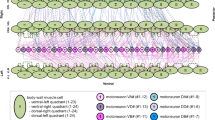
Abstract
During fictive locomotion cat lumbar motoneurons exhibit changes in membrane proprieties including a decrease in voltage threshold (Vth), afterhyperpolarization (AHP) and input resistance (Rin) and an increase in non-linear membrane property. The impact of these changes on the motoneuron recruitment remains unknown. Using modeling approach we investigated the channel mechanism regulating the motoneuron recruitment. Three types of motoneuron pools including slow (S), fatigue-resistant (FR) and fast-fatigable (FF) motoneurons were constructed based on the membrane proprieties of cat lumbar motoneurons. The transient sodium (NaT), persistent sodium (NaP), delayed-rectifier potassium [K(DR)], Ca2+-dependent K+ [K(AHP)] and L-type calcium (CaL) channels were included in the models. Simulation results showed that (1) Strengthening synaptic inputs increased the number of recruitments in all three types of motoneurons following the size principle. (2) Increasing NaT or NaP or decreasing K(DR) or K(AHP) lowered rheobase of spike generation thus increased recruitment of motoneuron pools. (3) Decreasing Rin reduced recruitment in all three types of motoneurons. (4) The FF-type motoneuron pool, followed by FR- and S-type, were the most sensitive to increase of synaptic inputs, reduction of Rin, upregulation of NaT and NaP, and downregulation of K(DR) and K(AHP). (5) Increasing CaL enhanced overall discharge rate of motoneuron pools with little effect on the recruitment. Simulation results suggested that modulation of ionic channels altered the output of motoneuron pools with either modulating the number of recruited motoneurons or regulating the overall discharge rate of motoneuron pools. Multiple channels contributed to the recruitment of motoneurons with interaction of excitatory and inhibitory synaptic inputs during walking.











Similar content being viewed by others
Change history
28 January 2021
The authors find several printing errors in the equations in the final versions on line and in print proof. However, there were no such errors in the submitted proof.
References
Binder, M. D., Heckman, C. J., & Powers, R. K. (2002). Relative strengths and distributions of different sources of synaptic input to the motoneurone pool: Implications for motor unit recruitment. Advances in Experimental Medicine and Biology, 508, 207–212. https://doi.org/10.1007/978-1-4615-0713-0_25.
Brownstone, R. M., Jordan, L. M., Kriellaars, D. J., Noga, B. R., & Shefchyk, S. J. (1992). On the regulation of repetitive firing in lumbar motoneurones during fictive locomotion in the cat. Experimental Brain Research, 90(3), 441–455. https://doi.org/10.1007/bf00230927.
Brownstone, R. M., Gossard, J. P., & Hultborn, H. (1994). Voltage-dependent excitation of motoneurones from spinal locomotor centres in the cat. Experimental Brain Research, 102(1), 34–44. https://doi.org/10.1007/bf00232436.
Carlin, K. P., Jones, K. E., Jiang, Z., Jordan, L. M., & Brownstone, R. M. (2000). Dendritic L-type calcium currents in mouse spinal motoneurons: Implications for bistability. European Journal of Neuroscience, 12(5), 1635–1646. https://doi.org/10.1046/j.1460-9568.2000.00055.x.
Carlin, K. P., Dai, Y., & Jordan, L. M. (2006). Cholinergic and serotonergic excitation of ascending commissural neurons in the thoraco-lumbar spinal cord of the neonatal mouse. Journal of Neurophysiology, 95(2), 1278–1284.
Cazalets, J. R., Sqalli-Houssaini, Y., & Clarac, F. (1992). Activation of the central pattern generators for locomotion by serotonin and excitatory amino acids in neonatal rat. The Journal of Physiology, 455, 187–204.
Cheng, Y., Ge, R., Chen, K., & Dai, Y. (2019). Modulation of NMDA-mediated intrinsic membrane properties of ascending commissural interneurons in neonatal rat spinal cord. Journal of Integrative Neuroscience, 18(2), 163–172.
