Abstract
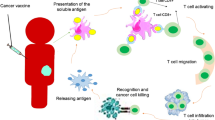
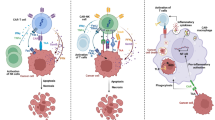
Utilization of oncolytic viruses (OVs) is a promising approach to inhibit the tumor growth. OVs exhibit both direct antitumor and indirect immunostimulatory effects. It is known that the recognition of tumor antigen by cells of the immune system is disturbed at cancer pathologies. Stimulation of dendritic cells (DCs) with tumor antigens together with viruses may enhance the generation of antigen-specific lymphocytes against tumor cells, which in turn can be an effective tool for antitumor cell technologies. The aim of this study was to clarify the possibility of using reovirus P-92 for the generation of tumor-specific T-lymphocytes in vitro. We found that the differentiation of DCs in the presence of reovirus P-92 increased the yield of mature DCs. In addition, DCs with loaded HeLa antigens after cocultivation with lymphocytes and HeLa cells did not demonstrate an active cytolytic activity. Only a cytostatic effect was observed, unlike in the case of samples with the added reovirus P-92. Thus, the antigen, which is HeLa cells cultured with reovirus P-92 under the conditions we described, can be proposed as an effective experimental product for the cellular cancer immunotherapy.


Similar content being viewed by others
REFERENCES
Andtbacka, R.H., Kaufman, H.L., Collichio, F., Amatruda, T., Senzer, N., Chesney, J., Delman, K.A., Spitler, L.E., Puzanov, I., Agarwala, S.S., Milhem, M., Cranmer, L., Curti, B., Lewis, K., Ross, M., Guthrie, T., Linette, G.P., Daniels, G.A., Harrington, K., Middleton, M.R., Miller, W.H., Jr., Zager, J.S, Ye, Y., Yao, B., Li, A., Doleman, S., VanderWalde, A., Gansert, J., and Coffin, R.S., Talimogenelaherparepvec improves durable response rate in patients with advanced melanoma, J. Clin. Oncol., 2015, vol. 33, pp. 2780—2788.
Baldueva, I.A., Novik, A.V., Moiseenko, V.M., Nexaeva, T.L., Danilova, A.B., Danilov, A.O., Procenko, S.A., Petrova, T.Yu., Ulejskaya, G.I., Shhyokina, L.A., Semyonova, A.I., Mixajlichenko, T.D., Teletaeva, G.M., Zhabina, A.S., Volkov, N.V., and Komarov, Yu.I., Clinical study (phase II) of vaccines based on autologous dendritic cells with an immunological adjuvant in patients with skin melanoma, Vopr. Onkol., 2012, vol. 58, no. 2, pp. 212–221.
Eissa, I., Bustos-Villalobos, I., Ichinose, T., Matsumura, S., Naoe, Y., MiyajimaMorimoto, D., Mukoyama, N., Zhiwen, W., Tanaka, M., Hasegawa, H., Sumigama, S., Aleksic, B., Kodera, Y., and Kasuya, H., The current status and future prospects of oncolytic viruses in clinical trials against melanoma, glioma, pancreatic, and breast cancers, Cancers, 2018, vol. 10, pp. 356–378.
Gabdrakhmanova, A.F., Viral oncolysis, Tavr. Nauchn. Obozr., 2017, vol. 1, no. 18, pp. 151–153.
Heinrich, B., Klein, J., Delic, M., Goepfert, K., Engel, V., Geberzahn, L., Lusky, M., Erbs, P., Preville, X., and Moehler, M., Immunogenicity of oncolytic vaccinia viruses JX-GFP and TG6002 in a human melanoma in vitro model: studying immunogenic cell death, dendritic cell maturation and interaction with cytotoxic T lymphocytes, Onco Targets Ther., 2017, vol. 10, pp. 2389–2401.
Kolpakov, S.A. and Kolpakova, E.P., Features of the isolation of reovirus RNA from clinical material, Zhiv. Biokosn. Sist., 2014, vol. 10. http://jbks.ru/archive/issue-10/article-5.
Marelli, G., Howells, A., Lemoine, N.R., and Wang, Y., Oncolytic viral therapy and the immune system: a double-edged sword against cancer, Front. Immunol., 2018, vol. 9, pp. 866–873.
Netesov, S.V., Kochneva, G.V., Loktev, V.B., Svyatchenko, V.A., Sergeev, A.N., Ternovoj, V.A., Tikunova, N.V., Shishkina, L.N., and Chumakov, P.M., Oncolytic viruses: achievements and challenges. Medical alphabet, Epidemiol. Sanit., 2011, vol. 3, pp. 26–33.
Ottolino-Perry, K., Diallo, J.S., Lichty, B.D., Bell, J.C., and McCart, J.A., Intelligent design: combination therapy with oncolytic viruses, Mol. Ther., 2010, vol. 18, pp. 251–263.
