Abstract
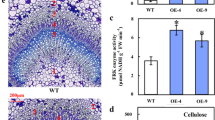
In plants, sucrose synthase (SUS, EC 2.4.1.13) is widely considered a multifunctional protein involved in modulating sink strength, cellulose biosynthesis, and carbon partitioning. However, supporting genetic evidence regarding the role of SUS from bamboo in fiber development is lacking. Here, we obtained transgenic poplar lines overexpressing the bamboo BeSUS5 gene and conducted functional analysis. We found that overexpression of BeSUS5 enhanced the activity of SUS and significantly promoted the growth of the plants, especially xylem growth. In BeSUS5 overexpressed poplar plants, the total soluble sugar (TSS) and starch contents were decreased in leaves, while the cellulose content was increased in stems, indicating that overexpression of BeSUS5 might enhance the partitioning of carbon to cellulose in poplar. Consistent with these results, the expression of cellulose biosynthesis and phloem loading–related genes, such as cellulose synthase (CesA7), KORRIGAN (KOR), and sucrose transporter (SUT1), was upregulated in transgenic plants. As a result, transgenic poplars displayed not only an increase in cell wall thickness and cell wall crystallinity but also an altered stem fiber phenotype. Taken together, our results imply the vast potential of BeSUS5 for the genetic improvement of wood cellulose production and fiber quality.









Similar content being viewed by others
References
Abbas M, Peszlen I, Shi R, Kim H, Katahira R, Kafle K, Xiang Z, Huang X, Min D, Mohamadamin M, Yang C, Dai X, Yan X, Park S, Li Y, Kim SH, Davis M, Ralph J, Sederoff RR, Chiang VL, Li Q (2019) Involvement of CesA4, CesA7-A/B and CesA8-A/B in secondary wall formation in Populus trichocarpa wood. Tree Physiology 40:73–89. https://doi.org/10.1093/treephys/tpz020
Ahmed M, Shahid AA, Akhtar S, Latif A, Din S, Fanglu M, Rao AQ, Sarwar MB, Husnain T, Xuede W (2018) Sucrose synthase genes: a way forward for cotton fiber improvement. Biologia 73:703–713. https://doi.org/10.2478/s11756-018-0078-6
Albrecht G, Mustroph A (2003) Sucrose utilization via invertase and sucrose synthase with respect to accumulation of cellulose and callose synthesis in wheat roots under oxygen deficiency. Russian Journal of Plant Physiology 50:813–820. https://doi.org/10.1023/B:RUPP.0000003280.10924.03
Ambavaram MMR, Krishnan A, Trijatmiko KR, Pereira A (2011) Coordinated activation of cellulose and repression of lignin biosynthesis pathways in rice. Plant Physiol 155:916–931. https://doi.org/10.1104/pp.110.168641
Arioli T, Peng L, Betzner AS, Burn J, Wittke W, Herth W, Camilleri C, Höfte H, Plazinski J, Birch R, Cork A, Glover J, Redmond J, Williamson RE (1998) Molecular analysis of cellulose biosynthesis in Arabidopsis. Science 279:717–720. https://doi.org/10.1126/science.279.5351.717
Baroja-Fernandez E, Munoz FJ, Li J, Bahaji A, Almagro G, Montero M, Etxeberria E, Hidalgo M, Sesma MT, Pozueta-Romero J (2012) Sucrose synthase activity in the sus1/sus2/sus3/sus4 Arabidopsis mutant is sufficient to support normal cellulose and starch production. Proceedings of the National Academy of Sciences 109:321–326. https://doi.org/10.1073/pnas.1117099109
Bieniawska Z, Paul Barratt DH, Garlick AP, Thole V, Kruger NJ, Martin C, Zrenner R, Smith AM (2007) Analysis of the sucrose synthase gene family in Arabidopsis: the sucrose synthase gene family in Arabidopsis. The Plant Journal 49:810–828. https://doi.org/10.1111/j.1365-313X.2006.03011.x
