Abstract
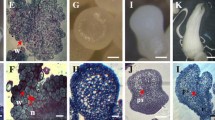
Carbon source plays an important role in the induction of embryogenic callus (EC). Therefore it is necessary to determine the effective carbohydrate for improving this process. In this study, different carbohydrates as sucrose, maltose, and sorbitol with different levels (0–30 g/L) were tested in the callus induction of Pyrus communis. At first, in solid culture, calli were divided in relation to their morphological appearance in EC and non-EC. EC was white or yellow with the granular aspect. The morphological study revealed the highest frequency of EC (28.5%) obtained at 30 g/L sucrose. Biochemical analyses showed EC exhibited both soluble sugar and protein high contents, while the starch was at the lowest level. As well as flow cytometry showed EC were genetically similar to donor explants. With transferring the EC into the liquid culture, then, subculturing calli along with 2 ml cell suspension on solid medium led to the globular embryo.







Similar content being viewed by others
References
Abbasi BH, Ali H, Yücesan B, Saeed S, Rehman K, Khan MA (2016) Evaluation of biochemical markers during somatic embryogenesis in Silybum marianum L. 3 Biotech 6(1):1–8
Acanda Y, Prado MJ, Gonzalez MV, Rey M (2013) Somatic embryogenesis from stamen filaments in grapevine (Vitis vinifera L. cv. Mencía): changes in ploidy level and nuclear DNA content. In Vitro Cell Dev Biol Plant 49:276–284
Antonelli M (1992) Regeneration from almond cotyledon: induction of proembryonal masses. Acta Hortic 300:255–259
Blanc G, Lardet L, Martin A, Jacob JL, Carron MP (2002) Differential carbohydrate metabolism conducts morphogenesis in embryogenic callus of Hevea brasiliensis. J Exp Bot 53:1453–1462
Blanc G, Michaux-ferriere N, Teisson C, Lardet L, Carron MP (1999) Effects of carbohydrate addition on the induction of somatic embryogenesis in Hevea brasiliensis. Plant Cell Tissue Organ Cult 59:103–112
Borji M, Bouamama-Gzara B, Chibani F, Teyssier C, Ammar AB, Mliki A, Zekri S, Ghorbel A (2018) Micromorphology, structural and ultrastructural changes during somatic embryogenesis of a Tunisian oat variety (Avena sativa L. var ‘Meliane’). Plant Cell Tissue Organ Cult 132(2):329–342
Bradford MM (1976) A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Cangahuala-Inocente GC, Silveira V, Caprestano CA, Ducroque JPHJ, Floh EIS, Guerra MP (2009b) Biochemical and histological changes during zygotic embryogenesis in Acca sellowiana. Plant Growth Regul 59:103–115
Cangahuala-Inocente GC, Silveira V, Caprestano CA, Floh EIS, Guerra MP (2014) Dynamics of physiological and biochemical changes during somatic embryogenesis of Acca sellowiana. In Vitro Cell Dev Biol Plant 50:166–175
Cangahuala-Inocente GC, Steiner N, Maldonado SB, Guerra MP (2009a) Patterns of protein and carbohydrate accumulation during somatic embryogenesis of Acca sellowiana. Pesqui Agropecu Bras 44:217–224
Chevreau E, Bell R (2005) Pyrus spp. Pear and Cydonia spp Quince. In: Litz RE (ed) Biotechnology of fruit and nut crops. Biotechnology in agriculture. CABI, Wallingford, p 543
David H, Domon JM, Miannay N, Sulmont G, Dargent R, David A (1992) Evidence for early stages of somatic embryo development in a protoplast-derived cell culture of Prunus avium. Physiol Plant 85:301–307
Druart PH (1999) Somatic embryogenesis in Prunus species. In: Jain SM, Gupta PK, Newton RJ (eds) Somatic embryogenesis in woody plants. Kluwer, Dordrecht, pp 215–235
Ganesan M, Jayabalan N (2005) Carbon source dependent somatic embryogenesis and plant regeneration in cotton, Gossypium hirsutum L. cv. SVPR2 through suspension cultures. Indian J Exp Biol 43(10):921–925
Gronroos L, Von Arnold S, Eriksson T (1989) Callus production and somatic embryogenesis from floral explants of basket willow (Salix viminalis L.). J Plant Physiol 134:558–566
James DJ, Passey AJ, Deeming DC (1984) Adventitious embryogenesis and the in vitro culture of apple seed parts. J Plant Physiol 115:217–229
Li XY, Huang FH, Murphy JB, Gbur EE (1998) Polyethylene glycol and maltose enhance somatic embryo maturation in loblolly pine (Pinus taeda L.). In Vitro Cell Dev Biol Plant 34(1):22–26
