Abstract
Background
Targeted anti-IL-1β therapy may be a valuable option for the management of gouty arthritis. The present meta-analysis has evaluated the effect of canakinumab, an anti-IL-1β monoclonal antibody in gouty arthritis.
Methods
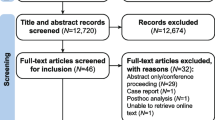
A standard meta-analysis protocol was developed and after performing a comprehensive literature search in MEDLINE, Cochrane, and International Clinical Trial Registry Platform (ICTRP), reviewers assessed eligibility and extracted data from three relevant articles. A random-effects model was used to estimate the pooled effect size as the mean difference in Visual Analouge Scale (VAS) score, serum hsCRP, serum Amyloid A, and risk ratio for global assessment between the groups. Quality assessment was done using the risk of bias assessment tool and summary of findings was prepared using standard Cochrane methodology with GradePro GDT.
Results
Treatment with canakinumab showed a mean reduction of VAS score by 14.59 mm [95% CI − 19.42 to − 9.77], serum hsCRP by 15.36 mg/L [95% CI 1.62–29.11], serum Amyloid A by 67.18 mg/L [95% CI 17.06–117.31], and improvement in patient global assessment (RR = 1.478; 95% CI 1.29–1.67) and physician global assessment (RR = 1.44; 95% CI 1.28–1.61). The probability that future studies may have a mean difference in VAS score less than zero has been calculated to be 27.3% using a cumulative distribution function (CDF) calculator.
Conclusion
This meta-analysis shows the beneficial effect of canakinumab over triamcinolone by reducing VAS score, serum hsCRP, serum amyloid A, and improvement in global assessments in acute gouty arthritis.





Similar content being viewed by others
References
Alten R, Gram H, Joosten LA, van den Berg WB, Sieper J, Wassenberg S et al (2008) The human anti-IL-1 beta monoclonal antibody ACZ885 is effective in joint inflammation models in mice and in a proof-of-concept study in patients with rheumatoid arthritis. Arthritis Res Ther 10:R67
Arromdee E, Michet CJ, Crowson CS, O'Fallon WM, Gabriel SE (2002) Epidemiology of gout: is the incidence rising? J Rheumatol 29:2403–2406
Balduzzi S, Rucker G, Schwarzer G (2019) How to perform a meta-analysis with R: a practical tutorial. Evid Based Ment Health 22:153–160
Berger P, McConnell JP, Nunn M, Kornman KS, Sorrell J, Stephenson K et al (2002) C-reactive protein levels are influenced by common IL-1 gene variations. Cytokine 17:171–174
Busso N, So A (2010) Mechanisms of inflammation in gout. Arthritis Res Ther 12:206
Chakraborty A, Van LM, Skerjanec A, Floch D, Klein UR, Krammer G, Sunkara G, Howard D (2013) Pharmacokinetic and pharmacodynamic properties of canakinumab in patients with gouty arthritis. J Clin Pharmacol 53(12):1240–1251
Challa DNV, Crowson CS, Davis JM 3rd (2017) The patient global assessment of disease activity in rheumatoid arthritis: identification of underlying latent factors. Rheumatol Ther 4:201–208
Clarson LE, Chandratre P, Hider SL, Belcher J, Heneghan C, Roddy E et al (2015) Increased cardiovascular mortality associated with gout: a systematic review and meta-analysis. Eur J Prev Cardiol 22:335–343
Cumulative Distribution Function (CDF) (2020) Calculator for the t-Distribution [Software]. [Online]. https://www.danielsoper.com/statcalc
GRADEpro GDT (2015) GRADEpro Guideline Development Tool [Software]. [Online]. https://gradepro.org
Harrold LR, Andrade SE, Briesacher BA, Raebel MA, Fouayzi H, Yood RA et al (2009) Adherence with urate-lowering therapies for the treatment of gout. Arthritis Res Ther 11:R46
Hirsch JD, Gnanasakthy A, Lale R, Choi K, Sarkin AJ (2014) Efficacy of Canakinumab vs. triamcinolone acetonide according to multiple gouty arthritis-related health outcomes measures. Int J Clini Pract 68 (12):1503–1507
Jordan KM, Cameron JS, Snaith M, Zhang W, Doherty M, Seckl J et al (2007) British Society for Rheumatology and British Health Professionals in Rheumatology guideline for the management of gout. Rheumatology (Oxford) 46:1372–1374
Khanna D, Khanna PP, Fitzgerald JD, Singh MK, Bae S, Neogi T et al (2012) 2012 American College of Rheumatology guidelines for management of gout. Part 2: therapy and antiinflammatory prophylaxis of acute gouty arthritis. Arthritis Care Res (Hoboken) 64:1447–1461
Khanna PP, Gladue HS, Singh MK, FitzGerald JD, Bae S, Prakash S et al (2014) Treatment of acute gout: a systematic review. Semin Arthritis Rheum 44:31–38
Kleinman NL, Brook RA, Patel PA, Melkonian AK, Brizee TJ, Smeeding JE et al (2007) The impact of gout on work absence and productivity. Value Health 10:231–237
Kuo CF, See LC, Luo SF, Ko YS, Lin YS, Hwang JS et al (2010) Gout: an independent risk factor for all-cause and cardiovascular mortality. Rheumatology (Oxford) 49:141–146
Martinon F (2010) Mechanisms of uric acid crystal-mediated autoinflammation. Immunol Rev 233:218–232
