Abstract
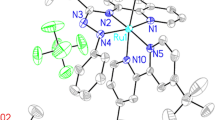
A series of new half-sandwich Ru(II) complex salts of the general formula [(η6-arene)RuX(L)]PF6 (where arene = p-cymene, X = Br and ligand L1 = N-tert-butyl-N-(pyridyl methylene) amine (complex 1), ligand L2 = N-isopropyl-2-pyridine carbaldimine (complex 2), ligand L3 = (2,5-dimethyl-phenyl)-pyridin-2-yl methylene amine (complex 3), ligand L4 = (3,5-dimethyl-phenyl)-pyridin-2-yl methylene (complex 4) and arene = C6H6, X = I, ligand L5 = (4-bromo-phenyl)-pyridin-2-yl methylene (complex 5) have been synthesized by reacting the ruthenium arene precursors [(η6-arene)Ru(μ-X)X]2 with N,N-bidentate ligands in a 1:2 ratio. Full characterization of the complexes was accomplished by physicochemical and spectroscopic methods and X-ray structures of all the complexes 1–5. The crystal structures reveal that the ruthenium centers in 1–5 are coordinated to the N,N-bidentate ligand in a bidentate manner, to the respective halide, and to the arene ring to give a pseudo-tetrahedral geometry around them. The whole arrangement is referred to as the three-legged piano stool, in which the N,N-bidentate ligand and the halide (Br or I) serve as the base occupying three coordination sites, while the arene ring serves as the apex of the stool and occupy the remaining coordination sites.









Similar content being viewed by others
References
Kumar P, Gupta RK, Pandey DS (2014) Half-sandwich arene ruthenium complexes: synthetic strategies and relevance in catalysis. Chem Soc Rev 43:707–733
Gichumbi JM, Friedrich HB, Omondi B (2016) Application of arene ruthenium(II) complexes with pyridine-2-carboxaldimine ligands in the transfer hydrogenation of ketones. J Mol Catal A Chem 416:29–38
Gichumbi JM, Friedrich HB, Omondi B (2016) Synthesis and characterization of half-sandwich ruthenium(II) complexes with N-alkyl pyridyl-imine ligands and their application in transfer hydrogenation of ketones. Trans Met Chem 41:867–877
Gichumbi JM, Friedrich HB, Omondi B (2017) Synthesis and characterization of some new half-sandwich ruthenium(II) complexes with bidentate N,N′-ligands and their application in alcohol oxidation. Inorg Chim Acta 456:55–63
Gichumbi JM, Friedrich HB, Omondi B (2018) Synthesis and characterization of piano-stool ruthenium complexes with N,N′-pyridine imine bidentate ligands and their application in styrene oxidation. J Organomet Chem 808:87–96
Furstner A, Picquet M, Bruneau C, Dixneuf PH (1998) Cationic ruthenium allenylidene complexes as a new class of performing catalysts for ring-closing metathesis. Chem Commun 12:1315–1316
Davenport AJ, Davies DL, Fawcett J, Russell DR (2004) Arene-ruthenium complexes with salicyloxazolines: diastereoselective synthesis, configurational stability and applications as asymmetric catalysts for Diels–Alder reactions. Dalton Trans 9:1481–1492
Murray BS, Babak MN, Hartinger CG, Dyson PJ (2016) The development of RAPTA compounds for the treatment of tumors. Coord Chem Rev 306:86–114
Gichumbi JM, Omondi B, Lazarus G, Singh M, Shaikh N, Chenia HY, Friedrich HB (2017) Influence of halogen substitution in the ligand sphere on the antitumor and antibacterial activity of half-sandwich ruthenium(II) complexes [RuX (η6-arene)(C5H4N-2-CH=N-Ar)]+. Z Anorg Allg Chem 643:699–711