Blaschek W, Koehler H, Semler U, Franz G (1982) Molecular weight distribution of cellulose in primary cell walls: Investigations with regenerating protoplasts, suspension cultured cells and mesophyll of tobacco. Planta 154:550–555. https://doi.org/10.1007/BF00402999
Brown DM, Zeef LAH, Ellis J, Goodacre R, Turner SR (2005) Identification of novel genes in Arabidopsis involved in secondary cell wall formation using expression profiling and reverse genetics. Plant Cell 17:2281–2295. https://doi.org/10.1105/tpc.105.031542
Cassab GI (1998) Plant cell wall proteins. Annu Rev Plant Physiol Plant Mol Biol 49:281–309. https://doi.org/10.1146/annurev.arplant.49.1.281
Chiu W-B, Lin C-H, Chang C-J, Hsieh MH, Wang AY (2006) Molecular characterization and expression of four cDNAs encoding sucrose synthase from green bamboo Bambusa oldhamii. New Phytol 170:53–63. https://doi.org/10.1111/j.1469-8137.2005.01638.x
Coleman HD, Yan J, Mansfield SD (2009) Sucrose synthase affects carbon partitioning to increase cellulose production and altered cell wall ultrastructure. Proceedings of the National Academy of Sciences 106:13118–13123. https://doi.org/10.1073/pnas.0900188106
Dietze MC, Sala A, Carbone MS, Czimczik CI, Mantooth JA, Richardson AD, Vargas R (2014) Nonstructural carbon in woody plants. Annu Rev Plant Biol 65:667–687. https://doi.org/10.1146/annurev-arplant-050213-040054
Fujii S, Hayashi T, Mizuno K (2010) Sucrose synthase is an integral component of the cellulose synthesis machinery. Plant and Cell Physiology 51:294–301. https://doi.org/10.1093/pcp/pcp190
Glass M, Barkwill S, Unda F, Mansfield SD (2015) Endo-β-1,4-glucanases impact plant cell wall development by influencing cellulose crystallization: endoglucanses alter cell wall formation. J Integr Plant Biol 57:396–410. https://doi.org/10.1111/jipb.12353
Gottwald JR, Krysan PJ, Young JC, Evert RF, Sussman MR (2000) Genetic evidence for the in planta role of phloem-specific plasma membrane sucrose transporters. Proceedings of the National Academy of Sciences 97:13979–13984. https://doi.org/10.1073/pnas.250473797
Haigler CH, Ivanova-Datcheva M, Hogan PS, Salnikov VV, Hwang S, Martin K, Delmer DP (2001) Carbon partitioning to cellulose synthesis. In: Carpita NC, Campbell M, Tierney M (eds) Plant cell walls. Springer Netherlands, Dordrecht, pp 29–51
Huang Y, Liao Q, Hu S, Cao Y, Xu G, Long Z, Lu X (2018) Molecular cloning and expression analysis of seven sucrose synthase genes in bamboo ( Bambusa emeiensis ): investigation of possible roles in the regulation of cellulose biosynthesis and response to hormones. Biotechnology & Biotechnological Equipment 32:316–323. https://doi.org/10.1080/13102818.2017.1412271
Jia Z, Sun Y, Yuan L, Tian Q, Luo K (2010) The chitinase gene (Bbchit1) from Beauveria bassiana enhances resistance to Cytospora chrysosperma in Populus tomentosa Carr. Biotechnol Lett 32:1325–1332. https://doi.org/10.1007/s10529-010-0297-6
Jin Y-L, Tang R-J, Wang H-H, Jiang CM, Bao Y, Yang Y, Liang MX, Sun ZC, Kong FJ, Li B, Zhang HX (2017) Overexpression of Populus trichocarpa CYP85A3 promotes growth and biomass production in transgenic trees. Plant Biotechnology Journal 15:1309–1321. https://doi.org/10.1111/pbi.12717
Kim W-C, Kim J-Y, Ko J-H, Kim J, Han KH (2013) Transcription factor MYB46 is an obligate component of the transcriptional regulatory complex for functional expression of secondary wall-associated cellulose synthases in Arabidopsis thaliana. Journal of Plant Physiology 170:1374–1378. https://doi.org/10.1016/j.jplph.2013.04.012
