Abstract
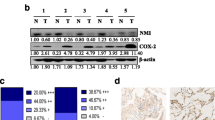
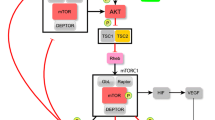
Aberrant expression of mTOR signaling pathway is significantly associated with gastric cancer. However, the effect of smoking on mTOR expression and its downstream signaling molecules in gastric cancer has not been explored. Our study aims to investigate the effect of smoking on p-mTOR and its correlation with various downstream targets and survival of the smoker and never-smoker in advanced gastric cancer patients. Forty-one smokers and 41 never-smokers patient sample with the advanced gastric carcinoma were chosen for this study. Immunohistochemistry and western blot analysis were performed to check the expression of p-mTOR and its downstream targets. The correlation of p-mTOR with its downstream targets was analyzed by linear regression analysis in Graph Pad Prism software. Survivability analysis was examined by Kaplan–Meier method with log rank test in SPSS. High expression of p-mTOR and its downstream targets were observed in advanced gastric cancer smoker patients as compared to never-smokers by immunohistochemistry and western blot analysis. Results revealed that over expressed p-mTOR in smoker patients were positively correlated with its downstream targets (P < 0.05) and poor survival (P = 0.034). Over expression of p-mTOR in gastric cancer male smoker patients had the worse outcome.




Similar content being viewed by others
Data availability
The data sets or information generated and analyzed during the present study are available in the CNCI repository. The datasets can be available from the corresponding author on reasonable requests.
References
Rawla P, Barsouk A (2019) Epidemiology of gastric cancer: global trends, risk factors and prevention. Gastroenterol Rev 14(1):26–38
Dikshit R, Gupta PC, Ramasundarahettige C et al (2012) Cancer mortality in India: a nationally representative survey. Lancet 379:1807–1816
Bray F, Ferlay J, Soerjomataram I et al (2018) Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 68(6):394–424
Jemal A, Center MM, DeSantis C, Ward EM (2010) Global patterns of cancer incidence and mortality rates and trends. Cancer Epidemiol Biomarkers Prev 19:1893–1907
Alberts SR, Cervantes A, van de Velde CJ (2003) Gastric cancer: epidemiology, pathology and treatment. Ann Oncol 14:ii31–ii36
Peleteiro B, Castro C, Morais S et al (2015) Worldwide burden of gastric cancer attributable to tobacco smoking in 2012 and predictions for 2020. Dig Dis Sci 60(8):2470–2476
Lopes RL, Pereira AK, Nogueira A et al (2008) Smoking and gastric cancer: systematic review and meta-analysis of cohort studies. Cancer Cause Control 19:689–701
Kalyani R, Das S, Kumar ML (2010) Pattern of cancer in adolescent and young adults—a ten-year study in India. Asian Pac J Cancer Prev 11:655–659
Tredaniel J, Boffetta P, Buiatti E et al (1997) Tobacco smoking and gastric cancer: review and meta-analysis. Int J Cancer 72:565–573
International Agency for Research on Cancer (2004) Tobacco smoke and involuntary smoking. In: IARC monographs on the evaluation of carcinogenic risks to humans, vol 83. IARC, Lyon
Nishino Y, Inoue M, Tsuji I et al (2006) Tobacco smoking and gastric cancer risk: an evaluation based on a systematic review of epidemiologic evidence among the Japanese population. Jpn J Clin Oncol 36:800–807
Minna JD (2003) Nicotine exposure and bronchial epithelial cell nicotinic acetylcholine receptor expression in the pathogenesis of lung cancer. J Clin Invest 111:31–33
Shin VY, Wu WK, Chu KM et al (2005) Nicotine induces cyclooxygenase-2 and vascular endothelial growth factor receptor- 2 in associationwith tumor-associated invasion and angiogenesis in gastric cancer. Mol Cancer Res 3:607–615
Nishioka T, Kim HS, Luo LY et al (2011) Sensitization of epithelial growth factor receptors by nicotine exposure to promote breast cancer cell growth. Breast Cancer Res 13:R113
Zhou H, Huang S (2010) mTOR signaling in cancer cell motility and tumor metastasis. Crit Rev Eukaryot Gene Expr 20:1–16
Brackett NO, Shariati M, Bernstam FM (2018) Role of PI3K/AKT/mTOR in cancer signaling. In: Badve S, Kumar G (eds) Predictive biomarkers in oncology. Springer, Cham, pp 263–270
Yao Y, Zhou D, Shi D et al (2019) GLI1 overexpression promotes gastric cancer cell proliferation and migration and induces drug resistance by combining with the AKT-mTOR pathway. Biomed Pharmacother 111:993–1004
Memmott RM, Dennis PA (2010) The role of the Akt/mTOR pathway in tobacco carcinogen-induced lung tumorigenesis. Clin Cancer Res 16(1):4–10
Yuge K, Kikuchi E, Hagiwara M et al (2015) Nicotine induces tumor growth and chemoresistance through activation of the PI3K/Akt/mTOR pathway in bladder cancer. Mol Cancer Ther 14(9):2112–2120
West KA, Brognard J, Clark AS et al (2003) Rapid Akt activation by nicotine and a tobacco carcinogen modulates the phenotype of normal human airway epithelial cells. J Clin Invest 111:81–90
