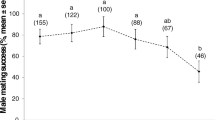
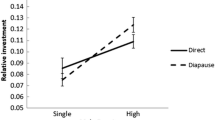
Abstract
Frequent mating can deplete sperm and seminal fluid, limiting male ability to sire offspring. Frequent mating could also deplete non-genetic ejaculate components that affect offspring quality. These effects of frequent mating on male reproductive success may be mediated by male condition, or by modification of subsequent male mating behaviour. Using the neriid fly Telostylinus angusticollis, we conducted two experiments to examine whether a history of frequent mating affects males’ subsequent mating rate and offspring traits. The first experiment tested whether male condition (manipulated by varying larval diet quality) and mating history affects male performance in a subsequent mating with a single novel female whereby we predicted effects of previous mating may be more prevalent in low-condition males. Prior mating resulted in a reduction in mating rate with the novel female, but we did not detect an effect of mating history or male condition on offspring quality or quantity. The second experiment tested whether costs of mating become more evident when males encounter multiple novel females. Surprisingly, while prior mating once again resulted in a reduction in mating rate with the novel females, we still did not detect an effect of condition or mating history on any offspring traits. Therefore, male neriid flies appear to be able to mate many times without suffering a reduction in offspring quality or quantity. The apparent lack of an effect of frequent mating on such traits could be mediated by a reduction in mating rate, reflecting male prudence with ejaculate expenditure.



Similar content being viewed by others
References
Adler MI, Bonduriansky R (2013) Paternal effects on offspring fitness reflect father’s social environment. Evol Biol 40:288–292. https://doi.org/10.1007/s11692-012-9211-6
Bates D, Maechler M, Bolker B, Walker S (2015) lme4: linear mixed-effects models using Eigen and S4. J Stat Softw 67:1–48
Bath E, Tatarnic N, Bonduriansky R (2012) Asymmetric reproductive isolation and interference in neriid flies: the roles of genital morphology and behaviour. Anim Behav 84:1331–1339
Bonduriansky R (2007) The evolution of condition-dependent sexual dimorphism. Am Nat 169:9–19. https://doi.org/10.1086/510214
Bonduriansky R, Crean AJ (2017) What are parental condition-transfer effects and how can they be detected? Methods Ecol Evol. https://doi.org/10.1111/2041-210X.12848
Bonduriansky R, Head M (2007) Maternal and paternal condition effects on offspring phenotype in Telostylinus angusticollis (Diptera: Neriidae). J Evol Biol 20:2379–2388. https://doi.org/10.1111/j.1420-9101.2007.01409.x
Chapman T (2001) Seminal fluid-mediated fitness traits in Drosophila. Heredity (Edinb) 87:511–521. https://doi.org/10.1046/j.1365-2540.2001.00961.x
Clutton-Brock TH (1991) The evolution of parental care. Princeton University Press, Princeton
Cordts R, Partridge L (1996) Courtship reduces longevity of male Drosophila melanogaster. Anim Behav 52:269–278
Crean AJ, Adler MI, Bonduriansky R (2016) Seminal fluid and mate choice: new predictions. Trends Ecol Evol 31:253–255. https://doi.org/10.1016/j.tree.2016.02.004
Crean AJ, Kopps AM, Bonduriansky R (2014) Revisiting telegony: offspring inherit an acquired characteristic of their mother’s previous mate. Ecol Lett 17:1545–1552. https://doi.org/10.1111/ele.12373
Delcurto H, Wu G, Satterfield MC (2013) Nutrition and reproduction: links to epigenetics and metabolic syndrome in offspring. Curr Opin Clin Nutr Metab Care 16:385–391. https://doi.org/10.1097/MCO.0b013e328361f96d
Dewsbury DA (1982) Ejaculate cost and male choice. Am Nat 119:601–610. https://doi.org/10.1086/283938
Dewsbury DAA (1981) Effects of novelty on copulatory behavior: the Coolidge effect and related phenomena. Psychol Bull 89:464–482. https://doi.org/10.1037/0033-2909.89.3.464