Cowley, K. C., & Schmidt, B. J. (1997). Regional distribution of the Locomotor pattern generating network in the neonatal rat spinal cord. Journal of Neurophysiology, 77, 247–259.
Crill, E. W. (1996). Persistent sodium current in mammalian central neurons. Annual Review of Physiology, 58(1), 349–362. https://doi.org/10.1146/annurev.ph.58.030196.002025.
Dai, Y., & Jordan, L. M. (2010). Multiple patterns and components of persistent inward current with serotonergic modulation in locomotor activity–related neurons in cfos-EGFP mice. Journal of Neurophysiology, 103(4), 1712–1727. https://doi.org/10.1152/jn.01111.2009.
Dai, Y., & Jordan, L. M. (2011). Tetrodotoxin-, dihydropyridine-, and riluzole-resistant persistent inward current: Novel sodium channels in rodent spinal neurons. Journal of Neurophysiology, 106(3), 1322–1340. https://doi.org/10.1152/jn.00918.2010.
Dai, Y., Jones, K. E., Fedirchuk, B., & Jordan, L. M. (2000). Effects of voltage trajectory on action potential voltage threshold in simulations of cat spinal motoneurons. Neurocomputing, 32-33(none), 105–111. https://doi.org/10.1016/s0925-2312(00)00150-8.
Dai, Y., Jones, K. E., Fedirchuk, B., Mccrea, D. A., & Jordan, L. M. (2002). A modelling study of locomotion-induced hyperpolarization of voltage threshold in cat lumbar motoneurones. Journal of Physiology (Cambridge), 544(2), 521–536. https://doi.org/10.1113/jphysiol.2002.026005.
Dai, Y., Carlin, K. P., Li, Z., McMahon, D. G., Brownstone, R. M., & Jordan, L. M. (2009). Electrophysiological and pharmacological properties of Locomotor activity-related neurons in cfos-EGFP mice. Journal of Neurophysiology, 102(6), 3365–3383. https://doi.org/10.1152/jn.00265.2009.
Dai Y., Cheng Y., Fedirchuk B., Jordan L.M., & Chu J.H. (2018). Motoneuron output regulated by ionic channels: A modeling study of motoneuron frequency-current relationships during fictive locomotion. Journal of Neurophysiology, 120, 1840–1858, 2018. 10.1152/jn.00068.2018
Fedirchuk, B., & Dai, Y. (2004). Monoamines increase the excitability of spinal neurones in the neonatal rat by hyperpolarizing the threshold for action potential production. Journal of Physiology, 557, 355–361.
Fuglevand, A. J., Lester, R. A., & Johns, R. K. (2015). Distinguishing intrinsic from extrinsic factors underlying firing rate saturation in human motor units. Journal of Neurophysiology, 113(5), 1310–1322. https://doi.org/10.1152/jn.00777.2014.
Ge, R., Chen, K., Cheng, Y., & Dai, Y. (2019). Morphological and electrophysiological properties of serotonin neurons with NMDA modulation in the Mesencephalic Locomotor region of neonatal ePet-EYFP mice. Experimental Brain Research, 237(12), 3333–3350.
Gosgnach, S., Quevedo, J., Fedirchuk, B., & Mccrea, D. A. (2000). Depression of group Ia monosynaptic EPSPs in cat hindlimb motoneurones during fictive locomotion. The Journal of physiology, 526 Pt 3(3), 639. https://doi.org/10.1111/j.1469-7793.2000.00639.x.
Goslow, G. E., Cameron, W. E., & Stuart, D. G. (1977). The fast twitch motor units of cat ankle flexors. 1. Tripartite classification on basis of fatigability. Brain Research, 134(1), 35–46. https://doi.org/10.1016/0006-8993(77)90923-4.
Harvey, P. J. (2006). Persistent sodium currents and repetitive firing in motoneurons of the sacrocaudal spinal cord of adult rats. Journal of Neurophysiology, 96(3), 1141–1157. https://doi.org/10.1152/jn.00335.2005.