Martinon F, Petrilli V, Mayor A, Tardivel A, Tschopp J (2006) Gout-associated uric acid crystals activate the NALP3 inflammasome. Nature 440:237–241
Moher D, Liberati A, Tetzlaff J, Altman DG (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med 6:e1000097
Moher D, Shamseer L, Clarke M, Ghersi D, Liberati A, Petticrew M, Shekelle P et al (2015) Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst Rev 4:1
Moriya M, Slade N, Brdar B, Medverec Z, Tomic K, Jelakovic B et al (2011) TP53 mutational signature for aristolochic acid: an environmental carcinogen. Int J Cancer 129:1532–1536
Neogi T (2011) Clinical practice. Gout N Engl J Med 364:443–452
Pascoe VL, Enamandram M, Corey KC, Cheng CE, Javorsky EJ, Sung SM et al (2015) Using the Physician Global Assessment in a clinical setting to measure and track patient outcomes. JAMA Dermatol 151:375–381
Riedel AA, Nelson M, Wallace K, Joseph-Ridge N, Cleary M, Fam AG (2004) Prevalence of comorbid conditions and prescription medication use among patients with gout and hyperuricemia in a managed care setting. J Clin Rheumatol 10:308–314
Schlesinger N, Thiele RG (2010) The pathogenesis of bone erosions in gouty arthritis. Ann Rheum Dis 69:1907–1912
Schlesinger N, De Meulemeester M, Pikhlak A, Yucel AE, Richard D, Murphy V et al (2011a) Canakinumab relieves symptoms of acute flares and improves health-related quality of life in patients with difficult-to-treat Gouty Arthritis by suppressing inflammation: results of a randomized, dose-ranging study. Arthritis Res Ther 13:R53
Schlesinger N, Mysler E, Lin HY, De Meulemeester M, Rovensky J, Arulmani U et al (2011b) Canakinumab reduces the risk of acute gouty arthritis flares during initiation of allopurinol treatment: results of a double-blind, randomised study. Ann Rheum Dis 70:1264–1271
Schlesinger N, Alten RE, Bardin T, Schumacher HR, Bloch M, Gimona A et al (2012) Canakinumab for acute gouty arthritis in patients with limited treatment options: results from two randomised, multicentre, active-controlled, double-blind trials and their initial extensions. Ann Rheum Dis 71:1839–1848
Schwarzer G (2007) meta: an R Package for meta-analysis, vol 7
Scire CA, Manara M, Cimmino MA, Govoni M, Salaffi F, Punzi L et al (2013) Gout impacts on function and health-related quality of life beyond associated risk factors and medical conditions: results from the KING observational study of the Italian Society for Rheumatology (SIR). Arthritis Res Ther 15:R101
So A (2008) Developments in the scientific and clinical understanding of gout. Arthritis Res Ther 10:221
So A, De Meulemeester M, Pikhlak A, Yucel AE, Richard D, Murphy V et al (2010) Canakinumab for the treatment of acute flares in difficult-to-treat gouty arthritis: results of a multicenter, phase II, dose-ranging study. Arthritis Rheum 62:3064–3076
Suurmond R, van Rhee H, Hak T (2017) Introduction, comparison, and validation of meta-essentials: a free and simple tool for meta-analysis. Res Synth Methods 8:537–553
Trifiro G, Morabito P, Cavagna L, Ferrajolo C, Pecchioli S, Simonetti M et al (2013) Epidemiology of gout and hyperuricaemia in Italy during the years 2005–2009: a nationwide population-based study. Ann Rheum Dis 72:694–700
Zhang W, Doherty M, Bardin T, Pascual E, Barskova V, Conaghan P et al (2006) EULAR evidence based recommendations for gout. Part II: management. Report of a task force of the EULAR Standing Committee for International Clinical Studies Including Therapeutics (ESCISIT). Ann Rheum Dis 65:1312–1324
Author information
Authors and Affiliations
Contributions
MJ concept and design, literature search, study selection and data collection, data extraction and management, data analysis and interpretation of data, critical revision of the manuscript for important intellectual content, and final approval of the manuscript. AT literature search, study selection and data collection, data extraction and management, and final approval of the manuscript. AM concept and design, data analysis and interpretation, drafting of the manuscript, and final approval of the manuscript. RM literature search, study selection and data collection, data extraction, critical revision of the manuscript for important intellectual content, and final approval of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
None to declare.
Consent for publication
Authors are responsible for the correctness of the statements provided in the manuscript.
Availability of data and material
All data generated or analysed during this study are included in this published article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Jena, M., Tripathy, A., Mishra, A. et al. Effect of canakinumab on clinical and biochemical parameters in acute gouty arthritis: a meta-analysis. Inflammopharmacol 29, 35–47 (2021). https://doi.org/10.1007/s10787-020-00753-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10787-020-00753-z