Abstract
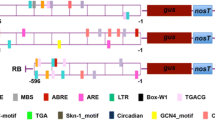
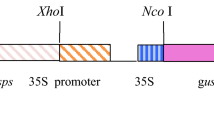
The present study was conducted to determine efficiency of green tissue-specific (pRCA) and stress-inducible promoters (pRD29A) to express E. coli beta-glucuronidase (gusA) gene in transgenic potatoes compared with constitutive promoter (35S CaMV). The promoter fragments were isolated from their original source and cloned upstream to gusA in pCAMBIA-1301 binary vector to develop plant expression constructs, i.e., pRCA-pCAMBIA and pRD29A-pCAMBIA. Agrobacterium strain GV2260 harboring recombinant plasmids were used to infect leaf discs and internodal explant of Lady Olympia cultivar. GUS histochemical analysis was performed at different stages to determine GUS activity in transgenic plants. To determine activity of stress-inducible promoter (pRD29A), transgenic plants were exposed to heat, drought and combination of both heat and drought stress. The real time (RT-qPCR) and GUS florimetric assays revealed that pRD29A promoter gets more activated under drought, heat and combination of both stresses. GUS expression levels were more than 10 folds high with pRD29A promoter compared to control. Likewise, the reduced transcripts levels of gusA gene under control of pRCA promoter were found in tuber/roots of transgenic plants compared to 35S promoter. GUS florimetric assays also showed decreased or no GUS expression in tubers. In conclusion, the results encourage the appropriate use of promoters to drive the expression of foreign gene(s) for the development of potato lines tolerant to biotic and abiotic stress while minimizing the risks of transgenic technology in potatoes.







Similar content being viewed by others
References
Ahmed HAA, Onarıcı S, Bakhsh A, Akdoğan G, Karakoç ÖC, Özcan SF, Aydın G, Aasim M, Ünlü L, Sancak C, Naimov S (2017) Targeted expression of insecticidal hybrid SN19 gene in potato leads to enhanced resistance against Colorado potato beetle (Leptinotarsa decemlineata Say) and tomato leafminer (Tuta absoluta Meyrick). Plant Biotechol Rep 11:315–329
Ahmed HA, Barpete S, Akdogan G, Aydin G, Sancak C, Ozcan S (2018) Efficient regeneration and Agrobacterium tumefaciens mediated genetic transformation of potato (Solanum tuberosum L.). Fresenius Environ Bull 27:3020–3027
Alisdair R, Fernie R, Willmitzer L (2001) Update on tuber formation, dormancy, and sprouting molecular and biochemical triggers of potato tuber development. Plant Physiol 127:1459–1465
Amarasinghe BHR, Nitschke EF, Wu Y, Udall JA, Dennis ES, Constable G, Llewellyn DJ (2006) Genomic approaches to the discovery of promoters for sustained expression in cotton (Gossypium hirsutum L.) under field conditions: expression analysis in transgenic cotton and Arabidopsis of a RuBisCo small subunit promoter identified using EST sequence analysis and cDNA microarrays. Plant Biotechnol 23:437–450
Anayol E, Bakhsh A, Karakoç ÖC, Onarıcı S, Köm D, Aasim M, Özcan SF, Barpete S, Khabbazi SD, Önol B, Sancak C (2016) Towards better insect management strategy: restriction of insecticidal gene expression to biting sites in transgenic cotton. Plant Biotechnol Rep 10:83–94
Analytical Software (2005) Statistix 8.1 for Windows. Analytical Software, Tallahassee, Florida
Bakhsh A (2020) Development of efficient, reproducible and stable Agrobacterium-mediated genetic transformation of five potato cultivars. Food Technol Biotechnol 58(1):57–63
Bakhsh A, Qayyum RA, Shamim Z, Husnain T (2011) A mini review: rubisco small subunit as a strong, green tissue-specific promoter. Arch of Biol Sci 63:299–307
Bakhsh A, Siddique S, Husnain T (2012) A molecular approach to combat spatio-temporal variation in insecticidal gene (Cry1Ac) expression in cotton. Euphytica 183:65–74
Bakhsh A, Anayol E, Ozcan SF (2014) Comparison of transformation efficiency of five Agrobacterium tumefaciens strains in Nicotiana Tabacum L. Emir J Food Agric 26:259–264
