Abstract
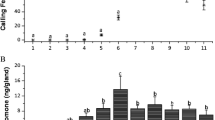
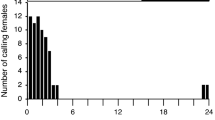
The complex agonistic repertoire between male lobster cockroaches (Nauphoeta cinerea) makes this species an excellent model for aggression studies. During the establishment of dominance hierarchies, 3-hydroxy-2-butanone (3H-2B) functions as a suppression pheromone, keeping the rivals in a submissive state. In the present study, we evaluated the release of 3H-2B by dominant individuals across four different time phases within the 24-h photoperiod, i.e., early scotophase (ES), late scotophase (LS), early photophase (EP), and late photophase (LP). For each time phase, we collected volatile pheromones during a 60-min first-encounter fight to measure the level of released 3H-2B. Subsequently, the amount of 3H-2B remaining in the sternal glands of dominant and subordinate individuals was measured and compared to socially naïve male controls. Release of 3H-2B was relatively high during ES or LP first-encounter fights, compared to LS or EP encounters. The attack duration and aggressive posture intensity in dominant males were positively correlated with the amount of 3H-2B release in all four phases. A similar statistical distribution was found between the amount of 3H-2B released by dominant males and the amount of 3H-2B in the sternal glands of naïve male sternal during LS, EP, and LP. However, during ES, the statistical distribution of 3H-2B released by the dominant was significantly greater than the distribution of 3H-2B content in socially naïve male sternal glands. The observed phase-dependence of 3H-2B release might be due to variations in 3H-2B biosynthesis or the scotophase-specific behavior of naïve males, wherein an aggressive posture is spontaneously adopted with concomitant 3H-2B release.




Similar content being viewed by others
References
Bell WJ, Gorton RE (1978) Informational analysis of agonistic behavior and dominance hierarchy formation in cockroach, Nauphoeta cinerea. Behaviour 67:217–235
Chen SC, Yang RL, Ho HY, Chou SY, Kou R (2007) Strategic 3-hydroxy-2-butanone release in the dominant male lobster cockroach, Nauphoeta cinerea. Naturwissenschaften 94:927–933
Chou SY, Huang ZY, Chen SC, Yang RL, Kou R (2007) Antenna contact and agonism in the male lobster cockroach, Nauphoeta cinerea. Horm Behav 52:252–260
Everaerts C, Fenaux-Benderitter F, Farine JP, Brossut R (1997) Male dominant/ subordinate relationships cuticular profiles and sex pheromone in Nauphoeta cinerea (Dictyoptera, Blaberidae). Insect Soc 44:277–287
Ewing LS (1967) Fighting and death from stress in a cockroach. Science 155:1035–1036
Kou R, Hsu CC (2013) Mating enhanced the probability of winning in lobster cockroaches. Horm Behav 64:546–556
Kou R, Chen SC, Chen YR, Ho HY (2006) 3-hydroxy-2-butanone and the first-encounter fight in the male lobster cockroach, Nauphoeta cinerea. Naturwissenschaften 93:286–291
Kou R, Chang HW, Huang ZY, Yang RL (2008a) Pheromone, juvenile hormone, and social status in the male lobster cockroach Nauphoeta cinerea. Arch Insect Biochem Physiol 68:144–155
Kou R, Chou SY, Huang ZY, Yang RL (2008b) Juvenile hormone levels are increased in winners of cockroach fights. Horm Behav 54:521–527
Kou R, Chang HW, Chen SC, Ho HY (2009) Suppression pheromone and cockroach rank formation. Naturwissenschaften 96:691–701
Kramer S (1964) Aggressive behavior in the courtship of cockroach Nauphoeta cinerea. Am Zool 4:164
Manning A, Johnstone G (1970) The effects of early adult experience on the development of aggressiveness in males of the cockroach Nauphoeta cinerea. Rev Comp Anim 4:12–16
Moore AJ (1997) The evolution of social signals: morphological, functional, and genetic integration of the sex pheromone in Nauphoeta cinerea. Evolution 51:1920–1928
Moore AJ, Cicconne WJ, Breed MD (1988) The influence of social experience on the behavior of males in the cockroach Nauphoeta cinerea. J Insect Behav 1:157–168
Moore AJ, Reagan NL, Haynes KF (1995) Conditional signalling strategies: effects of ontogeny, social experience and social status on the pheromonal signal of male cockroaches. Anim Behav 50:191–202
Moore PJ, Reagan-Wallin NL, Haynes KF, Moore AJ (1997) Odour conveys status on the cockroaches. Nature 389:25
Schal C, Bell WJ (1983) Determinants of dominant-subordinate interactions in males of the cockroach Nauphoeta cinerea. Biol Behav 8:117–139
Sirugue D, Bonnard O, Quere JL, Farine JP, Brossut R (1992) 2-methylthiazolidine and 4-ethyl-2-methoxyphenol, male sex pheromone components of the cockroach Nauphoeta cinerea (Dictyoptera, Blaberidae): a reinvestigation. J Chem Ecol 18:2261–2276
Smith SK, Breed MD (1982) Olfactory cues in discrimination among individuals in dominance hierarchies in the cockroach, Nauphoeta cinerea. Physiol Entomol 7:337–341
Sréng L (1990) Seducing, male sex pheromone of the cockroach Nauphoeta cinerea : isolation, identification, and bioassay. J Chem Ecol 16:2899–2912
Acknowledgments
We warmly thank the Data Science Statistical Cooperation Center of Academia Sinica (ASCFII-108-117) for statistical analysis support.
Funding
This work was financially supported by the National Science Council (NSC92- 2313-B-001-010) and Academia Sinica, Taipei, Taiwan, ROC.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethical approval
Nauphoeta cinerea were used for experiments in this article. All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. This article does not contain any studies with human participants performed by any of the authors.
Additional information
Communicated by: Matthias Waltert
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kou, R., Chen, SC., Yang, RL. et al. Photoperiod-dependent release of suppression pheromone in the male lobster cockroach Nauphoeta cinerea. Sci Nat 106, 56 (2019). https://doi.org/10.1007/s00114-019-1654-5
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00114-019-1654-5