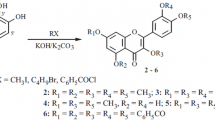
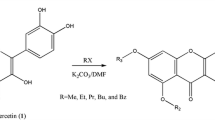
The tetra-O-methyl ether of quercetin (QU) 3 (54%), 3,7,4′-tri-O-methyl ether 4 (30%), and a previously unreported 3,7,3-tri-O-methyl ether of QU 5 (7%) were obtained via methylation of QU by an excess of diazomethane in dioxane. Their structures were established using 2D NMR (1H–1H COSY, 1H–1H NOESY, 1H–13C HSQC, 1H–13C HMBC). Tetra-O-methyl ether of QU 3 exhibited pronounced hypoglycemic activity, reduced alloxan-induced hyperglycemia in rats by 44.5% compared to a control, and was 2.7 times more active than QU.


Similar content being viewed by others
References
D. Yu. Korulkin, Zh. A. Abilov, R. A. Muzychkina, and G. A. Tolstikov, Natural Flavonoids [in Russian], Geo, Novosibirsk, 2007, 232 pp.
K. Murota and J. Terao, Arch. Biochem. Biophys., 417, 12 (2003).
Y. Cui, Y. Han, X. Yang, Y. Sun, and Y. Zhao, Molecules, 19, 291 (2014).
T. Taguri, T. Tanaka, and I. Kouno, Biol. Pharm. Bull., 29, 2226 (2006).
D. Chattopadhyay and T. N. Naik, Mini-Rev. Med. Chem., 7, 275 (2007).
N. Uchide and H. Toyoda, Anti-Infect. Agents Med. Chem., 7, 73 (2008).
D. K. Suh, E. J. Lee, and J. H. Kim, Arch. Pharm. Res., 33, 781 (2010).
H. Zhang, M. Zhang, L. Yu, Y. Zhao, N. He, and X. Yag, Food Chem. Toxicol., 50, 1589 (2012).
U. Undeger, S. Aydin, A. Basaran, and N. Basaran, Toxicol. Lett., 151, 143 (2004).
A. V. Kurkina, Flavonoids of Pharmacopoeial Plants [in Russian], Ofort, SamGMU, Samara, 2012, 290 pp.
C. Carpene, S. Gomez-Zorita, S. Deleruelle, and M. A. Carpene, Curr. Med. Chem., 22, 150 (2015).
Yu. S. Tarakhovskii, Yu. A. Kim, B. C. Abdrasilov, and E. N. Muzafarov, Flavonoids: Biochemistry, Biophysics, Medicine [in Russian], Synchrobook, Pushchino, 2013, 310 pp.
X. Cai, Z. Fang, J. Dou, A. Yu, and G. Zhai, Curr. Med. Chem., 20, 2572 (2013).
L. Biasutto, E. Marotta, U. De Marchi, M. Zoratti, and C. Paradisi, J. Med. Chem., 50, 241 (2007).
L. Chen, T. He, Y. Han, J.-Z. Sheng, S. Jin, and M.-W. Jin, Molecules, 16, 5754 (2011).
A. Mattarei, L. Biasutto, F. Rastrelli, S. Garbisa, E. Marotta, M. Zoratti, and C. Paradisi, Molecules, 15, 4722 (2010).
M. Bouktaib, S. Lebrun, A. Atmani, and C. Rolando, Tetrahedron, 58, 10001 (2002).
L. Jurd, J. Org. Chem., 27, 1294 (1962).
E. R. Karimova, L. A. Baltina, L. V. Spirikhin, R. M. Kondratenko, R. R. Farkhutdinov, and I. V. Petrova, Chem. Nat. Compd., 51, 851 (2015).
D. A. Sainiev, D. V. Mavrodiev, M. F. Abdullin, E. R. Karimova, S. V. Pikhtovnikov, L. A. Baltina, V. K. Mavrodiev, and I. I. Furlei, High Energy Chem., 49, 129 (2015).
A. N. Mironov (ed.), Handbook for Preclinical Drug Trials [in Russian], Part 1, Grif i K, Moscow, 2012, 670 pp.
N. A. Pulina, F. V. Sobin, A. I. Krasnova, T. A. Yushkova, V. V. Yushkov, P. A. Mokin, K. V. Yatsenko, and E. B. Babushkina, Khim.-farm. Zh., 45 (5), 18 (2011).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiya Prirodnykh Soedinenii, No. 5, September–October, 2020, pp. 718–721.
Rights and permissions
About this article
Cite this article
Karimova, E.R., Baltina, L.A., Spirikhin, L.V. et al. Methylation of Quercetin by Diazomethane and Hypoglycemic Activity of its Tetra-O-Methyl Ether. Chem Nat Compd 56, 837–841 (2020). https://doi.org/10.1007/s10600-020-03164-2
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10600-020-03164-2