Bakhsh A, Anayol E, Khabbazi SD, Karakoç ÖC, Sancak C, Özcan S (2016) Development of insect-resistant cotton lines with targeted expression of insecticidal gene. Arch of Biol Sci 68:773–780
Bakhsh A, Hussain T, Rahamkulov I, Ufuk D, Çalışkan ME (2020) Transgenic potato lines expressing CP4-EPSP synthase exhibit resistance against glyphosate. Plant Cell Tiss Organ Cult 140:23–34
Beaujean A, Sangwan R, Lecardonnel A, Sangwan-Norreel B (1998) Agrobacterium-mediated transformation of three economically important potato cultivars using sliced internodal explants, an efficient protocol of transformation. J Exp Bot 49:1589–1595
Behnam B, Kikuchi A, Celebi-Toprak F, Kasuga M, Yamaguchi-Shinozaki K, Watanabe KN (2007) Arabidopsis rd29A:DREB1A enhances freezing tolerance in transgenic potato. Plant Cell Rep 26:1275–1282
Bottino PJ (2018) GUS Gene assay in transformed tissues. Available at https://www.goldbio.com/documents/1053/Gus%2520Gene%2520Assay%2520Protocol.pdf. Retried on March 2020
Çalışkan ME, Onaran H, Arıoğlu H (2010) Overview of the Turkish potato sector: challenges, achievements and expectations. Potato Res 53:255–266
Conner AJ, Glare TR, Nap JP (2003) The release of genetically modified crops into the environment. Part II. Overview of ecological risk assessment. Plant J 33:19–46
Dandekar AM, Fisk H (2005) Plant transformation: Agrobacterium-mediated gene transformation. In: Pena L (ed) Transgenic plants: methods and protocols. Humana Press, New Jersey, pp 35–46
Dönmez BA, Dangol SD, Bakhsh A (2019) Transformation efficiency of five Agrobacterium strains in diploid and tetraploid potatoes. Sarhad J Agric 35:1344–1350
Gopal J, Iwama K (2007) In vitro screening of potato against water-stress mediated through sorbitol and polyethylene glycol. Plant Cell Rep 26:693–700
Hameed A, Tahir MN, Asad S, Bilal R, Van Eck J, Jander G, Mansoor S (2017) RNAi-mediated simultaneous resistance against three RNA viruses in potato. Molecular Biotechnol 59:73–83
Heidari Japelaghi R, Haddad R, Valizadeh M, Dorani Uliaie E, Jalali Javaran M (2018) High-efficiency Agrobacterium-mediated transformation of tobacco (Nicotiana tabacum). J Plant Mol Breed 6:38–50
Ho MW, Ryan A, Cummins J (1999) Cauliflower mosaic viral promoter-a recipe for disaster? Microb Ecol Health Dis 11:194–197
Hussain T, Aksoy E, Çalışkan ME, Bakhsh A (2019) Transgenic potato lines expressing hairpin RNAi construct of molting-associated EcR gene exhibit enhanced resistance against Colorado potato beetle (Leptinotarsa decemlineata, Say). Transgenic Res 28:151–164
Jefferson RA, Kavanagh TA, Bevan MW (1987) GUS fusions: beta-glucuronidase as a sensitive and versatile gene fusion marker in higher plants. EMBO J 6:3901–3907
Khabbazi SD, Khabbazi AD, Özcan SF, Bakhsh A, Başalma D, Özcan S (2018) Expression of GNA and biting site-restricted cry1Ac in cotton; an efficient attribution to insect pest management strategies. Plant Biotechnol Rep 12:273–282
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real time quantitative PCR and the 2∆∆C(T) method. Methods 25:402–408
Mithra SVA, Kulkarni K, Srinivasan R (2017) Plant promoters: characterization and applications in transgenic technology. In: Abdin M, Kiran U, Kamaluddin Ali A (eds) Plant biotechnology: principles and applications. Springer, Singapore
Msanne J, Lin J, Stone JM, Awada T (2011) Characterization of abiotic stress-responsive Arabidopsis thaliana RD29A and RD29B genes and evaluation of transgenes. Planta 234:97–107
Murashige T, Skoog F (1962) Revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497
Nicot N, Hausman JF, Hoffman L, Evers D (2005) Housekeeping gene selection for real time PCR normalization in potato during biotic and abiotic stress. J Exp Bot 56:2907–2914
Özcan S, Firek S, Draper J (1993) Selectable marker genes engineered for specific expression in target cells for plant transformation. Nat Biotechnol 11:218–221