Douglas T, Anderson R, Saltz JB (2020) Limits to male reproductive potential across mating bouts in Drosophila melanogaster. Anim Behav 160:25–33. https://doi.org/10.1016/j.anbehav.2019.11.009
Droney DC (1998) The influence of the nutritional content of the adult male diet on testis mass, body condition and courtship vigour in a Hawaiian Drosophila. Funct Ecol 12:920–928. https://doi.org/10.1046/j.1365-2435.1998.00266.x
Evans JP, Lymbery RA, Wiid KS et al (2017) Sperm as moderators of environmentally induced paternal effects in a livebearing fish. Biol Lett 13:10–13. https://doi.org/10.1098/rsbl.2017.0087
Fricke C, Adler MI, Brooks RC, Bonduriansky R (2015) The complexity of male reproductive success: effects of nutrition, morphology, and experience. Behav Ecol 26:617–624. https://doi.org/10.1093/beheco/aru240
Hihara F (1981) Effects of the male accessory gland secretion on oviposition and remating in females of Drosophila melanogaster. Zool Mag 90:307–316
Hopkins BR, Sepil I, Thézénas M-L et al (2019) Divergent allocation of sperm and the seminal proteome along a competition gradient in Drosophila melanogaster. Proc Natl Acad Sci 116:17925–17933. https://doi.org/10.1073/pnas.1906149116
Kawasaki N, Brassil CE, Brooks RC, Bonduriansky R (2008) Environmental effects on the expression of life span and aging: an extreme contrast between wild and captive cohorts of Telostylinus angusticollis (Diptera: Neriidae). Am Nat 172:346–357. https://doi.org/10.1086/589519
Koppik M, Ruhmann H, Fricke C (2018) The effect of mating history on male reproductive ageing in Drosophila melanogaster. J Insect Physiol 111:16–24. https://doi.org/10.1016/j.jinsphys.2018.10.003
Kotiaho JS, Simmons LW (2003) Longevity cost of reproduction for males but no longevity cost of mating or courtship for females in the male-dimorphic dung beetle Onthophagus binodis. J Insect Physiol 49:817–822. https://doi.org/10.1016/S0022-1910(03)00117-3
Kuznetsova A, Brockhoff P, Rune H (2017) lmerTest package: tests in linear mixed effects models. J Stat Softw 82:1–26
Linklater JR, Wertheim B, Wigby S, Chapman T (2007) Ejaculate depletion patterns evolve in response to experimental manipulation of sex ratio in Drosophila melanogaster. Evolution (N Y) 61:2027–2034. https://doi.org/10.1111/j.1558-5646.2007.00157.x
Macartney EL, Crean AJ, Bonduriansky R (2018a) Epigenetic paternal effects as costly, condition-dependent traits. Heredity (Edinb) 121:248–256. https://doi.org/10.1038/s41437-018-0096-8
Macartney EL, Crean AJ, Bonduriansky R (2017) Adult dietary protein has age- and context-dependent effects on male post-copulatory performance. J Evol Biol 38:42–49. https://doi.org/10.1111/jeb.13087
Macartney EL, Crean AJ, Nakagawa S, Bonduriansky R (2019) Effects of nutrient limitation on sperm and seminal fluid: a systematic review and meta-analysis. Biol Rev 94:1722–1739. https://doi.org/10.1111/brv.12524
Macartney EL, Nicovich PR, Bonduriansky R, Crean AJ (2018b) Developmental diet irreversibly shapes male post-copulatory traits in the neriid fly Telostylinus angusticollis. J Evol Biol 31:1894–1902. https://doi.org/10.1111/jeb.13384
Marcotte M, Delisle J, McNeil JN (2007) Effects of different male remating intervals on the reproductive success of Choristoneura rosaceana males and females. J Insect Physiol 53:139–145. https://doi.org/10.1016/j.jinsphys.2006.11.005
Maynard Smith J (1977) Parental investment: a prospective analysis. Anim Behav 25:1–9. https://doi.org/10.1016/0003-3472(77)90062-8
McNamara KB, Elgar MA, Jones TM (2008) A longevity cost of re-mating but no benefits of polyandry in the almond moth, Cadra cautella. Behav Ecol Sociobiol 62:1433–1440. https://doi.org/10.1007/s00265-008-0573-9
Michaud JP, Bista M, Mishra G, Singh O (2013) Sexual activity diminishes male virility in two Coccinella species: consequences for female fertility and progeny development. Bull Entomol Res 103:570–577. https://doi.org/10.1017/s0007485313000199