Konishi T, Ohmiya Y, Hayashi T (2004) Evidence that sucrose loaded into the phloem of a poplar leaf is used directly by sucrose synthase associated with various β-Glucan synthases in the stem. Plant Physiol 134:1146–1152. https://doi.org/10.1104/pp.103.033167
Kudlicka K, Brown RM (1996) Cellulose biosynthesis in higher plants. 65:149–156
Kumar M, Turner S (2015) Plant cellulose synthesis: CESA proteins crossing kingdoms. Phytochemistry 112:91–99. https://doi.org/10.1016/j.phytochem.2014.07.009
Kumar, Manoj, Gorzsas, et al (2014) Deficient sucrose synthase activity in developing wood does not specifically affect cellulose biosynthesis, but causes an overall decrease in cell wall polymers. 203:1220–1230
Kumar M, Campbell L, Turner S (2016) Secondary cell walls: biosynthesis and manipulation. EXBOTJ 67:515–531. https://doi.org/10.1093/jxb/erv533
Lalonde S, Wipf D, Frommer WB (2004) Transport mechanisms for organic forms of carbon and nitrogen between source and sink. Annu Rev Plant Biol 55:341–372. https://doi.org/10.1146/annurev.arplant.55.031903.141758
Li M-H, Xiao W-F, Wang S-G, Cheng GW, Cherubini P, Cai XH, Liu XL, Wang XD, Zhu WZ (2008) Mobile carbohydrates in Himalayan treeline trees I. Evidence for carbon gain limitation but not for growth limitation. Tree Physiology 28:1287–1296. https://doi.org/10.1093/treephys/28.8.1287
Li S, Ge F-R, Xu M, Zhao XY, Huang GQ, Zhou LZ, Wang JG, Kombrink A, McCormick S, Zhang XS, Zhang Y (2013) Arabidopsis COBRA-LIKE 10, a GPI-anchored protein, mediates directional growth of pollen tubes. Plant J 74:486–497. https://doi.org/10.1111/tpj.12139
Li S, Bashline L, Lei L, Gu Y (2014) Cellulose synthesis and its regulation. The Arabidopsis Book 12:e0169. https://doi.org/10.1199/tab.0169
Li C, Wang X, Ran L, Tian Q, Fan D, Luo K (2015) PtoMYB92 is a Transcriptional activator of the lignin biosynthetic pathway during secondary cell wall formation in Populus tomentosa. Plant Cell Physiol 56:2436–2446. https://doi.org/10.1093/pcp/pcv157
Li M, Wang S, Liu Y, Zhang Y, Ren M, Liu L, Lu T, Wei H, Wei Z (2019) Overexpression of PsnSuSy1, 2 genes enhances secondary cell wall thickening, vegetative growth, and mechanical strength in transgenic tobacco. Plant Mol Biol 100:215–230. https://doi.org/10.1007/s11103-019-00850-w
Ma Y, Wang Y, Liu J, Lv F, Chen J, Zhou Z (2014) the effects of fruiting positions on cellulose synthesis and sucrose metabolism during cotton (Gossypium hirsutum L.) Fiber Development. PLoS ONE 9:e89476. https://doi.org/10.1371/journal.pone.0089476
Maloney VJ, Samuels AL, Mansfield SD (2012) The endo-1,4-β-glucanase Korrigan exhibits functional conservation between gymnosperms and angiosperms and is required for proper cell wall formation in gymnosperms. New Phytologist 193:1076–1087. https://doi.org/10.1111/j.1469-8137.2011.03998.x
McFarlane HE, Döring A, Persson S (2014) The cell biology of cellulose synthesis. Annu Rev Plant Biol 65:69–94. https://doi.org/10.1146/annurev-arplant-050213-040240
Mitsuda N, Seki M, Shinozaki K, Ohme-Takagi M (2005) The NAC transcription factors NST1 and NST2 of Arabidopsis regulate secondary wall thickenings and are required for anther dehiscence. Plant Cell 17:2993–3006. https://doi.org/10.1105/tpc.105.036004
Mølhøj M, Ulvskov P, Dal Degan F (2001) Characterization of a functional soluble form of a Brassica napus membrane-anchored endo-1,4-β-glucanase heterologously expressed in Pichia pastoris. Plant Physiol 127:674–684. https://doi.org/10.1104/pp.010269
Nguyen QA, Luan S, Wi SG, Bae H, Lee DS, Bae HJ (2016) Pronounced Phenotypic changes in transgenic tobacco plants overexpressing sucrose synthase may reveal a novel sugar signaling pathway. Front Plant Sci 6: https://doi.org/10.3389/fpls.2015.01216