Heckman, C. J., & Binder, M. D. (1988). Analysis of effective synaptic currents generated by homonymous Ia afferent fibers in motoneurons of the cat. Journal of Neurophysiology, 60(6), 1946–1966. https://doi.org/10.1152/jn.1988.60.6.1946.
Heckman, C. J., & Binder, M. D. (1991). Computer simulation of the steady-state input-output function of the cat medial gastrocnemius motoneuron pool. Journal of Neurophysiology, 65(4), 952–994. https://doi.org/10.1016/0165-5728(91)90076-J.
Heckman, C. J., & Binder, M. D. (1992). Analysis of Ia-inhibitory synaptic input to cat spinal motoneurons evoked by vibration of antagonist muscles. Journal of Neurophysiology, 66(6), 1888–1893. https://doi.org/10.1152/jn.1991.66.6.1888.
Heckman, C. J., & Binder, M. D. (1993). Computer simulations of the effects of different synaptic input systems on motor unit recruitment. Journal of Neurophysiology, 70(5), 1827–1840. https://doi.org/10.1152/jn.1993.70.5.1827.
Helton, T. D., Xu, W., & Lipscombe, D. (2005). Neuronal L-type calcium channels open quickly and are inhibited slowly. The Journal of Neuroscience, 25, 10247–10251. https://doi.org/10.1523/jneurosci.1089-05.2005.
Henneman, E., Somjen, G., & Carpenter, D. O. (1965). Functional significance of cell size in spinal Motoneurons. Journal of Neurophysiology, 28(3), 560–580. https://doi.org/10.1152/jn.1965.28.3.560.
Hodgkin, A. L., & Huxley, A. F. (1990). A quantitative description of membrane current and its application to conduction and excitation in nerve. Bulletin of Mathematical Biology, 52(1–2), 25–71. https://doi.org/10.1113/jphysiol.1952.sp004764.
Hounsgaard, J., & Kiehn, O. (1989). Serotonin-induced bistability of turtle motoneurones caused by a nifedipine-sensitive calcium plateau potential. The Journal of Physiology, 414(1), 265–282. https://doi.org/10.1113/jphysiol.1989.sp017687.
Hounsgaard, J. O., & Kjaerulff. (1992). Ca2+-mediated plateau potentials in a subpopulation of interneurons in the ventral horn of the turtle spinal cord. European Journal of Neuroscience, 4, 183–188. https://doi.org/10.1111/j.1460-9568.1992.tb00865.x.
Hultborn, H., Brownstone, R. B., Toth, T. I., & Gossard, J. P. (2004). Key mechanisms for setting the input-output gain across the motoneuron pool. Progress in Brain Research, 143(143), 77–95. https://doi.org/10.1016/S0079-6123(03)43008-2.
Jones, K. E., & Bawa, P. (1999). A comparison of human motoneuron data to simulated data using cat motoneuron models. Journal of Physiology-Paris, 93(1–2), 43–59. https://doi.org/10.1016/S0928-4257(99)80135-1.
Kernell, D. (1969). Synaptic conductance changes and the repetitive impulse discharge of spinal motoneurones. Brain Research, 15(1), 291–294. https://doi.org/10.1016/0006-8993(69)90335-7.
Kernell, D., & Hultborn, H. (1990). Synaptic effects on recruitment gain: A mechanism of importance for the input-output relations of motoneurone pools. Brain Research, 507(1), 176–179. https://doi.org/10.1016/0006-8993(90)90542-J.
Kernell, D., & Zwaagstra, B. (1981). Input conductance, axonal conduction velocity and cell size among hindlimb motoneurones of the cat. Brain Research, 204(2), 311–326. https://doi.org/10.1016/0006-8993(81)90591-6.
Komendantov, A. O., & Ascoli, G. A. (2009). Dendritic excitability and neuronal morphology as determinants of synaptic efficacy. Journal of Neurophysiology, 101(4), 1847–1866. https://doi.org/10.1152/jn.01235.2007.
