Abstract
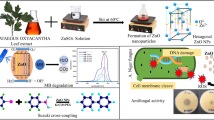
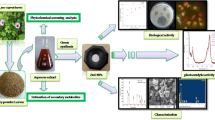
In the study, metabolites of Euphorbia sanguinea were used as benign reducing and stabilizing agents to obtain zinc oxide nanoparticles (ZnO-NPs). The nanoparticles were evaluated as dual agent for photodegradation of Malachite green dye and tyrosinase inhibitior of mushroom tyrosine enzyme. Surface plasmon bands and energy band gaps of the ZnO-NPs were within the range 356–378 nm and 2.72–4.37 eV respectively as obtained from the UV–Vis spectra. SEM/EDS elemental mapping of the nanoparticles gave flower-like shape and even distribution of zinc and oxygen. XRD result revealed crystallographic peaks assigned to hexagonal phase of zinc oxide. The metal oxide nanoparticles were used to achieve 53% percentage degradation of Malachite green dye solution in less than a minute of solar radiation, which increased to 92% in 60 min. A first order kinetics with correlation coefficient R2 of 0.937, rate constant of 0.0084 min−1 and half-life of 82.52 min was established for the photodegradation process. The ZnONPs exhibited good tyrosinase inhibition with IC50 of 49.016 µg/ml. The mode of enzymatic inhibition was competitive with an inhibition constant (Ki) of 0.525 mM using Lineweaver–Burk kinetic model.











Similar content being viewed by others
References
V.K. Vidhu, P. Daizy, Micron 56, 54–62 (2014)
M.C. Mahesh, E.N. Shivayogeeswar, K.R. Kotresh, SN Appl. Sci. 1, 117 (2019). https://doi.org/10.1007/s42452-018-0095-7
J. Huang, L. Lin, D. Sun, H. Chen, D. Yang, Q. Li, Chem. Soc. Rev. 44(17), 6330–6374 (2015)
B. Nagaraj, I. Akber, R.L. Yong, Mater. Lett. 129, 28–30 (2014). https://doi.org/10.1016/j.matlet.2014.05.008
E.E. Elemike, D.C. Onwudiwe, D.F. Ogeleka, J. InorgOrganometPolym. 29, 1798–1806 (2019). https://doi.org/10.1007/s10904-019-01142-y
D. Das, R.K. Mohapatra, P.K. Parhi, A.K. Sarangi, R. Sahu, R.S. Barik, ACS Omega 5, 7716–7721 (2020)
D. Das, A.K. Sarangi, R.K. Mohapatra, P.K. Parhi, A. Mahal, R. Sahu, Md Kudrat-E-Zahan, J. Mol. Liq. 309, 113133 (2020)
D. Das, S. Pattanaik, P.K. Parhi, R.K. Mohapatra, R.K. Jyothi, L. Jin-Young, H.I. Kim, ACS Omega 4(25), 21604–21611 (2019)
A. Routray, P.K. Senapati, M. Padhy, D. Das, R.K. Mohapatra, Int. J. Coal Prep. Util (2019). https://doi.org/10.1080/19392699.2019.1674843
M. Sakir, S.M. Onses, Results Phys. 12, 1133–1141 (2019)
S. Bhakya, S. Muthukrishman, M. Sukumaran, M. Muthukumar, K.T. Senthul, M.V. Rao, J. Bioremed. Biodegr. 6, 1–9 (2015)
A. Ucar, M. Findik, I.H. Gubbuk, N. Kocak, H. Bingol, Mater. Chem. Phys. 196, 21–28 (2017)
S. Kamaljit, A. Sucharita, Crit. Rev. Environ. Sci. Technol. 41(9), 807–878 (2011). https://doi.org/10.1080/10643380903218376
F. Xu, Y. Yuan, D. Wu, M. Zhao, Z. Gao, K. Jiang, Mater. Res. Bull. 48, 2066–2070 (2013)
E.E. Elemike, D.C. Onwudiwe, L. Wei, L. Chaogang, Z. Zhiwei, J. Environ. Chem. Eng. 7, 103190 (2019)
X. Chen, Z. Wu, D. Liu, Z. Gao, Bull. Chem Soc. Ethiop. 31, 4–13 (2017)
T. Mulla, S. Patil, S. Sistla, J. Jadhav, Biochem. Res. Int. (2019). https://doi.org/10.1155/2019/8284968
H.S. Abdillahi, J.F. Finnie, J.V. Staden, J. Ethnopharmacol. 136(3), 496–503 (2011)
P.V. Bruyns, Bolhalia 42(2), 117–245 (2012)
N.I.F. Morah, W.W. Okoi, Int. J. Adv. Sci. Res. 1(1), 08–09 (2016)
J. Sharifi-Rad, S.M. Hoseini-Alfatemi, A. Miri, M. Sharifi-Rad, L. Soufi, M. Sharifi-Rad, W.N. Setzer, M. Hoseini, M. Sharifi-Rad, M. Rokni, Environ. Exp. Biol. 13, 169–175 (2015)
X. Dewanto, K. Wu, K. Adom, R.H. Liu, J. Agric. Food Chem. 50, 3010–3014 (2002)
R. Yuvakkumar, J. Suresh, A.J. Nathanael, M. Sundrarajan, S.I. Hong, Mater. Sci. Eng. 41, 17–27 (2014)
E. Varghese, M. George, Int. J. Adv. Res. Sci. Eng. 4(1), 307–314 (2015)
N. Basavegowda, A. Idhayadhulla, Y.R. Lee, Mater. Sci. Eng. C 43, 58–64 (2014)
M.L. Curri, R. Comparelli, P.D. Cozzoli, G. Mascolo, A. Agostiano, Mater. Sci. Eng. C 23, 285–289 (2003)
