Abstract
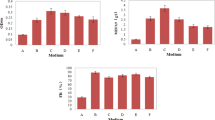
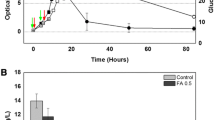
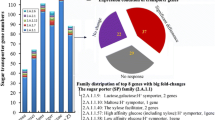
The low yield as bottleneck problem limits the application of microbial flocculant in water treatment. However, genetic information of microbial flocculant-producing strains can guide the regulation of microbial flocculant production, but it remains unknown. Agrobacterium tumefaciens F2 produced polysaccharide-based microbial flocculants in the fermentation medium but none in Luria Bertani medium; hence, the transcriptome was used to analyze the potentially associated genes with the production of microbial flocculants. Glucose, mannose, rhamnose, and galactose are the main sugar monomers, and genes (manA, glmM, manC, rfb genes, exo genes, etc.) with changed expression levels related to sugar monomers metabolism potentially participated in the biosynthesis of polysaccharide-based microbial flocculants. exoC, exoP, and manC were confirmed to participate in the biosynthesis via constructing the mutants F2-dexoC, F2-dexoP, and F2-dmanC. An exoF2 gene cluster was annotated due to the high percentage of matches between the genome sequences of strains F2 and C58, and exo genes in their genome sequences showed the similarity of 86~92%. The hypothetical pathway for the biosynthesis of polysaccharide-based microbial flocculants in strain F2 was proposed, laying the basis for the production yield regulation.
Key points
• An exo F2 gene cluster in the polysaccharide biosynthesis was annotated.
• exoC, exoP, and manC genes participated in the polysaccharide biosynthesis.
• A hypothetical biosynthesis pathway of polysaccharide in flocculant was proposed.

Graphical abstract





Similar content being viewed by others
Change history
04 September 2020
In the original publication of the article, it was published under the title ���Applied microbiology and biotechnology uncovering the biosynthetic pathway of polysaccharide-based microbial flocculant in Agrobacterium tumefaciens F2���.
References
Becker A (2015) Challenges and perspectives in combinatorial assembly of novel exopolysaccharide biosynthesis pathways. Front Microbiol 6:687. https://doi.org/10.3389/fmicb.2015.00687
Cangelosi GA, Hung L, Puvanesarajah V, Stacey G, Ozga DA, Leigh JA, Nester EW (1987) Common loci for Agrobacterium tumefaciens and Rhizobium meliloti exopolysaccharide synthesis and their roles in plant interactions. J Bacteriol 169(5):2086–2091. https://doi.org/10.1128/jb.169.5.2086-2091.1987
Chouly C, Colquhoun IJ, Jodelet A (1995) NMR studies of succinoglycan repeating-unit octasaccharides from Rhizobium meliloti and Agrobacterium radiobacter. Int J Biol Macromol 17(6):357–363. https://doi.org/10.1016/0141-8130(96)81846-0
Evans LR, Linker A, Impallomeni G (2000) Structure of succinoglycan from an infectious strain of Agrobacterium radiobacter. Int J Biol Macromol 27:319–326. https://doi.org/10.1016/s0141-8130(00)00131-8
Ferreira AS, Silva IN, Oliveira VH, Cunha R, Moreira LM (2011) Insights into the role of extracellular polysaccharides in Burkholderia adaptation to different environments. Front Cell Infect Microbiol 1:16. https://doi.org/10.3389/fcimb.2011.00016
Freitas F, Alves VD, Reis MA (2011) Advances in bacterial exopolysaccharides: from production to biotechnological applications. Trends Biotechnol 29(8):388–398. https://doi.org/10.1016/j.tibtech.2011.03.008