Krawitz, S., Fedirchuk, B., Dai, Y., Jordan, L. M., & McCrea, D. A. (2001). State-dependent hyperpolarization of voltage threshold enhances motoneurone excitability during fictive locomotion in the cat. The Journal of Physiology, 532, 271–281. https://doi.org/10.1111/j.1469-7793.2001.0271g.x.
Kuo, J. J., Lee, R. H., Zhang, L., & Heckman, C. J. (2006). Essential role of the persistent sodium current in spike initiation during slowly rising inputs in mouse spinal neurones. The Journal of Physiology, 574, 819–834. https://doi.org/10.1113/jphysiol.2006.107094.
Lee, R. H., & Heckman, C. J. (2001). Essential role of a fast persistent inward current in action potential initiation and control of rhythmic firing. Journal of Neurophysiology, 85(1), 472–478. https://doi.org/10.1007/s002320010038.
Liu, J., & Jordan, L. M. (2005). Stimulation of the parapyramidal region of the neonatal rat brain stem produces locomotor-like activity involving spinal 5-HT7 and 5-HT2A receptors. Journal of Neurophysiology, 94(2), 1392–1404.
Lombardo, J., & Harrington, M. A. (2016). Nonreciprocal mechanisms in up- and downregulation of spinal motoneuron excitability by modulators of KCNQ/Kv7 channels. Journal of Neurophysiology, 116(5), 2114–2124. https://doi.org/10.1152/jn.00446.2016.
Mccrea, D. A., & Rybak, I. A. (2007). Modeling the mammalian locomotor CPG: Insights from mistakes and perturbations. Progress in Brain Research, 165, 235–253. https://doi.org/10.1016/S0079-6123(06)65015-2.
Nielsen, J. B., Morita, H., Wenzelburger, R., Deuschl, G., Gossard, J. P., & Hultborn, H. (2019). Recruitment gain of spinal motor neuron pools in cat and human. Experimental Brain Research, 237(11), 2897–2909. https://doi.org/10.1007/s00221-019-05628-6.
Power, K. E., Carlin, K. P., & Fedirchuk, B. (2012). Modulation of voltage-gated sodium channels hyperpolarizes the voltage threshold for activation in spinal motoneurones. Experimental Brain Research, 217(2), 311–322. https://doi.org/10.1007/s00221-011-2994-3.
Powers, R. K., & Heckman, C. J. (2015). Contribution of intrinsic motoneuron properties to discharge hysteresis and its estimation based on paired motor unit recordings: A simulation study. Journal of Neurophysiology, 114(1), 184–198. https://doi.org/10.1152/jn.00019.2015.
Powers, R. K., & Heckman, C. J. (2017). Synaptic control of the shape of the motoneuron pool input-output function. Journal of Neurophysiology, 117(3), 1171–1184. https://doi.org/10.1152/jn.00850.2016.
Powers, R. K., Binder, M. D., & Matthews, P. B. C. (2000). Relationship between the time course of the afterhyperpolarization and discharge variability in cat spinal motoneurones. The Journal of Physiology, 528(1), 131–150. https://doi.org/10.1111/j.1469-7793.2000.t01-1-00131.x.
Powers, R. K., Elbasiouny, S. M., Rymer, W. Z., & Heckman, C. J. (2012). Contribution of intrinsic properties and synaptic inputs to motoneuron discharge patterns: A simulation study. Journal of Neurophysiology, 107(3), 808–823. https://doi.org/10.1152/jn.00510.2011.
Pratt, C. A., & Jordan, L. M. (1987). Ia inhibitory interneurons and Renshaw cells as contributors to the spinal mechanisms of fictive locomotion. Journal of Neurophysiology, 57(1), 56–71. https://doi.org/10.1152/jn.1987.57.1.56.
Qiang Z., Dongyan Y., Xingyu W., Weihang X., & Dai Y. A Simulation Study of Spinal Motoneuron Recruitment Regulated By Ionic Channels. The 13th Biennial Conference of Chinese Neuroscience Society (CNS 2019) P-082.
