Abstract
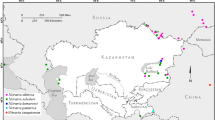
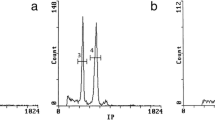
Four spontaneous diploid taxa (2n = 2x = 16 chromosomes) of the genus Hedysarum L. were studied for the first time using molecular cytogenetic approach and a tetraploid one as an additional comparative control. This study follows the work undertaken on this genus as part of the preservation and valorization of fodder and pastoral phytogenetic resources in Algeria. Fluorescent in situ hybridization (FISH) results revealed the same number of 35S rDNA loci for all diploid taxa, but differences in the number and position of 5S ribosomal genes. Chromomycin fluorochrome banding displayed that all chromomycin positive bands (GC-rich heterochromatin) were colocalized with rDNA signals. 2C DNA values (genome size), estimated by flow cytometry, indicated greater than two-fold difference among diploid taxa where nuclear DNA amount ranged from 2C = 1.26 pg in H. carnosum Desf. to 2C = 3.36 pg in H. naudinianum Coss. Genome size of tetraploid H. perrauderianum Coss. & Durieu (2n = 4x = 32) also estimated for the first time, was 6.64 pg which is almost twice of the value of diploid H. naudinianum 2C = 3.36 pg. Palynological analysis by light microscopy shows two types of pollen grains: prolate and tricolpate for H. naudinianum and H. perrauderianum, subprolate and tricolporate for H. carnosum, H. spinosissimum subsp. capitatum (Desf.) Asch. & Gr. and H. pallidum Desf. These new data enrich the knowledge about relationships among these species and also allow proposing and discussing the possible origin of the tetraploid H. perrauderianum from the diploid H. naudinianum.



Similar content being viewed by others
Code availability
Not applicable.
References
Abdelguerfi-Berrekia R, Abdelguerfi A, Bounaga N, Guittonneau CG (1986) Chromosome number reports 90. Taxon 35:197
Abdelguerfi-Berrekia R, Abdelguerfi A, Bounaga N, Guittonneau GG (1988) Contribution à l’étude des espèces spontanées du genre Hedysarum L. en Algérie. III Dénombrement chromosomique chez 29 populations de huit (8) espèces. Ann Inst Nat Agro El- Harrach 12(1):233–247
Abdelguerfi-Berrekia R, Abdelguerfi A, Bounaga N, Guittonneau GG (1991) Répartition des espèces spontanées du genre Hedysarum selon certains facteurs du milieu en Algérie. Fourrages 126:187–207
Akpinar N, Yildiz B (1999) Nuclear DNA contents of some endemic Hedysarum L. species. Turk J Bot 23:229–232
Ansari HA, Ellison NW, Reader SM, Badaeva ED, Friebe B, Miller TE, Williams WM (1999) Molecular cytogenetic organization of 5S and 18S–16S rDNA loci in white clover (Trifolium repens L.) and related species. Ann Bot 83:199–206. https://doi.org/10.1006/anbo.1998.0806
Arano H, Saito H (1980) Cytological studies in family Umbelliferae 5. Karyotypes of seven species in subtribe Seselinae. La Kromosomo II 17:471–480
Arslan E, Ertugrul K, Tugay O, Dural H (2012) Karyological studies of the genus Onobrychis Mill. and the related genera Hedysarum L. and Sartoria Boiss. and Heldr. (Fabaceae, Hedysareae) from Turkey. Caryologia 65:11–17. https://doi.org/10.1080/00087114.2012.678079
Bancheva S, Greilhuber J (2006) Genome size in Bulgarian Centaurea s.l. (Asteraceae). Plant Syst Evol 257:95–117. https://doi.org/10.1007/s00606-005-0384-7
Baziz K, Benamara-Bellagha M, Pustahija F, Brown SC, Siljak-Yakovlev S, Khalfallah N (2014) First karyotype analysis, physical rDNA mapping, and genome size assessment in 4 North African Astragalus taxa (Fabaceae). Turk J Bot 38:1248–1258. https://doi.org/10.3906/bot-1405-40
Benhizia H, Benhizia Y, Ghernoub L, Siljak-Yakovlev S, Khalfallah N (2013) Meiotic behavior and karyotype features of endangered endemic fodder species Hedysarum perrauderianum (Fabaceae) in some populations from Algeria. Caryologia 66:195–204. https://doi.org/10.1080/00087114.2013.821838