Giraud MF, Naismith JH (2000) The rhamnose pathway. Curr Opin Struct Biol 10(6):687–696. https://doi.org/10.1016/S0959-440X(00)00145-7
Glucksmann MA, Reuber TL, Walker GC (1993) Genes needed for the modification, polymerization, export, and processing of succinoglycan by Rhizobium meliloti: a model for succinoglycan biosynthesis. J Bacteriol 175(21):7045–7055. https://doi.org/10.1111/j.1365-2672.1993.tb02806.x
Guan NZ, Du B, Li JH, Shin HD, Chen RR, Du GC, Chen J, Liu L (2018) Comparative genomics and transcriptomics analysis-guided metabolic engineering of Propionibacterium acidipropionici for improved propionic acid production. Biotechnol Bioeng 115(2):483–494. https://doi.org/10.1002/bit.26478
Janczarek M (2011) Environmental signals and regulatory pathways that influence exopolysaccharide production in Rhizobia. Int J Mol Sci 12:7898–7933. https://doi.org/10.3390/ijms12117898
Kovach ME, Elzer PH, Hill DS, Robertson GT, Farris MA, Roop RI, Peterson KM (1995) Four new derivatives of the broad-host-range cloning vector pBBR1MCS, carrying different antibiotic-resistance cassettes. Gene 166:175–176. https://doi.org/10.1016/0378-1119(95)00584-1
Li A, Geng JN, Cui D, Shu C, Zhang S, Yang JX, Xing J, Wang JN, Ma F, Hu SN (2011) Genome sequence of Agrobacterium tumefaciens strain F2, a bioflocculant-producing bacterium. J Bacteriol 193(19):5531. https://doi.org/10.1128/JB.05690-11
Quandt J, Hynes MF (1993) Versatile suicide vectors which allow direct selection for gene replacement in Gram-negative bacteria. Gene 127:15–21. https://doi.org/10.1016/0378-1119(93)90611-6
Rehm BH (2010) Bacterial polymers: biosynthesis, modifications and applications. Nat Rev Microbiol 8(8):578–592. https://doi.org/10.1038/nrmicro2354
Reinhold BB, Chan SY, Reuber TL, Marra A, Walker GC, Reinhold VN (1994) Detailed structural characterization of succinoglycan, the major exopolysaccharide of Rhizobium meliloti Rm1021. J Bacteriol 176(7):1997–2002. https://doi.org/10.1128/jb.176.7.1997-2002.1994
Reuber TL, Walker GC (1993) Biosynthesis of succinoglycan, a symbiotically important exopolysaccharide of Rhizobium meliloti. Cell 74:269–280. https://doi.org/10.1016/0092-8674(93)90418-P
Ruffing A, Chen RR (2006) Metabolic engineering of microbes for oligosaccharide and polysaccharide synthesis. Microb Cell Factories 5:25. https://doi.org/10.1186/1475-2859-5-25
Salehizadeh H, Yan N, Farnood R (2018) Recent advances in polysaccharide bio-based flocculants. Biotechnol Adv 36:92–119. https://doi.org/10.1016/j.biotechadv.2017.10.002
Schmid J, Sieber V, Rehm B (2015) Bacterial exopolysaccharides: biosynthesis pathways and engineering strategies. Front Microbiol 6. https://doi.org/10.3389/fmicb.2015.00496
Skorupska A, Janczarek M, Marczak M, Mazur A, Król J (2006) Rhizobial exopolysaccharides: genetic control and symbiotic functions. Microb Cell Factories 5:7. https://doi.org/10.1186/1475-2859-5-7
Stredansky M, Conti E (1999) Succinoglycan production by solid-state fermentation with Agrobacterium tumefaciens. Appl Microbiol Biotechnol 52:332–337. https://doi.org/10.1007/s002530051528
Stredansky M, Conti E, Bertocchi C, Matulova M, Zanetti F (1998) Succinoglycan production by Agrobacterium tumefaciens. J Ferment Bioeng 85(4):398–403. https://doi.org/10.1016/s0922-338x(98)80083-4