Niu, Erli, Fang, et al (2018) Ectopic expression of GhCOBL9A, a cotton glycosyl-phosphatidyl inositol-anchored protein encoding gene, promotes cell elongation, thickening and increased plant biomass in transgenic Arabidopsis. 1–14
Persia D, Cai G, Del Casino C et al (2008) Sucrose synthase is associated with the cell wall of tobacco pollen tubes. Plant Physiol 147:1603–1618. https://doi.org/10.1104/pp.108.115956
Raessler M, Wissuwa B, Breul A, Unger W, Grimm T (2010) Chromatographic analysis of major non-structural carbohydrates in several wood species – an analytical approach for higher accuracy of data. Anal Methods 2:532. https://doi.org/10.1039/b9ay00193j
Rai V, Ghosh JS, Pal A, Dey N (2011) Identification of genes involved in bamboo fiber development. Gene 478:19–27. https://doi.org/10.1016/j.gene.2011.01.004
Robert S, Bichet A, Grandjean O, Kierzkowski D, Satiat-Jeunemaître B, Pelletier S, Hauser MT, Höfte H, Vernhettes S (2005) An Arabidopsis endo-1,4-β-d-glucanase involved in cellulose synthesis undergoes regulated intracellular cycling. Plant Cell 17:3378–3389. https://doi.org/10.1105/tpc.105.036228
Roudier F, Fernandez AG, Fujita M, Himmelspach R, Borner GHH, Schindelman G, Song S, Baskin TI, Dupree P, Wasteneys GO, Benfey PN (2005) COBRA, an Arabidopsis extracellular glycosyl-phosphatidyl inositol-anchored protein, specifically controls highly anisotropic expansion through its involvement in cellulose microfibril orientation. Plant Cell 17:1749–1763. https://doi.org/10.1105/tpc.105.031732
Ruan Y-L, Llewellyn DJ, Furbank RT (2003) Suppression of sucrose synthase gene expression represses cotton fiber cell initiation, elongation, and seed development. Plant Cell 15:952–964. https://doi.org/10.1105/tpc.010108
Ruan Y-L, Jin Y, Yang Y-J, Li GJ, Boyer JS (2010) sugar input, metabolism, and signaling mediated by invertase: roles in development, yield potential, and response to drought and heat. Molecular Plant 3:942–955. https://doi.org/10.1093/mp/ssq044
Rueda-López M, Cañas RA, Canales J, Cánovas FM, Ávila C (2015) The overexpression of the pine transcription factor PpDof5 in Arabidopsis leads to increased lignin content and affects carbon and nitrogen metabolism. Physiol Plantarum 155:369–383. https://doi.org/10.1111/ppl.12381
Scheible W-R, Pauly M (2004) Glycosyltransferases and cell wall biosynthesis: novel players and insights. Current Opinion in Plant Biology 7:285–295. https://doi.org/10.1016/j.pbi.2004.03.006
Segal L, Creely JJ, Martin AE, Conrad CM (1959) An empirical method for estimating the degree of crystallinity of native cellulose using the X-ray diffractometer. Textile Research Journal 29:786–794. https://doi.org/10.1177/004051755902901003
Singh S, Bhatt, Singha (2004) Commercial edible bamboo species of the North-Eastern Himalayan Region. India. Part I: young shoot sales. Journal of Bamboo and Rattan 3:337–364. https://doi.org/10.1163/1569159042464680
Smith AM, Kruger NJ, Lunn JE (2012) Source of sugar nucleotides for starch and cellulose synthesis. Proceedings of the National Academy of Sciences 109:E776–E776. https://doi.org/10.1073/pnas.1200878109
Somerville C (2006) Cellulose synthesis in higher plants. Annu Rev Cell Dev Biol 22:53–78. https://doi.org/10.1146/annurev.cellbio.22.022206.160206
Song X, Peng C, Zhou G, Gu H, Li Q, Zhang C (2016) Dynamic allocation and transfer of non-structural carbohydrates, a possible mechanism for the explosive growth of Moso bamboo (Phyllostachys heterocycla). Sci Rep 6:25908. https://doi.org/10.1038/srep25908
