei, M., Duan, W., Ma, J., Ma, Q., and Ma, Z., Loss of AMPK activation promotes the invasion and metastasis of pancreatic cancer through an HSF1-dependent pathway, Mol. Oncol., 2017, vol. 11, pp. 1475–1492.
Jacobs, A.T. and Marnett, L.J., HSF1-mediated BAG3 expression attenuates apoptosis in 4-hydroxynonenal-treated colon cancer cells via stabilization of anti-apoptotic Bcl-2 proteins, J. Biol. Chem., 2009, vol. 284, pp. 9176–9183.
Juhasz, K., Lipp, A.M., Nimmervoll, B., Sonnleitner, A., Hesse, J., Haselgruebler, T., and Balogi, Z., The complex function of Hsp70 in metastatic cancer, Cancers (Basel), 2013, vol. 6, pp. 42–66.
Kim, S.J., Lee, S.C., Kang, H.G., Gim, J., Lee, K.H., Lee, S.H., and Chun, K.H., Heat shock factor 1 predicts poor prognosis of gastric cancer, Yonsei Med. J., 2018, vol. 59, pp. 1041–1048.
Liao, T.T. and Yang, M.H., Revisiting epithelial-mesenchymal transition in cancer metastasis: the connection between epithelial plasticity and stemness, Mol. Oncol., 2017, vol. 11, pp. 792–804.
Nikotina, A.D., Koludarova, L., Komarova, E.Y., Mikhaylova, E.R., Aksenov, N.D., Suezov, R., Kartzev, V.G., Margulis, B.A., and Guzhova, I.V., Discovery and optimization of cardenolides inhibiting HSF1 activation in human colon HCT-116 cancer cells, Oncotarget, 2018, vol. 9, pp. 27 268–27 279.
Paul, S., Ghosh, S, .Mandal, S., Sau, S., and Pal, M., NRF2 transcriptionally activates the heat shock factor 1 promoter under oxidative stress and affects survival and migration potential of MCF7 cells, J. Biol. Chem., 2018, vol. 293, pp. 19303–19316.
Qin, G., Luo, M., Chen, J., Dang, Y., Chen, G., Li, L., Zeng, J., Lu, Y., and Yang, J., Reciprocal activation between MMP-8 and TGF-β1 stimulates EMT and malignant progression of hepatocellular carcinoma, Cancer Lett., 2016, vol. 374, pp. 85–95.
Riihimäki, M., Hemminki, A., Sundquist, J., and Hemminki, K., Patterns of metastasis in colon and rectal cancer, Sci. Rep., 2016, vol. 6, p. 29765. https://doi.org/10.1038/srep29765
Roche, J., The epithelial-to-mesenchymal transition in cancer, Cancers (Basel), 2018, vol. 10, no. 2. pii: E52. https://doi.org/10.3390/cancers10020052
Slingerland, M., Cerella, C., Guchelaar, H.J., Diederich, M., and Gelderblom, H., Cardiac glycosides in cancer therapy: from preclinical investigations towards clinical trials, Invest. New Drugs, 2013, vol. 31, pp. 1087–1094.
Tang, Y., Herr, G., Johnson, W., Resnik, E., and Aho, J., Induction and analysis of epithelial to mesenchymal transition, J. Vis. Exp., 2013, vol. 78. https://doi.org/10.3791/50478
Van der Geest, L.G., Lam-Boer, J., Koopman, M., Verhoef, C., Elferink, M.A., and de Wilt, J.H., Nationwide trends in incidence, treatment and survival of colorectal cancer patients with synchronous metastases, Clin. Exp. Metastasis, 2015, vol. 32, pp. 457–465.
Vega, S., Morales, A.V., Ocaña, O.H., Valdés, F., Fabregat, I., and Nieto, M.A., Snail blocks the cell cycle and confers resistance to cell death, Genes Dev., 2004, vol. 18, pp. 1131–1143.
Vilaboa, N., Boré, A., Martin-Saavedra, F., Bayford, M., Winfield, N., Firth-Clark, S., Kirton, S.B., and Voellmy, R., New inhibitor targeting human transcription factor HSF1: effects on the heat shock response and tumor cell survival, Nucleic Acids Res., 2017, vol. 45, pp. 5797–5817.
Wendt, M.K., Tian, M., and Schiemann, W.P., Deconstructing the mechanisms and consequences of TGF-β-induced EMT during cancer progression, Cell Tiss. Res., 2012, vol. 347, pp. 85–101.
Westerheide, S.D., Kawahara, T.L., Orton, K., and Morimoto, R.I., Triptolide, an inhibitor of the human heat shock response that enhances stress-induced cell death, J. Biol. Chem., 2006, vol. 281, pp. 9616–9622.
Wrighton, K.H., Lin, X., and Feng, X.H., Critical regulation of TGFbeta signaling by Hsp90, Proc. Natl. Acad. Sci. U. S. A., 2008, vol. 105, pp. 9244–9249.
Xi, C., Peng, S., Wu, Z., Zhou, Q., and Zhou, J., Toxicity of triptolide and the molecular mechanisms involved, Biomed. Pharmacother., 2017, vol. 90, pp. 531–541.
Yang, W., Feng, B., Meng, Y., Wang, J., Geng, B., Cui, Q., Zhang, H., Yang, Y., and Yang, J., FAM3C–YY1 axis is essential for TGFβ-promoted proliferation and migration of human breast cancer MDA-MB-231 cells via the activation of HSF1, J. Cell Mol. Med., 2019. https://doi.org/10.1111/jcmm.14243
Yokota, S., Kitahara, M., and Nagata, K., Benzylidene lactam compound, KNK437, a novel inhibitor of acquisition of thermotolerance and heat shock protein induction in human colon carcinoma cells, Cancer Res., 2000, vol. 60, pp. 2942–2948.
ACKNOWLEDGMENTS
We thank N. Tararova (DAPCEL Inc., United States) for providing TGFβ1 and E.V. Martova for help in setting up experiments.
Funding
This work was supported by the Russian Science Foundation (project no. 19-74-20161) and Russian Foundation for Basic Research (project no. 18-34-00973).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflict of interest. This article does not contain any studies involving animals or human participants performed by any of the authors.
Additional information
Translated by I. Fridlyanskaya
Rights and permissions
About this article
Cite this article
Nikotina, A.D., Kartsev, V.G., Margulis, B.A. et al. CL-43, a New Inhibitor of HSF1 Activity, Inhibits Epithelial-Mesenchymal Transition of DLD1 Colon Cancer Cells. Cell Tiss. Biol. 13, 418–422 (2019). https://doi.org/10.1134/S1990519X19060075
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1990519X19060075