Mirhosseini MA, Michaud JP, Jalali MA, Ziaaddini M (2014) Paternal effects correlate with female reproductive stimulation in the polyandrous ladybird Cheilomenes sexmaculata. Bull Entomol Res 104:480–485. https://doi.org/10.1017/s0007485314000194
Muller K, Arenas L, Thiéry D, Moreau J (2016) Direct benefits from choosing a virgin male in the European grapevine moth, Lobesia botrana. Anim Behav 114:165–172. https://doi.org/10.1016/j.anbehav.2016.02.005
Parker GA, Ball MA (2005) Sperm competition, mating rate and the evolution of testis and ejaculate sizes: a population model. Biol Lett 1:235–238. https://doi.org/10.1098/rsbl.2004.0273
Parker GA, Pizzari T (2010) Sperm competition and ejaculate economics. Biol Rev 85:897–934. https://doi.org/10.1111/j.1469-185X.2010.00140.x
Perry JC, Tse CT (2013) Extreme costs of mating for male two-spot ladybird beetles. PLoS ONE 8:1–5. https://doi.org/10.1371/journal.pone.0081934
Pitnick S, Markow TA (1994) Male gametic strategies - sperm size, testis size, and the allocation of ejaculate among successive mates by the sperm-limited fly Drosophila pachea and its relatives. Am Nat 143:785–819. https://doi.org/10.1086/285633
Pizzari T, Cornwallis CK, Hanne L et al (2003) Sophisticated sperm allocation in male fowl. Nature 426:70–74. https://doi.org/10.1038/nature02025.1
Polak M, Simmons LW, Benoit JB et al (2017) Nutritional geometry of paternal effects on embryo mortality. Proc R Soc B Biol Sci 284:1–9. https://doi.org/10.1098/rspb.2017.1492
Preston BT, Stevenson IR, Pemberton JM, Wilson K (2001) Dominant rams lose out by sperm depletion. Nature 409:681–682. https://doi.org/10.1038/35055617
Rasband WS (2015) ImageJ, U. S. National Institutes of Health, Bethesda, Maryland, USA. http://imagej.nih.gov/ij/
Reinhardt K, Naylor R, Siva-Jothy MT (2011) Male mating rate is constrained by seminal fluid availability in bedbugs, Cimex lectularius. PLoS ONE 6:1–8. https://doi.org/10.1371/journal.pone.0022082
Schaalje GB, McBride JB, Fellingham GW (2002) Adequacy of approximations to distributions of test statistics in complex mixed linear models. J Agric Biol Environ Stat 7:512–524. https://doi.org/10.1198/108571102726
Sentinella A, Crean A, Bonduriansky R (2013) Dietary protein mediates a trade-off between larval survival and the development of male secondary sexual traits. Funct Ecol 27:1134–1144. https://doi.org/10.1111/1365-2435.12104
Sheldon BC (2002) Relating paternity to paternal care. Philos Trans R Soc B Biol Sci 357:341–350. https://doi.org/10.1098/rstb.2001.0931
Torres-Vila LM, Jennions MD (2005) Male mating history and female fecundity in the Lepidoptera: do male virgins make better partners? Behav Ecol Sociobiol 57:318–326. https://doi.org/10.1007/s00265-004-0857-7
Vahed K (1998) The function of nuptial feeding in insects: review of empirical studies. Biol Rev Camb Philos Soc 73:43–78. https://doi.org/10.1017/s0006323197005112
Wigby S, Sirot LK, Linklater JR et al (2009) Seminal fluid protein allocation and male reproductive success. Curr Biol 19:751–757. https://doi.org/10.1016/j.cub.2009.03.036
Wylde Z, Crean A, Bonduriansky R (2020) Effects of condition and sperm competition risk on sperm allocation and storage in neriid flies. Behav Ecol 31:202–212. https://doi.org/10.1093/beheco/arz178
Acknowledgements
We would like to thank the Australian Research Council for a Discovery Grant awarded to RB (Grant Number DP170702449). We would also like to thank two anonymous reviewers and the Associate Editor for their thorough and insightful comments. The authors declare no conflicts of interest. No permission was needed to collect flies from Fred Hollows Reserve, Coogee, Sydney.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Macartney, E.L., Bonduriansky, R. & Crean, A.J. Frequent mating reduces male mating rate but not offspring quality or quantity in a neriid fly. Evol Ecol 34, 915–927 (2020). https://doi.org/10.1007/s10682-020-10076-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10682-020-10076-8