Pinhero R, Pazhekattu R, Marangoni AG, Liu Q, Yada RY (2011) Alleviation of low temperature sweetening in potato by expressing Arabidopsis pyruvate decarboxylase gene and stress-inducible rd29A: a preliminary study. Physiol Mol Biol Plants 17:105–114
Pino MT, Ávila A, Molina A, Jeknic Z, Chen TH (2013) Enhanced in vitro drought tolerance of Solanum tuberosum and Solanum commersonii plants overexpressing the ScCBF1 gene. Cien. Inv. Agr. 40 (1): 171–184. Int J Agric Nat Resour 40:171–184
Podevin N, Du Jardin P (2012) Possible consequences of the overlap between the CaMV 35S promoter regions in plant transformation vectors used and the viral gene VI in transgenic plants. GM crops food 3:296–300
Porto MS, Pinheiro MPN, Batista VGL, dos Santos RC, de Albuquerque Melo Filho P, de Lima LM (2014) Plant promoters: an approach of structure and function. Mol Biotechnol 56:38–49
Potenza C, Aleman L, Sengupta-Gopalan C (2004) Targeting transgene expression in research, agricultural, and environmental applications: promoters used in plant transformation. Vitro Cell Devel Biol Plant 40:1–22
Qu D, Song Y, Li WM, Pei XW, Wang ZX, Jia SR, Zhang YQ (2011) Isolation and characterization of the organ-specific and light-inducible promoter of the gene encoding rubisco activase in potato (Solanum tuberosum). Genet Mol Res 10:621–631
Rahamkulov I (2019) Comparison of different promoters activity in transgenic potatoes. Nigde Omer Halisdemir University, Nigde, Turkey, Page No, p 74
Rowe CR (1993) Potato health manegement. Ohio State University, USA, Department of Plant Pathology
Sambrook J, Russell DW (2001) Molecular cloning: a laboratory manual. Cold Spring Harbor Laboratory Press, New York
Somssich M (2019) A short history of the CaMV 35S promoter. PeerJ Prepr. https://doi.org/10.7287/peerj.preprints.27096v2
Sunilkumar G, Mohr L, Lopata-Finch E, Emani C, Rathore KS (2002) Developmental and tissue-specific expression of CaMV 35S promoter in cotton as revealed by GFP. Plant Mol Biol 50:463–474
Tabashnik BE, Carrie`re Y (2017) Surge in insect resistance to transgenic crops and prospects for sustainability. Nat Biotechnol 35:926–935
Tanabe N, Tamoi M, Shigeoka S (2015) The sweet potato RbcS gene (IbRbcS1) promoter confers high-level and green tissue-specific expression of the GUS reporter gene in transgenic Arabidopsis. Gene 567:244–250
Veale MA, Slabbert MM, Van Emmenes L (2012) Agrobacterium-mediated transformation of potato cv. Mnandi for resistance to the potato tuber moth (Phthorimaea operculella). S Afr J Bot 80:7–74
Zhu LP, Yu Z, Zou CX, Li QL, Chuan Y (2010) Plant stress-inducible promoters and their function. Yi Chuan = Hereditas 32:229–234
Zuraida AR, Zulkifli RK, Alizah Z, Zamri Z, Aziz A (2013) Hygromycin as selective marker in Agrobacterium-mediated genetic transformation of indica rice MR 219. J Trop Agric Food Sci 41:71–79
Acknowledgements
The present study is a part of research project supported by TÜBİTAK (Grant No. 215O520), where we used pRCA promoter to drive the expression of EcR dsRNAs in green part of potato plants. The authors are highly thankful to Tübitak for providing fellowship to Mr. Ilhom Rahamkulov from the project budget. Authors are grateful to Prof. Dr. Mehmet Emin Çalışkan for providing seed tuber of Lady Olympia used in the study. Special thanks to Dr. Emre Aksoy for his technical help during experiments and interpretation of results. We are also thankful to Dr. Halil Toktay for providing access to microscope facility.
Author information
Authors and Affiliations
Contributions
The data presented in the manuscript is MS thesis work of IR who completed his studies under the supervision of AB.
Corresponding author
Ethics declarations
Conflict of interest
Authors declare no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Rahamkulov, I., Bakhsh, A. Tissue-specific and stress-inducible promoters establish their suitability for containment of foreign gene(s) expression in transgenic potatoes. 3 Biotech 10, 426 (2020). https://doi.org/10.1007/s13205-020-02350-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-020-02350-x