Abstract
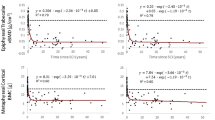
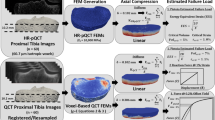
Spinal cord injury (SCI) is associated with bone fragility and fractures around the knee. The purpose of this investigation was to validate a computed tomography (CT) based finite element (FE) model of the proximal tibia and distal femur under biaxial loading, and to retrospectively quantify the relationship between model predictions and fracture incidence. Twenty-six cadaveric tibiae and femora (n = 13 each) were loaded to 300 N of compression, then internally rotated until failure. FE predictions of torsional stiffness (K) and strength (Tult) explained 74% (n = 26) and 93% (n = 7) of the variation in experimental measurements, respectively. Univariate analysis and logistic regression were subsequently used to determine if FE predictions and radiographic measurements from CT and dual energy X-ray absorptiometry (DXA) were associated with prevalent lower-limb fracture in 50 individuals with SCI (n = 14 fractures). FE and CT measures, but not DXA, were lower in individuals with fracture. FE predictions of Tult at the tibia demonstrated the highest odds ratio (4.98; p = 0.006) and receiver operating characteristic (0.84; p = 0.008) but did not significantly outperform other metrics. In conclusion, CT-based FE model predictions were associated with prevalent fracture risk after SCI; this technique could be a powerful tool in future clinical research.




Similar content being viewed by others
References
Abderhalden, L., F. M. Weaver, M. Bethel, H. Demirtas, S. Burns, J. Svircev, H. Hoenig, K. Lyles, S. Miskevics, and L. D. Carbone. Dual-energy X-ray absorptiometry and fracture prediction in patients with spinal cord injuries and disorders. Osteoporos. Int. 28:925–934, 2017.
Biering-Serensen, F., H. Bohr, and O. Schaadt. Bone mineral content of the lumbar spine and lower extremities years after spinal cord lesion. Paraplegia 26:293–301, 1988.
Biering-Sorensen, F., H. H. Bohr, and O. P. Schaadt. Longitudinal study of bone mineral content in the lumbar spine, the forearm and the lower extremities after spinal cord injury. Eur. J. Clin. Invest. 20:330–335, 1990.
Cezayirlioglu, H., E. Bahniuk, D. T. Davy, and K. G. Heiple. Anisotropic yield behavior of bone under combined axial force and torque. J. Biomech. 18:61–69, 1985.
Cirnigliaro, C. M., M. J. Myslinski, M. F. La Fountaine, S. C. Kirshblum, G. F. Forrest, and W. A. Bauman. Bone loss at the distal femur and proximal tibia in persons with spinal cord injury: imaging approaches, risk of fracture, and potential treatment options. Osteoporos. Int. 28:747–765, 2017.
Cody, D. D., G. J. Gross, F. J. Hou, H. J. Spencer, S. A. Goldstein, and D. P. Fyhrie. Femoral strength is better predicted by finite element models than QCT and DXA. J. Biomech. 32:1013–1020, 1999.
Crawford, R. P., C. E. Cann, and T. M. Keaveny. Finite element models predict in vitro vertebral body compressive strength better than quantitative computed tomography. Bone 33:744–750, 2003.
Currey, J. D. Tensile yield in compact bone is determined by strain, post-yield behaviour by mineral content. J. Biomech. 37:549–556, 2004.
Edwards, W. B., T. J. Schnitzer, and K. L. Troy. Bone mineral and stiffness loss at the distal femur and proximal tibia in acute spinal cord injury. Osteoporos. Int. 24:2461–2469, 2013.
Edwards, W. B., T. J. Schnitzer, and K. L. Troy. Torsional stiffness and strength of the proximal tibia are better predicted by finite element models than DXA or QCT. J. Biomech. 46:1655–1662, 2013.
Edwards, W. B., T. J. Schnitzer, and K. L. Troy. The mechanical consequence of actual bone loss and simulated bone recovery in acute spinal cord injury. Bone 60:141–147, 2014.
Edwards, W. B., N. Simonian, I. T. Haider, A. S. Anschel, D. Chen, K. E. Gordon, E. K. Gregory, K. H. Kim, R. Parachuri, K. L. Troy, and T. J. Schnitzer. Effects of teriparatide and vibration on bone mass and bone strength in people with bone loss and spinal cord injury: a randomized controlled trial. J. Bone Miner. Res. 33:1729–1740, 2018.