Wang LX, Wang Y, Pellock B, Walker GC (1999) Structural characterization of the symbiotically important low-molecular-weight succinoglycan of Sinorhizobium meliloti. J Bacteriol 181(21):6788–6796. https://doi.org/10.1038/228178a0
Wood DW, Setubal JC, Kaul R, Monks DE, Kitajima JP, Okura VK, Zhou Y, Chen L, Wood GE, Almeida NF Jr, Woo L, Chen Y, Paulsen IT, Eisen JA, Karp PD, Bovee D Sr, Chapman P, Clendenning J, Deatherage G, Gillet W, Grant C, Kutyavin T, Levy R, Li MJ, McClelland E, Palmieri A, Raymond C, Rouse G, Saenphimmachak C, Wu Z, Romero P, Gordon D, Zhang S, Yoo H, Tao Y, Biddle P, Jung M, Krespan W, Perry M, Gordon-Kamm B, Liao L, Kim S, Hendrick C, Zhao ZY, Dolan M, Chumley F, Tingey SV, Tomb JF, Gordon MP, Olson MV, Nester EW (2001) The genome of the natural genetic engineer Agrobacterium tumefaciens C58. Science 294(5550):2317–2323. https://doi.org/10.1126/science.1066804
Wu D, Li A, Yang JX, Ma F, Chen H, Pi SS, Wei W (2015) N-3-Oxo-octanoyl-homoserine lactone as a promotor to improve the microbial flocculant production by an exopolysaccharide bioflocculant-producing bacterium Agrobacterium tumefaciens F2. RSC Adv 5:89531–89538. https://doi.org/10.1039/c5ra15657b
Wu D, Li A, Ma F, Yang JX, Xie YT (2016) Genetic control and regulatory mechanisms of succinoglycan and curdlan biosynthesis in genus Agrobacterium. Appl Microbiol Biotechnol 100:6183–6192. https://doi.org/10.1007/s00253-016-7650-1
Yang R, Li HJ, Huang M, Yang H, Li AM (2016) A review on chitosan-based flocculants and their applications in water treatment. Water Res 95:59–89. https://doi.org/10.1016/j.watres.2016.02.068
Yu WC, Chen Z, Shen L, Wang YP, Li QB, Yan S, Zhong CJ, He N (2015) Proteomic profiling of Bacillus licheniformis reveals a stress response mechanism in the synthesis of extracellular polymeric flocculants. Biotechnol Bioeng 113(4):797–806. https://doi.org/10.1002/bit.25838
Yu WC, Chen Z, Ye H, Liu PZ, Li ZP, Wang YP, Li QB, Yan S, Zhong CJ, He N (2017) Effect of glucose on poly-γ-glutamic acid metabolism in Bacillus licheniformis. Microb Cell Factories 16:22. https://doi.org/10.1186/s12934-017-0642-8
Zevenhuizen LPTM (1997) Succinoglycan and galactoglucan. Carbohydr Polym 33(2–3):139–144. https://doi.org/10.1016/s0144-8617(97)00054-4
Zlosnik JEA, Hird TJ, Fraenkel MC, Moreira LM, Henry DA, Speert DP (2008) Differential mucoid exopolysaccharide production by members of the Burkholderia cepacia complex. J Clin Microbiol 46(4):1470–1473. https://doi.org/10.1128/JCM.02273-07
Acknowledgments
This work was supported by the National Natural Science Foundation of China (no. 51578179).
Author contribution statement
A. Li and F. Ma conceived and designed the research. S.S. Pi and L. Feng conducted the experiments. J.G. Qiu and H.P. Zhao contributed new reagents or analytical tools. S.S. Pi and D. Wu analyzed the data. S.S. Pi wrote the manuscript. All authors read and approved the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 698 kb)
Rights and permissions
About this article
Cite this article
Pi, S., Qiu, J., Li, A. et al. Applied microbiology and biotechnology uncovering the biosynthetic pathway of polysaccharide-based microbial flocculant in Agrobacterium tumefaciens F2. Appl Microbiol Biotechnol 104, 8479–8488 (2020). https://doi.org/10.1007/s00253-020-10850-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-020-10850-2