Romero-Canelón I, Salassa L, Sadler PJ (2013) The contrasting activity of iodido versus chlorido ruthenium and osmium arene azo- and imino-pyridine anticancer complexes: control of cell selectivity, cross-resistance, p53 dependence, and apoptosis pathway. J Med Chem 56:1291–1300
Murray BS, Menin L, Scopelliti R, Dyson PJ (2014) Conformational control of anticancer activity: the application of arene-linked dinuclear ruthenium(II) organometallics. Chem Sci 5:2536–2545
Ackermann L (2015) Robust ruthenium(II)-catalyzed C–H arylations: carboxylate assistance for the efficient synthesis of angiotensin-II-receptor blockers. Org Process Res Dev 19:260–269
Fernandez FE, Puerta MC, Valerga P (2012) Ruthenium(II) picolyl-NHC complexes: synthesis, characterization, and catalytic activity in amine N-alkylation and transfer hydrogenation reactions. Organometallics 31:6868–6879
A CSD (Cambridge Structural Database) search of arene ruthenium(II)-X reveals (February 2020), 4 hits for X = Br, 8 hits for X = I and 587 hits for X = Cl
Gichumbi JM, Friedrich HB, Omondi B (2016) Solvato-polymorph of [(η6-C6H6) RuCl (L)] PF6 (L = (2, 6-dimethyl-phenyl-pyridin-2-yl methylene amine). J Mol Struct 1113:55–59
Gichumbi OB, Friedrich HB (2017) Crystal structure of (η6-benzene)-(N-(2, 6-dimethyl phenyl)-1-(pyridin-2-yl) methanimine-κ2 N,N′) ruthenium(II) perchlorate monohydrate, C20H20Cl2N2O5Ru. Z Kristallogr New Cryst Struct 232:301–303
Gichumbi JM, Friedrich HB, Omondi B (2017) Crystal structure of chlorido-(η6-1-isopropyl-4-methyl benzene)-(1-(pyridin-2-yl)-N-(p-tolyl) methanimine-κ2 N,N′) ruthenium(II) hexafluorophosphate (V), C23H26ClF6N2PRu. Z Kristallogr New Cryst Struct 232:285–287
Bennett MA, Smith AK (1974) Arene ruthenium(II) complexes formed by dehydrogenation of cyclohexadiene with ruthenium(III) trichloride. J Chem Soc Dalton Trans 2:233–241
Mendoza-Ferri MG, Hartinger CG, Nazarov AA, Eichinger RE, Jakupec MA, Severin K, Keppler BK (2009) Influence of the arene ligand, the number and type of metal centers, and the leaving group on the in vitro antitumor activity of polynuclear organometallic compounds. Organometallics 28:6260–6265
Hu H, Zhang L, Gao H, Zhu F, Wu Q (2014) Design of thermally stable amine-imine nickel catalyst precursors for living polymerization of ethylene: effect of ligand substituents on catalytic behavior and polymer properties. Chem Eur J 20:3225–3233
Bruker-AXS, Bruker-AXS, Madison, Wisconsin, USA, 2009
Sheldrick G (2008) Acta Cryst Sec A 64:112–122
Marchetti F, Pettinari C, Cerquetella A, Cingolani A, Pettinari R, Monari M, Wanke R, Kuznetsov ML, Pombeiro AJL (2009) Switching between κ2 and κ3 Bis(pyrazol-1-yl)acetate Ligands by tuning reaction conditions: synthesis, spectral, electrochemical, structural, and theoretical studies on arene-Ru(II) derivatives of bis(azol-1-yl)acetate ligands. Inorg Chem 48:6096–6108
Chow MJ, Licona C, Yuan Qiang Wong D, Pastorin G, Gaiddon C, Ang WH (2014) Discovery and investigation of anticancer ruthenium-arene Schiff-base complexes via water-promoted combinatorial three-component assembly. J Med Chem 57:6043–6059