Edwards, W. B., N. Simonian, K. L. Troy, and T. J. Schnitzer. Reduction in torsional stiffness and strength at the proximal tibia as a function of time since spinal cord injury. J. Bone Miner. Res. 30:1422–1430, 2015.
Edwards, W. B., and K. L. Troy. Finite element prediction of surface strain and fracture strength at the distal radius. Med. Eng. Phys. 34:290–298, 2012.
Eser, P., A. Frotzler, Y. Zehnder, and J. Denoth. Fracture threshold in the femur and tibia of people with spinal cord injury as determined by peripheral quantitative computed tomography. Arch. Phys. Med. Rehabil. 2005. https://doi.org/10.1016/j.apmr.2004.09.006.
Eser, P., A. Frotzler, Y. Zehnder, L. Wick, H. Knecht, J. Denoth, and H. Schiessl. Relationship between the duration of paralysis and bone structure: a pQCT study of spinal cord injured individuals. Bone 34:869–880, 2004.
Frey-Rindova, P., E. D. De Bruin, E. Stüssi, M. A. Dambacher, and V. Dietz. Bone mineral density in upper and lower extremities during 12 months after spinal cord injury measured by peripheral quantitative computed tomography. Spinal Cord 38:26–32, 2000.
Garland, D., R. Adkins, and C. Stewart. Fracture threshold and risk for osteoporosis and pathologic fractures in individuals with spinal cord injury. Top. Spinal Cord Inj. Rehabil. 11:61–69, 2005.
Gordon, K. E., M. J. Wald, and T. J. Schnitzer. Effect of parathyroid hormone combined with gait training on bone density and bone architecture in people with chronic spinal cord injury. PM&R 5:663–671, 2013.
Grassner, L., B. Klein, D. Maier, V. Bühren, and M. Vogel. Lower extremity fractures in patients with spinal cord injury characteristics, outcome and risk factors for non-unions. J. Spinal Cord Med. 41:676–683, 2018.
Gray, H. A., F. Taddei, A. B. Zavatsky, L. Cristofolini, and H. S. Gill. Experimental validation of a finite element model of a human cadaveric tibia. J. Biomech. Eng. 130:031016, 2008.
Haider, I. T., S. M. Lobos, N. Simonian, T. J. Schnitzer, and W. B. Edwards. Bone fragility after spinal cord injury: reductions in stiffness and bone mineral at the distal femur and proximal tibia as a function of time. Osteoporos. Int. 30:1422–1430, 2018.
Haider, I. T., N. Simonian, A. S. Saini, F. M. Leung, W. B. Edwards, and T. J. Schnitzer. Open-label clinical trial of alendronate after teriparatide therapy in people with spinal cord injury and low bone mineral density. Spinal Cord 57:832–842, 2019.
Hayes, W. C., and D. R. Carter. Postyield behavior of subchondral trabecular bone. J. Biomed. Mater. Res. 10:537–544, 1976.
Hill, R. A theory of the yielding and plastic flow of anisotropic metals. Proc. R. Soc. A Math. Phys. Eng. Sci. 193:281–297, 1948.
Imai, K., I. Ohnishi, T. Matsumoto, S. Yamamoto, and K. Nakamura. Assessment of vertebral fracture risk and therapeutic effects of alendronate in postmenopausal women using a quantitative computed tomography-based nonlinear finite element method. Osteoporos. Int. 20:801–810, 2009.
Keating, J. F., M. Kerr, and M. Delargy. Minimal trauma causing fractures in patients with spinal cord injury. Disabil. Rehabil. 14:108–109, 1992.
Kirshblum, S. C., W. Waring, F. Biering-Sorensen, S. P. Burns, M. Johansen, M. Schmidt-Read, W. Donovan, D. Graves, A. Jha, L. Jones, M. J. Mulcahey, and A. Krassioukov. International standards for neurological classification of spinal cord injury (Revised 2011). J. Spinal Cord Med. 34:547–554, 2011.
Lala, D., B. C. Craven, L. Thabane, A. Papaioannou, J. D. Adachi, M. R. Popovic, and L. M. Giangregorio. Exploring the determinants of fracture risk among individuals with spinal cord injury. Osteoporos. Int. 25:177–185, 2014.
Lambach, R. L., N. E. Stafford, J. A. Kolesar, B. J. Kiratli, G. H. Creasey, R. S. Gibbons, B. J. Andrews, and G. S. Beaupre. Bone changes in the lower limbs from participation in an FES rowing exercise program implemented within two years after traumatic spinal cord injury. J. Spinal Cord Med. 43:306–314, 2018.