Szyjanowicz PMJ, McKinnon I, Taylor NG, Gardiner J, Jarvis MC, Turner SR (2004) The irregular xylem 2 mutant is an allele of korrigan that affects the secondary cell wall of Arabidopsis thaliana. Plant J 37:730–740. https://doi.org/10.1111/j.1365-313X.2003.02000.x
Taylor-Teeples M, Lin L, de Lucas M, Turco G, Toal TW, Gaudinier A, Young NF, Trabucco GM, Veling MT, Lamothe R, Handakumbura PP, Xiong G, Wang C, Corwin J, Tsoukalas A, Zhang L, Ware D, Pauly M, Kliebenstein DJ, Dehesh K, Tagkopoulos I, Breton G, Pruneda-Paz JL, Ahnert SE, Kay SA, Hazen SP, Brady SM (2015) An Arabidopsis gene regulatory network for secondary cell wall synthesis. Nature 517:571–575. https://doi.org/10.1038/nature14099
Updegraff DM (1969) Semimicro determination of cellulose inbiological materials. Analytical Biochemistry 32:420–424. https://doi.org/10.1016/S0003-2697(69)80009-6
Xi W, Song D, Sun J, Shen J, Li L (2017) Formation of wood secondary cell wall may involve two type cellulose synthase complexes in Populus. Plant Mol Biol 93:419–429. https://doi.org/10.1007/s11103-016-0570-8
Xu S-M, Brill E, Llewellyn DJ, Furbank RT, Ruan YL (2012) Overexpression of a potato sucrose synthase gene in cotton accelerates leaf expansion, reduces seed abortion, and enhances fiber production. Molecular Plant 5:430–441. https://doi.org/10.1093/mp/ssr090
Yang Y, Yoo CG, Winkeler KA, Collins CM, Hinchee MAW, Jawdy SS, Gunter LE, Engle NL, Pu Y, Yang X, Tschaplinski TJ, Ragauskas AJ, Tuskan GA, Chen JG (2017) Overexpression of a domain of unknown function 231-containing protein increases O-xylan acetylation and cellulose biosynthesis in Populus. Biotechnol Biofuels 10:311. https://doi.org/10.1186/s13068-017-0998-3
Yu L, Chen H, Sun J, Li L (2014) PtrKOR1 is required for secondary cell wall cellulose biosynthesis in Populus. Tree Physiology 34:1289–1300. https://doi.org/10.1093/treephys/tpu020
Zhang W, Yi Z, Huang J, Li F, Hao B, Li M, Hong S, Lv Y, Sun W, Ragauskas A, Hu F, Peng J, Peng L (2013) Three lignocellulose features that distinctively affect biomass enzymatic digestibility under NaOH and H2SO4 pretreatments in Miscanthus. Bioresource Technology 130:30–37. https://doi.org/10.1016/j.biortech.2012.12.029
Zhong R, Richardson EA, Ye Z-H (2007) The MYB46 transcription factor is a direct target of SND1 and regulates secondary wall biosynthesis in Arabidopsis. Plant Cell 19:2776–2792. https://doi.org/10.1105/tpc.107.053678
Funding
This work was supported by the National Natural Science Found of China (31400257), the Science and Technology Project of Sichuan Province, China (18YYJC0920), and the Demonstration Project of the Industrial Chain of Sichuan Province, China (19Z21842).
Author information
Authors and Affiliations
Contributions
HY and WLL designed the experiments and performed the study; HY and CY drafted the manuscript; WLL performed the transgene study; HSL and LXG proposed and supervised the overall project. All authors read and approved the final version of this manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Data archiving statement
The full-length CDS of BeSUS5 (accession number KJ525750) has been submitted to GenBank.
Additional information
Communicated by A. De La Torre
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Huang, Y., Wang, L., Hu, S. et al. Overexpression of the bamboo sucrose synthase gene (BeSUS5) improves cellulose production, cell wall thickness and fiber quality in transgenic poplar. Tree Genetics & Genomes 16, 75 (2020). https://doi.org/10.1007/s11295-020-01464-w
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11295-020-01464-w