Abstract
Purpose
To determine whether texture analysis of contrast-enhanced computed tomography (CECT) and apparent diffusion coefficient (ADC) maps could predict tumor grade (G1 vs G2–3) in patients with pancreatic neuroendocrine tumor (PNET).
Materials and methods
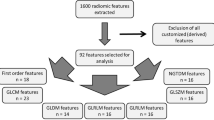
Thirty-three PNETs (22 G1 and 11 G2–3) were retrospectively reviewed. Fifty features were individually extracted from the arterial and portal venous phases of CECT and ADC maps by two radiologists. Diagnostic performance was assessed by receiver operating characteristic curves while inter-observer agreement was determined by calculating intraclass correlation coefficients (ICCs).
Results
G2–G3 tumors were significantly larger than G1. Seventeen features significantly differed among the two readers on univariate analysis, with ICCs > 0.6; the largest area under the curve (AUC) for features of each CECT phase and ADC map was log-sigma 1.0 joint-energy = 0.855 for the arterial phase, log-sigma 1.5 kurtosis = 0.860 for the portal venous phase, and log-sigma 1.0 correlation = 0.847 for the ADC map. The log-sigma 1.5 kurtosis of the portal venous phase showed the largest AUC in the CECT and ADC map, and its sensitivity, specificity, and accuracy were 95.5%, 72.7%, and 87.9%, respectively.
Conclusion
Texture analysis may aid in differentiating between G1 and G2–3 PNET.



Similar content being viewed by others
References
Ansari NA, Ramalho M, Semelka RC, Buonocore V, Gigli S, Maccioni F. Role of magnetic resonance imaging in the detection and characterization of solid pancreatic nodules: an update. World J Radiol. 2015;7:361–74.
Lewis RB, Lattin GE Jr, Paal E. Pancreatic endocrine tumors: radiologic-clinicopathologic correlation. Radiographics. 2010;30:1445–644.
Lloyd RV, Osamura RY, Klöppel G, Rosai J. WHO classification of tumours of endocrine organs. 4th ed. Lyon: International Agency for Research on Cancer; 2017. p. 209–240.
Singhi AD, Klimstra DS. Well-differentiated pancreatic neuroendocrine tumours (PanNETs) and poorly differentiated pancreatic neuroendocrine carcinomas (PanNECs): concepts, issues and a practical diagnostic approach to high-grade (G3) cases. Histopathology. 2018;72:168–77.
Amin MB, Edge SB, Greene F, Byrd DR, Brookland RK, Washington MK, et al. AJCC cancer staging manual. 8th ed. New York: Springer; 2017.
Plöckinger U, Rindi G, Arnold R, Eriksson B, Krenning EP, de Herder WW, et al. European Neuroendocrine Tumour Society Guidelines for the diagnosis and treatment of neuroendocrine gastrointestinal tumours: a consensus statement on behalf of the European Neuroendocrine Tumour Society (ENETS). Neuroendocrinology. 2004;80:394–424.
Sahani DV, Bonaffini PA, Fernández-Del Castillo C, Blake MA. Gastroenteropancreatic neuroenodocrine tumors: role of imaging in diagnosis and management. Radiology. 2013;266:38–61.
Yang G, Ji M, Chen J, Chen R, Chen Y, Fu D, et al. Surgery management for sporadic small (≤2 cm), non-functioning pancreatic neuroendocrine tumors: a consensus statement by the Chinese Study Group for Neuroendocrine Tumors (CSNET). Int J Oncol. 2017;50:567–74.
Smith JK, Ng SC, Hill JS, Simmmons JP, Arous EJ, Shah SA, et al. Complications after pancreatectomy for neuroendocrine tumors: a national study. J Surg Res. 2010;163:63–8.
Sallinen VJ, Le Large TYS, Tieftrunk E, Galeev S, Kovalenko Z, Haugvik SP, et al. Prognosis of sporadic resected small (≤2 cm) nonfunctional pancreatic neuroendocrine tumors—a multi-institutional study. HPB. 2018;20:251–9.
Rodallec M, Vilgrain V, Couvelard A, Rufat P, O'Toole D, Sauvanet A, et al. Endocrine pancreatic tumours and helical CT: contrast enhancement is correlated with microvascular density, histoprognostic factors and survival. Pancreatology. 2006;6:77–85.