Benhizia H, Rached-Mosbah O, Benhizia Y, Kouachi A, Khalfallah N (2003) Etude cytogénétique d’Hedysarum pallidum Desf., espèce endémique Nord-africaine, tolérante à l’antimoine. Univ Const Sci Technol 20:7–13
Bogunić F, Siljak-Yakovlev S, Muratović E, Ballian D (2011) Different karyotype patterns among allopatric Pinus nigra (Pinaceae) populations revealed by molecular cytogenetics. Plant Biol 13:194–200. https://doi.org/10.1111/j.1438-8677-2010.00326x
Bou Dagher-Kharrat M, Abdel-Samad N, Douaihy BC, Abdel-Samad F, Bourge M, Siljak-Yakovlev S, Brown S (2013) Nuclear DNA C-values for biodiversity screening: case of the Lebanese flora. Plant Biosyst 147:1228–1237. https://doi.org/10.1080/11263504.2013.861530
Bourge M, Brown SC, Siljak-Yakovlev S (2018) Flow cytometry as tool in plant sciences, with emphasis on genome size and ploidy level assessment. Genet Appl 2:1–12. https://doi.org/10.31383/ga.vol2iss2pp1-12
Burchardt P, Souza-Chies TT, Chauveau O, Callegari-Jacques SM, Brisolara-Corrêa L, Inácio CD, Eggers L, Siljak-Yakovlev S, de Campos SJM, Kaltchuk-Santos E (2018) Cytological and genome size data analyzed in a phylogenetic frame: evolutionary implications concerning Sisyrinchium taxa (Iridaceae: Iridoideae). Genet Mol Biol 41:288–307. https://doi.org/10.1590/1678-4685-gmb-2017-0077
Chalup L, Grabiele M, Neffa VS, Seijo G (2014) DNA content in South American endemic species of Lathyrus. J Plant Res 127:469–480. https://doi.org/10.1007/s10265-014-0637-z
Chennaoui H, Marghali S, Marrakchi M, Trifi-Farah N (2007) Phylogenetic relationships in the North African genus Hedysarum as inferred from ITS sequences of nuclear ribosomal DNA. Genet Resour Crop Evol 54:389–397. https://doi.org/10.1007/s10722-006-0001-9
Dural H, Citak BY (2015) Morphology and anatomy of Hedysarum pannosum (Boiss.) Boiss. (Fabaceae). Act Bot Croat 74:19–29. https://doi.org/10.1515/botcro-2015-0009
Erdtman G (1945) Pollen morphology and plant taxonomy. Labiateae, Verbenaceae and Avicenniaceae. Svensk Botanisk Tidskrift 39:279–285
Erdtman G (1952) Pollen morphology and plant taxonomy: angiosperms (an introduction to palynology). Almqvist & Wikse, Stockholm, Sweden
Falistocco E, Maurelli L (1990) Chromosome study in Hedysarum coronarium L. Annali di Botanica (Rome) 48:81–86
Furuta Y, Nishikawa K (1991) 4-variation in nuclear and individual chromosomal DNA content and its role in evolution of plants. Dev Plant Genet Breed 2(A):71–85. https://doi.org/10.1016/B978-0-444-88259-2.50008-4
Garcia S, Garnatje T, Hidalgo O, McArthur ED, Siljak-Yakovlev S, Vallès J (2007) Extensive ribosomal DNA (18S–5,8S–26S and 5S) colocalization in the North American endemic sagebrushes (subgenus Tridentatae, Artemisia, Asteraceae) revealed by FISH. Plant Syst Evol 267:79–92. https://doi.org/10.1007/s00606-007-0558-6
Garcia S, Garnatje T, Kovařík A (2012) Plant rDNA database: ribosomal DNA loci information goes online. Chromosoma 121:389–394. https://doi.org/10.1007/s00412-012-03687
Garcia S, Garnatje T, Pellicer J, McArthur ED, Siljak-Yakovlev S, Vallès J (2009) Ribosomal DNA, heterochromatin and correlation with genome size in diploid and polyploidy North American endemic sagebrushes (Artemisia, Asteraceae). Genome 52:1012–1024. https://doi.org/10.1139/G09-77
Garcia S, Panero JL, Siroky J, Kovařík A (2010) Repeated reunions and splits feature the highly dynamic evolution of 5S and 35S ribosomal RNA genes (rDNA) in the Asteraceae family. BMC Plant Biol 10:176. https://doi.org/10.1186/1471-2229-10-176
Ghanavati F, Amirabadizadeh H (2012) Pollen grain morphology in Iranian Hedysareae (Fabaceae). Crop Breed J 2:25–33. https://doi.org/10.22092/cbj.2012.100413
Greilhuber J, Doležele J, Lysak MA, Bennett MD (2005) The origin evolution and proposed stabilization of the terms ʽgenome sizeʼ and ʽC-valueʼ to describe nuclear DNA contents. Ann Bot 95:255–260. https://doi.org/10.1093/aob/mci019