Rybak, I. A., Shevtsova, N. A., Lafreniere-Roula, M., & Mccrea, D. A. (2006). Modelling spinal circuitry involved in locomotor pattern generation: Insights from deletions during fictive locomotion. The Journal of Physiology, 577(2), 617–639. https://doi.org/10.1113/jphysiol.2006.118703.
SAH, P. (1996). Ca2+-activated K+ currents in neurons: Types, physiological roles and modulation. Trends in Neurosciences, 19, 150–154. https://doi.org/10.1016/S0166-2236(96)80026-9.
Schwindt, P. C., & Crill, W. E. (1980a). Properties of a persistent inward current in normal and tea-injected motoneurons. Journal of Neurophysiology, 43(6), 1700–1724. https://doi.org/10.1152/jn.1980.43.6.1700.
Schwindt, P., & Crill, W. (1980b). Role of a persistent inward current in motoneuron bursting during spinal seizures. Journal of Neurophysiology, 43(5), 1296–1318. https://doi.org/10.1152/jn.1980.43.5.1296.
Shefchyk, S. J., & Jordan, L. M. (1985). Motoneuron input-resistance changes during fictive locomotion produced by stimulation of the mesencephalic locomotor region. Journal of Neurophysiology, 54(5), 1101–1108. https://doi.org/10.1152/jn.1985.54.5.1101.
Tano, C., Martín, V., Eugenia, M., & Szczupak, L. (2015). Graded boosting of synaptic signals by low-threshold voltage-activated calcium conductance. Journal of Neurophysiology, 114(1), 332–340. https://doi.org/10.1152/jn.00170.2015.
Tansey, K. E., & Botterman, B. R. (1996). Activation of type-identified motor units during centrally evoked contractions in the cat medial gastrocnemius muscle. Ii. Motoneuron firing-rate modulation. Journal of Neurophysiology, 75(1), 38–50. https://doi.org/10.1152/jn.1996.75.1.38.
Theiss, R. D., Kuo, J. J., & Heckman, C. J. (2007). Persistent inward currents in rat ventral horn neurones. The Journal of Physiology, 580(2), 507–522. https://doi.org/10.1113/jphysiol.2006.124123.
Urban, N. N., Henze, D. A., & Barrionuevo, G. (1998). Amplification of Perforant-path EPSPs in CA3 pyramidal cells by LVA calcium and sodium channels. Journal of Neurophysiology, 80(3), 1558–1561. https://doi.org/10.1152/jn.1998.80.3.1558.
Vieira, M. F., & Kohn, A. F. (2007). Compartmental models of mammalian motoneurons of types S, FR and FF and their computer simulation. Computers in Biology & Medicine, 37(6), 842–860. https://doi.org/10.1016/j.compbiomed.2006.09.001.
Yakovenko, S., McCrea, D. A., Stecina, K., & Prochazka, A. (2005). Control of locomotor cycle durations. Journal of Neurophysiology, 94, 1057–1065.
Zengel, J. E., Reid, S. A., Sypert, G. W., & Munson, J. B. (1985). Membrane electrical properties and prediction of motor-unit type of medial gastrocnemius motoneurons in the cat. Journal of Neurophysiology, 53(5), 1323–1344.
Grants
This work was funded by The National Natural Science Foundation of China (31,571,222, Yue Dai).
Author information
Authors and Affiliations
Contributions
Y.D. proposed the research plan and Q.Z. designed simulation; Q.Z. performed experiments and analyzed data; Q.Z. drafted manuscript; Y.D. and Q.Z. edited and revised manuscript; Q.Z. and Y.D., approved final version of manuscript.
Corresponding author
Ethics declarations
Conflict of interest
No conflicts of interest, financial or otherwise, are declared
Disclosures
No conflicts of interest, financial or otherwise, are declared by the authors.
Additional information
Action Editor: Steve I. Perlmutter
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, Q., Dai, Y. A modeling study of spinal motoneuron recruitment regulated by ionic channels during fictive locomotion. J Comput Neurosci 48, 409–428 (2020). https://doi.org/10.1007/s10827-020-00763-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10827-020-00763-4