Saxton RA, Sabatini DM (2017) mTOR signaling in growth, metabolism, and disease. Cell 168:960–976
Sengupta S, Peterson TR, Sabatini DM (2010) Regulation of the mTOR complex 1 pathway by nutrients, growth factors, and stress. Mol Cell 40:310–322
Chen M, Gu J, Delclos GL et al (2010) Genetic variations of the PI3K-AKT-mTOR pathway and clinical outcome in muscle invasive and metastatic bladder cancer patients. Carcinogenesis 31:13871391
Sahin F, Kannangai R, Adegbola O et al (2004) mTOR and P70 S6 kinase expression in primary liver neoplasms. Clin Cancer Res 10:8421–8425
Matsuoka T, Yashiro M (2014) The role of PI3K/Akt/mTOR. Signal Gastric Carcinoma Cancers 6:1441–1463
Riquelme I, Tapia O, Espinoza JA et al (2016) The gene expression status of the PI3K/AKT/mTOR pathway in gastric cancer tissues and cell lines. Pathol Oncol Res 22(4):797–805
Chevallier M, Guerret S, Chosseqros P et al (1994) A histological semiquantitative scoring system for evaluation of hepatic fibrosis in needle liver biopsy specimens: comparison with morphometric studies. Hepatology 20(2):349–355
Smyth EC, Capanu M, Janjigian YY et al (2012) Tobacco use is associated with increased recurrence and death from gastric cancer. Ann Surg Oncol 19:2088–2094
Hoffman D, Hoffman I (1998) Chemistry and toxicology. In: Burns D, Cummings KM, Hoffman D (eds) Cigars: health effects and trends, smoking and tobacco control monograph no. 9. National Cancer Institute, Bethesda, NIH 98-4302
Hecht SS, Hoffmann D (1988) Tobacco-specific nitrosamines, an important group of carcinogens in tobacco and tobacco smoke. Carcinogenesis 9(6):875–884
Hecht SS, Chen CHB, Hirota N et al (1978) Tobacco-specific nitrosamines: formation from nicotine in vitro and during Tobacco curing and carcinogenicity in strain a mice. J Natl Cancer Inst 60:819–824
Wu W, Song S, Ashley DL, Watson CH (2004) Assessment of tobaccospecific nitrosamines in the tobacco and mainstream smoke of Bidi cigarettes. Carcinogenesis 25:283–287
Shin VY, Cho CH (2005) Nicotine and gastric cancer. Alcohol 35:259–269
Dyke GW, Craven JL, Hall R, Garner RC (1992) Smoking-related DNA adducts in human gastric cancers. Int J Cancer 52:847-850 22
Kneller RW, You WC, Chang YS et al Liu (1992) Cigarette smoking and other risk factors for progression of precancerous stomach lesions. J Natl Cancer Inst 84:1261–1266 23
Vignot S, Faivre S, Aguirre D, Raymond E (2005) mTOR-targeted therapy of cancer with rapamycin derivatives. Ann Oncol 16:525–537
Mahalingam D, Sankhala K, Mita A et al (2009) Targeting the mTOR pathway using deforolimus in cancer therapy. Future Oncol 5:291–303
Tapia O, Riquelme I, Leal P et al (2014) The PI3K/AKT/mTOR pathway is activated in gastric cancer with potential prognosis and predictive significance. Virchows Arch 465(1):25–33
Asaka M, Takeda H, Sugiyama T, Kato M (1997) What role does Helicobacter pylori play in gastric cancer? Gastroenterology 113:56–60
Siman JH, Forsgren A, Berglund G, Floren CH (2001) Tobacco smoking increases the risk for gastric adenocarcinoma among Helicobacter pylori-infected individuals. Scand J Gastroenterol 36:208–13
Acknowledgements
This study was supported in part by the Chittaranjan National Cancer Institute, Kolkata, India and by University Grants commission of India. Nabendu Murmu sincerely thanks to Dr. Jayanta Chakrabarti, Director, Chittaranjan National Cancer Institute (CNCI), Kolkata, India for his active support.
Funding
This project is funded by University Grant Commission (UGC) Ref. No.: 17 − 06/2012(i)EU-V.
Author information
Authors and Affiliations
Contributions
PG and NM are responsible for the conception or design of the work. NA helped in providing the tissue samples of gastric cancer patients required for the project. PG and SM performed the acquisition, analysis, or interpretation of data of the work. SMM helped in reviewing the histopathology data of this project and provided the data. All authors finally approved the manuscript version to be published. Nabendu Murmu is the guarantor of the article. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interests
The authors of the manuscript do not have any conflicts of interest.
Ethics approval
The study was approved by the Institutional Ethics Committee (IEC) of Chittaranjan National Cancer Institute, Kolkata, India (IEC Ref: A-4.311/1/2018-2).
Informed consent
Consent was obtained from all patients included within the study. All the procedures of the present study were carried out in accordance with the Helsinki declaration and its later amendments.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ghosh, P., Alam, N., Mandal, S. et al. Association of mTOR pathway with risk of gastric cancer in male smoker with potential prognostic significance. Mol Biol Rep 47, 7489–7495 (2020). https://doi.org/10.1007/s11033-020-05808-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-020-05808-6