Hamon P, Siljak-Yakovlev S, Srisuwan S, Robin O, Poncet V, Hamon S, de Kochko A (2009) Physical mapping of rDNA and heterochromatin in chromosomes of 16 Coffea species: a revised view of species differentiation. Chromosome Res 17:291–304. https://doi.org/10.1007/s10577-009-9033-2
Hubac JM (1972) Relations entre les dimensions du pollen et le niveau de polyploïdie dans le complexe du Campanula rotundifolia L. s.l. Bull Soc bot Fr 119:279–286. https://doi.org/10.1080/00378941.1972.10839032
Inceer H, Kalmuk NA, Čížková J, Doležel J (2018) Genome size in some taxa of Crepis L. (Asteraceae) from Turkey. Caryologia 71:217–223. https://doi.org/10.1080/00087114.2018.1460057
Issolah R (2018) Diversité et valorisation des ressources fourragères en Algérie : cas des genres Trifolium L. et Hedysarum L. (Fabaceae). In: Issolah R. et Abbas K., éd. INRAA (2018) Actes du Congrès national sur les ressources phytogénétiques: évaluation, conservation et valorisation; Oct 22–23 et 24; Alger. Algérie, pp 87–104
Issolah R, Benhizia H, Khalfallah N (2006) Karyotype variation within some natural populations of sulla (Hedysarum coronarium L., Fabaceae) in Algeria. Genet Resour Crop Evol 53:1653–1664. https://doi.org/10.1007/s10722-005-5287-5
Kolano B, Tomczak H, Molewska R, Jellen EN, Maluszynska J (2012) Distribution of 5S and 35S rRNA gene sites in 34 Chenopodium species (Amaranthaceae). Bot J Linn Soc 170:220–231. https://doi.org/10.1111/j.1095-8339.2012.01286.x
Leitch IJ, Chase MW, Bennett MD (1998) Phylogenetic analysis of DNA C-Values provides evidence for a small ancestral genome size in flowering plants. Ann Bot 82:85–94. https://doi.org/10.1006/anbo.1998.0783
Leitch IJ, Johnston E, Pellicer J, Hidalgo O, Bennett MD (2019) Plant DNA C-values database release 7.1, April 2019. https://cvalues.science.kew.org
Levan A, Fredga K, Sandberg AA (1964) Nomenclature for centromeric position on chromosomes. Hereditas 52:201–220. https://doi.org/10.1111/j.1601-5223.1964.tb01953.x
Maluszynska J, Heslop-Harrison JS (1991) Localization of tandemly repeated DNA sequences in Arabidopsis thaliana. Plant J 12:159–166. https://doi.org/10.1111/j.1365-313x.1991.00159
Maluszynska J, Heslop-Harrison JS (1993) Physical mapping of rDNA loci in Brassica species. Genome 36:774–781. https://doi.org/10.1139/g93-102
Marie D, Brown SC (1993) A cytometric exercise in plant DNA histograms with, 2C values for 70 species. Biol Cell 78:41–51. https://doi.org/10.1016/0248-4900(93)90113-S
Marinho RC, Mendes-Rodrigues C, Bonetti AM, Oliveira PE (2013) Pollen and stomata morphometrics and polyploidy in Eriotheca (Malvaceae-Bombacoideae). Plant Biol 16:508–511. https://doi.org/10.1111/plb.12135
Pavlova DK, Manova VI (2000) Pollen morphology of the genera Onobrychis and Hedysarum (Hedysareae, Fabaceae) in Bulgaria. Ann Bot Fenn 37(3):207–217
Pire SML (1974) Estudio palynológico de la tribu "Hedysareae" (Leguminosae). Bonplandia 3(12):143–169
Poggio L, Rosato M, Chiavarino AM, Naranjo CA (1998) Genome size and environmental correlations in Maize (Zea mays ssp. mays, Poaceae). Ann Bot 82(Suppl 1):107–115. https://doi.org/10.1006/anbo.1998.0757
Pons A (1958) Le pollen. Que sais-je? Presses universitaires de France
Quezel P, Santa S (1962) Nouvelle flore de l’Algérie et des régions désertiques méridionales (ed). Centre National de la Recherche Scientifique (CNRS), France, pp 539–542
Reille M (1992) Pollen et spores d’Europe et d’Afrique du Nord. (ed). Marseille: Laboratoire de Botanique Historique et Palynologie
Roa F, Guerra M (2015) Non-random distribution of 5S rDNA sites and its association with 45S rDNA in plant chromosomes. Cytogenet Genome Res 146:243–249. https://doi.org/10.1159/000440930
Sastri DC, Hilu K, Appels R, Lagudah ES, Playford J, Baum BR (1992) An overview of evolution on plant 5S-DNA. Plant Syst Evol 183:169–181. https://doi.org/10.1007/BF00940801