A CSD (Cambridge Structural Database) search of arene ruthenium(II) -with imino N atom attached to a substituted phenyl ring or an alkyl group (January 27, 2020), reveals 46 hits
Li J, Guo L, Tian Z, Zhang S, Xu Z, Han Y, Li R, Li Y, Liu Z (2018) Half-sandwich iridium and ruthenium complexes: effective tracking in cells and anticancer studies. Inorg Chem 57:13552–13563
Thangavel S, Boopathi S, Mahadevaiah N, Kolandaivel P, Pansuriya PB, Friedrich HB (2016) Catalytic oxidation of primary aromatic alcohols using half sandwich Ir(III), Rh(III) and Ru(II) complexes: a practical and theoretical study. J Mol Catal A Chem 423:160–171
Tian M, Li J, Zhang S, Guo L, He X, Kong D, Zhang H, Liu Z (2017) Half-sandwich ruthenium(II) complexes containing N,N-chelated imino-pyridyl ligands that are selectively toxic to cancer cells. Chem Commun 53:12810–12813
Lalrempuia R, Kollipara MR, Carroll PJ (2003) Syntheses and characterization of arene ruthenium (II) complexes containing N,N′-donor Schiff base ligands. Crystal and molecular structure of [(η6-C6Me6)Ru(C5H4N-2-CH-N-C6H4-p-NO2)]PF6. Polyhedron 22:605–609
Govender P, Renfrew AK, Clavel CM, Dyson PJ, Therrien B, Smith GS (2011) Antiproliferative activity of Chelating N, O-and N,N-ruthenium(II) arene functionalised poly(propyleneimine) dendrimer scaffolds. Dalton Trans 40:1158–1167
Mandal SK, Chakravarty AR (1992) Arene ruthenium complexes of N,N′- and N,O-donor Schiff base ligands: an X-ray structure of [(η6-p-cymene) RuCl(C5H4N-2-CH-NC6H4-p-Me)Cl·C6H6·H2O. Polyhedron 11:823–827
Gichumbi JM, Omondi B, Friedrich HB (2018) Crystal structure of (η6-1-isopropyl-4-methyl benzene)-(N-(2,5-dichloro phenyl)-1-(pyridin-2-yl) methanimine-κ2 N, N′) ruthenium(II) perchlorate, C22H22Cl4N2O4Ru. Z Kristallogr New Cryst Struct 233:423–425
Gichumbi JM, Omondi B, Friedrich HB (2017) Crystal structure of chlorido-(N-(2, 5-dichloro phenyl)-1-(pyridin-2-yl) methanimine-κ2N, N′)(η6-1-isopropyl-4-methyl benzene) ruthenium(II) tetrafluoroborate, C22H22Cl3N2BF4Ru. Z Kristallogr New Cryst Struct 232:309–311
Dolomanov OV, Bourhis LJ, Gildea RJ, Howard JAK, Puschmann H (2009) OLEX2: a complete structure solution, refinement and analysis program. J Appl Cryst 42:339–341
Binnani C, Rai RK, Tyagi D, Mobin SM, Singh SK (2018) Ligand-tuned C–H bond activation/arylation of 2-arylpyridines over pyridine-based N, O/N, N ligated ruthenium-arene complexes. Eur J Inorg Chem 2018:1435–1445
Richter S, Singh S, Draca D, Kate A, Kumbhar A, Kumbhar AS, Maksimovic-Ivanic D, Mijatovic S, Lönnecke P, Hey-Hawkins E (2016) Antiproliferative activity of ruthenium(II) arene complexes with mono- and bidentate pyridine-based ligands. Dalton Trans 45:13114–13125
Acknowledgements
We gratefully acknowledge the financial support from the NRF, THRIP (Grant No. TP. 1208035643), and UKZN (URF). Gichumbi, JM thanks Prof. E. N. Njoka for his support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gichumbi, J.M., Friedrich, H.B. & Omondi, B. Comparative structural description of five arene ruthenium(II) complexes of N,N-bidentate Schiff base ligands to related complexes from literature. Transit Met Chem 46, 17–28 (2021). https://doi.org/10.1007/s11243-020-00417-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11243-020-00417-5