Manfredi R, Bonatti M, Mantovani W, Graziani R, Segala D, Capelli P, et al. Non-hyperfunctioning neuroendocrine tumors of the pancreas: MR imaging appearance and correlation with their biological behaviour. Eur Radiol. 2013;23:3029–39.
Lotfalizadeh E, Ronot M, Wagner M, Cros J, Couvelard A, Vullierme MP, et al. Prediction of pancreatic neuroendocrine tumour grade with MR imaging features: added value of diffusion-weighted imaging. Eur Radiol. 2016;27:1748–59.
Wang Y, Chen ZE, Yaghmai V, Nikolaidis P, McCarthy RJ, Merrick L, et al. Diffusion-weighted MR imaging in pancreatic endocrine tumors correlated with histopathologic characteristics. J Magn Reson Imaging. 2011;33:1071–9.
Toshima F, Inoue D, Komori T, Yoshida K, Yoneda N, Minami T, et al. Is the combination of MR and CT findings useful in determining the tumor grade of pancreatic neuroendocrine tumors? Jpn J Radiol. 2017;35:242–53.
Park HJ, Kim HJ, Kim KW, Kim SY, Choi SH, You M-W, et al. Comparison between neuroendocrine carcinomas and well-differentiated neuroendocrine tumors of the pancreas using dynamic enhanced CT. Eur Radiol. 2020. https://doi.org/10.1007/s00330-020-06867-w.
Lubner MG, Smith AD, Sandrasegaran K, Sahani DV, Perry JP. CT texture analysis: definitions, applications, biologic correlates, and challenges. Radiographics. 2017;37:1483–503.
Yun G, Kim YH, Lee YJ, Kim B, Hwang J-H, Choi DJ. Tumor heterogeneity of pancreas head cancer assessed by CT texture analysis: association with survival outcomes after curative resection. Sci Rep. 2018;8:7226.
Canellas R, Burk KS, Parakh A, Sahani D. Prediction of pancreatic neuroendocrine tumor grade based on CT features and texture analysis. AJR Am J Roentgenol. 2018;210:341–6.
Choi TW, Kim JH, Yu MH, Park SJ, Han JK. Pancreatic neuroendocrine tumor: prediction of the tumor grade using CT findings and computerized texture analysis. Acta Radiol. 2018;59:383–92.
Pereira JA, Rosado E, Bali M, Metens T, Chao SL. Pancreatic neuroendocrine tumors: correlation between histogram analysis of apparent diffusion coefficient maps and tumor grade. Abdom Imaging. 2015;40:3122–8.
De Robertis R, Maris B, Cardobi N, Martini PT, Gobbo S, Capelli P, et al. Can histogram analysis of MR image predict aggressiveness in pancreatic neuroendocrine tumors? Eur Radiol. 2018;28:2582–91.
Guo CG, Ren S, Chen X, Wang QD, Xiao WB, Zhang JF, et al. Pancreatic neuroendocrine tumor: prediction of the tumor grade using magnetic resonance imaging findings and texture analysis with 3-T magnetic resonance. Cancer Manag Res. 2019;11:1933–44.
D'Onofrio M, Ciaravino V, Cardobi N, De Robertis R, Cingarlini S, Landoni L, et al. CT enhancement and 3D texture analysis of pancreatic neuroendocrine neoplasms. Sci Rep. 2019;9:2176.
Liu Y, Liu S, Qu F, Qian L, Cheng R, Ye Z. Tumor heterogeneity assessed by texture analysis on contrast-enhanced CT in lung adenocarcinoma: association with pathologic grade. Oncotarget. 2017;8:53664–74.
Al-Hawary MM, Francis IR, Chari ST, Fishman EK, Hough DM, Lu DS, et al. Pancreatic ductal adenocarcinoma radiology reporting template: consensus statement of the Society of Abdominal Radiology and the American Pancreatic Association. Radiology. 2014;270:248–60.
Ducreux M, Cuhna AS, Caramella C, Hollebecque A, Burtin P, Goéré D, et al. Cancer of the pancreas: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2015;26(Suppl 5):v56–v68.
Acknowledgements
The authors would like to thank Soumei Nozu (Siemens Japan K. K., Customer Service Division) for providing information about detailed MR parameters.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Ohki, K., Igarashi, T., Ashida, H. et al. Usefulness of texture analysis for grading pancreatic neuroendocrine tumors on contrast-enhanced computed tomography and apparent diffusion coefficient maps. Jpn J Radiol 39, 66–75 (2021). https://doi.org/10.1007/s11604-020-01038-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11604-020-01038-9