Sato S, Katoh N, Iwai S, Hagimori M (2005) Frequency of Spontaneous Polyploidization of Embryos Regenerated from Cultured Anthers or Microspores of Brassica rapa var. pekinensis L. and B. oleracea var. capitata L. Breed Sci 55:99–102
Schinkel CCF, Kirchheimer B, Dullinger S, Geelen D, De Storme N, Hörandl E (2017) Pathways to polyploidy: indications of a female triploid bridge in the alpine species Ranunculus kuepferi (Ranunculaceae). Plant Syst Evol 303:1093–1108. https://doi.org/10.1007/s00606-017-1435-6
Schmidt T, Schwarzacher T, Heslop-Harrison JS (1994) Physical mapping of rRNA genes by fluorescent in situ hybridization and structural analysis of 5S rRNA genes and intergenic spacer sequences in sugar beet (Beta vulgaris). Theor Appl Genet 88:629–636. https://doi.org/10.1007/BF01253964
Siljak-Yakovlev S, Cerbah M, Coulaud J, Stoian V, Brown SC, Zoldos V, Jelenic S, Papes D (2002) Nuclear DNA content, base composition, heterochromatin and rDNA in Picea omorika and Picea abies. Theor Appl Genet 104:505–512. https://doi.org/10.1007/s001220100755
Siljak-Yakovlev S, Godelle B, Zoldos V, Vallès J, Garnatje T, Hidalgo O (2017) Evolutionary implications of heterochromatin and rDNA in chromosome number and genome size changes during dysploidy: a case study in Reichardia genus. PLoS ONE 12:1–21. https://doi.org/10.1371/journal.pone.0182318
Siljak-Yakovlev S, Pustahija F, Šolić EM, Bogunić F, Muratović F, Bašić N, Catrice O, Brown SC (2010) Towards a genome size and chromosome number database of Balkan flora: C-values in 343 taxa with novel values for 242. Adv Sci Lett 3:190–213. https://doi.org/10.1166/asl.2010.1115
Trifi-Farah N, Marrakchi M (2001) Hedysarum phylogeny mediated by RFLP analysis of nuclear ribosomal DNA. Genet Resour Crop Evol 48:339–345. https://doi.org/10.1023/A:1012051410501
Ubera JL, Prados AM, Tormo R, Domínguez E (1988) Estudio del sistema apertural de Hedysarum L. y Onobrychis miller (Fabaceae) y sus implicaciones taxonomicas. Anales de la asociación de Palinólogos de Lengua Española 4:7–13
Wicke S, Costa A, Munoz J, Quandt D (2011) Restless 5S: the re-arrangement(s) and evolution of the nuclear ribosomal DNA in land plants. Mol Phylogenet Evol 61:321–332. https://doi.org/10.1016/j.ympev.2011.06.023
Wielgorskaya T (1995) Dictionary of generic names of seed plants. Takhtajan A. Consulting editor, Columbia University Press
Wodehouse RP (1935) Pollen grains. 3st ed 1965. Hafner, NewYork
Yamauchi A, Hosokawa A, Nagata H, Shimoda M (2004) Triploid bridge and role of parthenogenesis in the evolution of autopolyploidy. Am Nat 164(1):101–112. https://doi.org/10.1086/421356
Yildiz K, Gücel S (2006) Chromosome numbers of 16 endemic plants taxa from Northern Cyprus. Turk J Bot 30(3):181–192
Acknowledgements
The authors are very grateful to Odile Robin for her help in cytogenetics and Dr. Michael Bourge for his expert assistance in flow cytometry on the Platform of Cytometry, Institute of Integrative Biology of the Cell (I2BC), Gif-sur-Yvette.
Funding
This work was partially supported by the North Atlantic Treaty Organization-Science for Peace and Security Program (Collaborative Linkage Grant No. 983838).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No potential conflict of interest was reported by the authors.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Availability of data and material
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Benhizia, H., Benhizia, Y., Djeghar, R. et al. Cytogenetic characterization, nuclear genome size, and pollen morphology of some Hedysarum L. taxa (Fabaceae) from Algeria, with emphasis on the origin of H. perrauderianum Coss. & Durieu. Genet Resour Crop Evol 68, 679–691 (2021). https://doi.org/10.1007/s10722-020-01016-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10722-020-01016-0