Razumov, I.A., Svyatchenko, V.A., Protopopova, E.V., Kochneva, G.V., Kiselyov, N.N., Gubanova, N.V., Shilov, A.G., Mordvinov, V.A., Netyosov, S.V., Chumakov, P.M., and Loktev, V.B., Evaluation of the oncolytic activity of a number of orthopoxviruses, adenoviruses and parvoviruses in relation to human gliomas, Vestn. Ross. Akad. Med. Nauk, 2013, vol. 12, pp. 4–8.
Santry, L.A., McAusland, T.M., Susta, L., Wood, G.A., Major, P.P., Petrik, J.J., Bridle, B.W., and Wootton, S.K., Production and purification of high-titer Newcastle disease virus for use in preclinical mouse models of cancer, Mol. Ther. Meth. Clin. Dev., 2018, vol. 9, pp. 181–191.
Schirrmacher, V., From chemotherapy to biological therapy: a review of novel concepts to reduce the side effects of systemic cancer treatment (review), Int. J. Oncol., 2018, vol. 54, pp. 407–419.
Schirrmacher, V., Lorenzen, D., Van, Gool, S.W., and Stuecker, W., A new strategy of cancer immunotherapy combining hyperthermia/oncolytic virus pretreatment with specific autologous anti-tumor vaccination. A review, Austin Oncol. Case Rep., 2017, vol. 2, no. 1, pp. 1–8.
Sitkovskaya, A.O., Zlatnik, E.Yu., Novikova, I.A., and Kit, O.I., Newcastle disease virus and immunity is an effective alliance against cancer (literature review), Sib. Onkol. Zh., 2018a, vol. 17, no. 6, pp. 105–113.
Sitkovskaya, A.O., Zlatnik, E.Yu., Novikova, I.A., Kolpakov, S.A., Kolpakova, E.P., Potemkin, D.S., Vasilchen-ko, N.G., Triandafilidi, E.I., and Bykadorova, O.V., Study of the possibility of in vitro oncolytic effects of the viruses of the families Reoviridae and Paramyxoviridae, Izv. Vysch. Uchebn. Zaved., Severo-Kavkaz. Reg.,Estestv. Nauki, 2018b, vol. 4, no. 200, pp. 124–130.
Teng, M.W., Ngiow, S.F., Ribas, A., and Smyth, M.J., Classifying cancers based on T-cell infiltration and PD-L1, Cancer Res., 2015, vol. 75, pp. 2139–2145.
Vasaturo, A., Di, Blasio, S., Peeters, D.G., de, Koning, C.C., de, Vries, J.M., Figdor, C.G., and Hato, S.V., Clinical implications of co-inhibitory molecule expression in the tumor microenvironment for DC vaccination: a game of stop and go, Front. Immunol., 2013, vol. 4, p. 417. https://doi.org/10.3389/fimmu.2013.00417
Vodolazhsky, D.I., Kit, O.I., Mogushkova, Kh.A., Pushkin, A.A., and Timoshkina, N.N., Cancer testicular antigens in immunotherapy of malignant tumors, Sib. Onkol. Zh., 2017, vol. 16, no. 2, pp. 71–81.
Zlatnik, E.Y., Sitkovskaya, A.O., Nepomnyashchaya, E.M., Jandigova, F.R., and Vashchenko, L.N., Achievements and perspectives of cell technologies based on activated lymphocytes in the treatment of malignant tumors, Kazan. Med. Zh., 2018, vol. 99, no. 5, pp. 792–801.
Funding
This work was performed as part of a state order (reg. no. R&D AAAA-A18-118072790017-9 “Development and Application of New Methods of Cellular Technologies for Tumor Immunotherapy”) of the Rostov Oncological Research Institute, Ministry of Health of the Russian Federation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interests. The authors declare that they have no conflict of interest.Statement on the welfare of animals. The study was approved by the Ethical Committee of the Rostov Oncological Research Institute, Ministry of Health of the Russian Federation, protocol no. 33/1 of November 29, 2018. The donors signed an informed consent.
Additional information
Translated by I. Fridlyanskaya
Abbreviations: DC—dendritic cell, DCV—dendrite-cellular vaccine, PBMC—peripheral blood mononuclear cell, OV—oncolytic virus, and SPEV—swine embryo kidney cell culture.
Rights and permissions
About this article
Cite this article
Sitkovskaya, A.O., Zlatnik, E.Y., Mezhevova, I.V. et al. The Effect of Oncolytic Reovirus P-92 on Dendritic Cell Maturation and Generation of Tumor-Specific Lymphocytes in vitro. Cell Tiss. Biol. 13, 434–438 (2019). https://doi.org/10.1134/S1990519X19060099
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1990519X19060099