Martin AB, Cuadrado Y, Guerra H, Gallego P, Hita O, Martin L, Dorado A, Villalobos N (2000) Differences in the contents of total sugars, reducing sugars, starch and sucrose in embryogenic and non-embryogenic calli from Medicago arborea L. Plant Sci 154:955–960
Meng X, Zhou W (1981) Induction of embryoid and production of plantlets in vitro from endosperm of peach. Acta Agric Univ Peking 4:95–98
Morgan DR, Soltis DE, Robertson KR (1994) Systematic and evolutionary implications of rbcL sequence variation in Rosaceae. Am J Bot 81:890–903
Mujib A, Banerjee S, Ghosh PD (2005) Origin, development and structure of somatic embryos in selected bulbous ornamentals: BAP as inducer. In: Mujib A, Samaj J (eds) Somatic embryogenesis. Springer, Berlin, pp 15–25
Mukherjee A, Debata BK, Mukherjee PS, Malik SK (2001) Morphohistobiochemical characteristics of embryogenic and non-embryogenic callus cultures of Sweet potato (Ipomoea batatas L.). Cytobios 106:113–124
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497
Ng TL, Karim R, Tan YS, Teh HF, Danial AD, Ho LS, Khalid N, Appleton DR, Harikrishna JA (2016) Amino acid and secondary metabolite production in embryogenic and non-embryogenic callus of fingerroot ginger (Boesenbergia rotunda). PLoS ONE 11(6):e156714
Nieves N, Segura-Nieto M, Blanco MA, Sanches A, Gonzalez M, Gonzalez JL, Castillo B (2003) Biochemical characterization of embryogenic and non-embryogenic calluses of sugarcane. In Vitro Cell Dev Biol Plant 39:343–345
Pierik RLM (1997) In vitro culture of higher plants. Kluwer, Dordrecht
Qingrong S, Qingzhong L, Ruihua Z (2003) Somatic embryo genesis from in vitro leaves of pear. Acta Hortic Sin 30:85–86
Reidiboym-Talleux L, Diemer F, Sourdioux M, Chapelain K, Grenier-De march G (1998) Improvement of somatic embryogenesis in wild cherry (Prunus avium). Effect of maltose and ABA supplements. Plant Cell Tiss Organ Cult 55(3):199–209
Saeed T, Shahzad A (2015) High frequency plant regeneration in Indian Siris via cyclic somatic embryogenesis with biochemical, histological and SEM investigations. Ind Crops Prod 76:623–637
Santos Filho PR, Santos BR, Barbosa S, Vieira LR, Freitas NC, Dias DF, Santos MH (2014) Growth curve, biochemical profile and phytochemical analyses in calli obtained from the procambium segments of Bacupari. Braz Arch Biol Technol 57(3):326–333
Shang HH, Liu CL, Zhang CJ, Li FL, Hong WD, Li FG (2009) Histological and ultrastructural observation reveals significant cellular differences between agrobacterium transformed embryogenic and non-embryogenic calli of cotton. J Integr Plant Biol 51:456–465
Swedlund B, Locy RD (1993) Sorbitol as the primary carbon source for the growth of embryogenic callus of maize. Plant Physiol 103:1339–1346
Tang W, Newton RJ (2006) Genome-wide expression analysis of genes involved in somatic embryogenesis. In: Mujib A, Samaj J (eds) Somatic embryogenesis. Springer, Berlin, pp 69–83
Thorpe TA, Stasolla C (2001) Somatic embryogenesis. In: Bhojwani SS, Soh WY (eds) Current trends in the embryology of lingiosperms. Kluwer, Dordrecht, pp 279–336
Visarada KBRS, Sailaja M, Sarma NP (2002) Effect of callus induction media on morphology of embryogenic in rice genotypes. Biol Plant 45:495–502
Von Aderkas P, Teyssier C, Charpentier JP, Gutmann M, Pâques L, Le Metté C, Ader K, Label P, Kong L, Lelu-Walter MA (2015) Effect of light conditions on anatomical and biochemical aspects of somatic and zygotic embryos of hybrid larch (Larix× marschlinsii). Ann Bot 115(4):605–615
Wallin A, Nyman M, Svensson M (1995) Somatic embryogenesis in apple (Malus). In: Jain S, Gupta P, Newton R (eds) Somatic embryogenesis in woody plants. Kluwer, Dordrecht, pp 445–460
Warchoł M, Skrzypek E, Kusibab T, Dubert F (2015) Induction of somatic embryogenesis and biochemical characterization of Cordyline australis (G. Forst.) Endl. ‘Red Star’ callus. Sci Hortic 192:338–345
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
A. Ameri, G.H. Davarynejad, N. Moshtaghi and A. Tehranifar declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Ameri, A., Davarynejad, G.H., Moshtaghi, N. et al. The Role of Carbohydrates on The Induction of Somatic Embryogenesis and The Biochemical State of The Embryogenic Callus in Pyrus communis L. Cv. ‘Dar Gazi’. Erwerbs-Obstbau 62, 411–419 (2020). https://doi.org/10.1007/s10341-020-00518-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10341-020-00518-6