Lazo, M. G., P. Shirazi, M. Sam, A. Giobbie-Hurder, M. J. Blacconiere, and M. Muppidi. Osteoporosis and risk of fracture in men with spinal cord injury. Spinal Cord 39:208–214, 2001.
Les, C. M., S. M. Stover, J. H. Keyak, K. T. Taylor, and A. J. Kaneps. Stiff and strong compressive properties are associated with brittle post-yield behavior in equine compact bone material. J. Orthop. Res. 20:607–614, 2002.
Lewis, G. Properties of acrylic bone cement: state of the art review. J. Biomed. Mater. Res. 38:155–182, 1997.
Martínez, Á. A., J. Cuenca, A. Herrera, and J. Domingo. Late lower extremity fractures in patients with paraplegia. Injury 33:583–586, 2002.
McPherson, J. G., W. B. Edwards, A. Prasad, K. L. Troy, J. W. Griffith, and T. J. Schnitzer. Dual energy X-ray absorptiometry of the knee in spinal cord injury: methodology and correlation with quantitative computed tomography. Spinal Cord 52:821–825, 2014.
Modlesky, C. M., S. Majumdar, A. Narasimhan, and G. A. Dudley. Trabecular bone microarchitecture is deteriorated in men with spinal cord injury. J. Bone Miner. Res. 19:48–55, 2004.
Moons, K. G. M., J. A. H. de Groot, W. Bouwmeester, Y. Vergouwe, S. Mallett, D. G. Altman, J. B. Reitsma, and G. S. Collins. Critical appraisal and data extraction for systematic reviews of prediction modelling studies: the CHARMS checklist. PLoS Med. 11:15, 2014. https://doi.org/10.1371/journal.pmed.1001744.
Morgan, E. F., H. H. Bayraktar, and T. M. Keaveny. Trabecular bone modulus–density relationships depend on anatomic site. J. Biomech. 36:897–904, 2003.
Morse, L. R., K. L. Troy, Y. Fang, N. Nguyen, R. Battaglino, R. F. Goldstein, R. Gupta, and J. A. Taylor. Combination therapy with zoledronic acid and FES-row training mitigates bone loss in paralyzed legs: results of a randomized comparative clinical trial. JBMR Plus 3:e10167, 2019.
Nishiyama, K. K., S. Gilchrist, P. Guy, P. Cripton, and S. K. Boyd. Proximal femur bone strength estimated by a computationally fast finite element analysis in a sideways fall configuration. J. Biomech. 46:1231–1236, 2013.
Orwoll, E. S., L. M. Marshall, C. M. Nielson, S. R. Cummings, J. Lapidus, J. A. Cauley, K. Ensrud, N. Lane, P. R. Hoffmann, D. L. Kopperdahl, and T. M. Keaveny. Finite element analysis of the proximal femur and hip fracture risk in older men. J. Bone Miner. Res. 24:475, 2009.
Reiter, A. L., A. Volk, J. Vollmar, B. Fromm, and H. J. Gerner. Changes of basic bone turnover parameters in short-term and long-term patients with spinal cord injury. Eur. Spine J. 16:771–776, 2007.
Schileo, E., F. Taddei, L. Cristofolini, and M. Viceconti. Subject-specific finite element models implementing a maximum principal strain criterion are able to estimate failure risk and fracture location on human femurs tested in vitro. J. Biomech. 41:356–367, 2008.
Vestergaard, P., K. Krogh, L. Rejnmark, and L. Mosekilde. Fracture rates and risk factors for fractures in patients with spinal cord injury. Spinal Cord 36:790–796, 1998.
Zehnder, Y., M. Lüthi, D. Michel, H. Knecht, R. Perrelet, I. Neto, M. Kraenzlin, G. Zäch, and K. Lippuner. Long-term changes in bone metabolism, bone mineral density, quantitative ultrasound parameters, and fracture incidence after spinal cord injury: a cross-sectional observational study in 100 paraplegic men. Osteoporos. Int. 15:180–189, 2004.
Acknowledgments
This study was funded by the Department of Defense U.S. Army Medical Research and Materiel Command (Grant #: SC090010 and BA150039). Research infrastructure for this study was funded by a Canadian Foundation for Innovation (CFI) John R. Evans Leaders Fund (JELF; Project #37134).
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Eiji Tanaka oversaw the review of this article.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Haider, I.T., Simonian, N., Schnitzer, T.J. et al. Stiffness and Strength Predictions From Finite Element Models of the Knee are Associated with Lower-Limb Fractures After Spinal Cord Injury. Ann Biomed Eng 49, 769–779 (2021). https://doi.org/10.1007/s10439-020-02606-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-020-02606-w