D. Suresh, R.M. Shobharani, P.C. Nethravathi, M.A.P. Kumar, H. Nagabhushana, S.C. Sharma, Spectrochim. Acta Part A 141, 128–134 (2015)
R.R. Khoushika, B. Durairaj, Int. J. App. Pharm. 9(5), 116–120 (2017)
A. Datta, C. Patra, H. Bharadwaj, S. Kaur, N. Dimri, R. Khajuria, J. Biotechnol. Biomater. 7, 271 (2017). https://doi.org/10.4172/2155-952X.1000271
M. Gupta, R.S. Tomar, S. Kaushik, K.R. Mishra, D. Sharma, Front. Microbiol. 9, 2030–2043 (2018). https://doi.org/10.3389/fmicb.2018.02030
I. Fatimah, R.Y. Pradita, A. Nurfalinda, Int. Conf. Process Eng. Adv. Mater. Proc. Eng. 148, 43–48 (2016). https://doi.org/10.1016/j.proeng.2016.06.483
D. Gnanasangeetha, S.D. Thambavani, Int. J. Pharm. Sci. Res. 5(7), 2866–2873 (2014)
W. Khan, Z.A. Khan, A.A. Saad, S. Shervani, A. Saleem, A.H. Naqvi, Int. J. Mod. Phys. 22, 630–636 (2013)
M. Romero-Sáez, L. Jaramillo, R. Saravanan, N. Benito, E. Pabón, E. Mosquera, F. Gracia, Express Polym. Lett. 11, 899–909 (2017)
T. Kokila, P.S. Ramesh, D. Geetha, Appl. Nanosci. 5, 911–920 (2015)
Z.E.J. Pérez, R. Mathiyalagan, J. Markus, Y.J. Kim, H.M. Kang, R. Abbai, K.H. Seo, D. Wang, V. Soshnikova, D.C. Yang, Int. J. Nanomed. 12, 709 (2017). https://doi.org/10.2147/IJN.S118373
J. Markus, D. Wang, Y.J. Kim, S. Ahn, R. Mathiyalagan, C. Wang, D.C. Yang, Nanoscale Res. Lett. 12, 46 (2017). https://doi.org/10.1186/s11671-017-1833-2
E.J. Rupa, L. Kaliraj, S. Abid, Y. Deok-Chun, J. Seok-Kyu, Nanomaterials 9, 1692 (2019). https://doi.org/10.3390/nano9121692
M.H. Kahsay, A. Tadesse, D. RamaDevi, N. Belachew, K. Basavaiah, RSC Adv. 9, 36967 (2019)
Z. Tao, X. Yu, J. Liu, L. Yang, S. Yang, J. Alloys Compd. 459, 395–398 (2008)
T. Islam, A. Dominguez, B. Alvarado-Tenorio, R.A. Bernal, M.O. Montes, J.C. Noveron, ACS Omega 4, 6560–6572 (2019). https://doi.org/10.1021/acsomega.9b00023
Z.H. Diao, M.Y. Li, F.Y. Zeng, L. Song, R.L. Qiu, J. Hazard. Mater. 260, 585–592 (2013)
Y. Ma, M. Ni, S. Li, Nanomaterials 8, 428 (2018). https://doi.org/10.3390/nano8060428
Q. Wang, C. Cai, M. Wang, Q. Guo, B. Wang, W. Luo, Y. Wang, C. Zhang, L. Zhou, D. Zhang, Z. Tong, Y. Liu, J. Chen, Materials (2018). https://doi.org/10.3390/ma11061004
N. Kaneva, A. Bojinova, K. Papazova, J. Phys (2015). https://doi.org/10.1088/1742-6596/682/1/012022
Y. Chiu, T.M. Chang, C. Chen, M. Sone, Y. Hsu, Catalysts 9, 430 (2019). https://doi.org/10.3390/catal9050430
K. Singh, S. Arora, Crit. Rev. Environ. Sci. Technol. 41(9), 807–878 (2011). https://doi.org/10.1080/10643380903218376
M.M. Khan, J. Lee, M.H. Cho, J. Ind. Eng. Chem. 20(4), 1584–1590 (2014)
S. Senapati, S.K. Srivastava, S.B. Singh, Nanoscale 4(20), 6604–6612 (2012)
X. Chen, Z. Wu, D. Liu, Z. Gao, Nanoscale Res. Lett. 12, 143 (2017)
K.R. Raajshree, B. Durairaj, Int. J. Appl. Pharm. 9, 20 (2017). https://doi.org/10.22159/ijap.2017v9i5.20847
L. Yung-Sheng, C. Hui-Ju, H. Jung-Ping, L. Pei-Chi, T. Ching-Ru, H. Tzu-Fang, H. Wen-Ying, BioMed Res. Int. (2017). https://doi.org/10.1155/2017/5232680
Z. Ashraf, M. Rafiq, H. Nadeem, M. Hassan, S. Afzal, M. Waseem, K. Afzal, J. Latip, PLoS ONE 12, 5 (2017). https://doi.org/10.1371/journal.pone.0178069
Acknowledgements
The authors are grateful to Alex Ekwueme Federal University Ndufu-Alike, Nigeria for the provision of laboratory space and research equipment.
Author information
Authors and Affiliations
Contributions
The research idea was conceptualized by ACE and DNU; all authors participated in synthesis, applications and manuscript preparations.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ekennia, A.C., Uduagwu, D.N., Nwaji, N.N. et al. Green Synthesis of Biogenic Zinc Oxide Nanoflower as Dual Agent for Photodegradation of an Organic Dye and Tyrosinase Inhibitor. J Inorg Organomet Polym 31, 886–897 (2021). https://doi.org/10.1007/s10904-020-01729-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10904-020-01729-w