Abstract
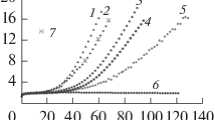
Based on the reference data on the dynamic viscosity of the melts of halides of the elements of Groups 1 and 11, the approximating dependences in the form of cluster-associate and Frenkel’s models were constructed at various temperatures. In the framework of the cluster-associate viscosity model, which is being developed by the authors, entities in the form of virtual clusters and associates determine the melt viscosity and serve as the flow units to that the energy of fluid motion is applied. The Frenkel’s model allows us to estimate the activation energy of fluidity. Calculations show that there is a linear correlation between this energy and the cluster association degree obtained in the framework of the cluster-associate model, and the proportionality coefficient has the meaning of the activation energy per a cluster. This energy does not exceed the van der Waals energy of an unsaturated intermolecular bond characterizing the interaction of particles in a liquid.
Similar content being viewed by others
References
I. V. Melikhov, Fiziko-khimicheskaya evolutsiya tverdogo veshchestva [Physicochemical Evolution of the Solid], BINOM. Laboratoriya Znanii, Moscow, 2006, 309 pp. (in Russian).
G. N. Elansky, D. G. Elansky, Stroenie i svoistva metallicheskikh rasplavov [The Structure and Properties of Metal Melts], Izd-vo MGVMI, Moscow, 2006, 228 pp. (in Russian).
B. A. Baum, Metallicheskie zhidkosti [Metal Fluids], Nauka, Moscow, 1979, 120 pp. (in Russian).
B. A. Baum, G. V. Tyagunov, E. E. Baryshev, E. S. Tsepelev, Fundamentalnye issledovaniya fiziko-khimii metallicheskikh rasplavov [Basic Research of the Physical Chemistry of Metal Melts], Akademkniga, Moscow, 2002, 469 pp. (in Russian).
I. V. Gavrilin, Plavlenie i kristallizatsiya metallov i splavov [Melting and Crystallization of Metals and Alloys], Izd-vo VGU, Vladimir, 2000, 260 pp. (in Russian).
A. M. Zborschik, Teoreticheskie osnovy metallurgicheskogo proizvodstva [The Theoretical Foundations of Metallurgical Production], Izd-vo DonNTU, Donetsk, 2008, 189 pp. (in Russian).
Fundamental’nye issledovaniya fizikokhimii metallicheskikh rasplavov [Basic Research of the Physical Chemistry of Metal Melts], Ed N. P. Lyakishev, Akademkniga, Moscow, 2002, 467 pp. (in Russian).
A. A. Cherepakhin, I. I. Koltunov, V. A. Kuznetsov, Materialovedenie [Materials Science], Knorus, Moscow, 2009, 235 pp. (in Russian).
A. Maksimov, Novaya interpretatsiya v termodinamike [New Interpretation in Thermodynamics], Lambert, Academic Publishing, Saarbrucken, 2014, 76 pp. (in Russian).
A. D. Zimon, Fizicheskaya khimiya [Physical Chemistry], Krasand, Moscow, 2015, 318 pp. (in Russian).
V. V. Budanov, A. I. Maksimov, Khimicheskaya termodinamika [Chemical Thermodynamics], Lan’, Saint Petersburg, 2016, 320 pp. (in Russian).
Yu. S. Efremov, Statisticheskaya fizika i termodinamika [Statistical Physics and Thermodynamics], Yurait, Moscow, 2017, 209 pp. (in Russian).
D. P. Zarubin, Fizicheskaya khimiya [Physical Chemistry], Infra-M, Moscow, 2017, 476 pp. (in Russian).
P. Schmidt, R. Schafer, Methods in Physical Chemistry, John Willey & Sons Limited, Manchester, 2017, 370 pp.
P. W. Atkins, J. de Paula, Elements of Physical Chemistry, W. H. Freeman and Company, Oxford, 2016, 656 pp.
T. Yazachev, Physical Chemistry, Lambert, Academic Publishing, Saarbrucken, 2014, 148 pp.
P. M. S. Monk, Physical Chemistry: Understanding our Chemical World, John Wiley & Sons Ltd, Manchester, 2004, 619 pp.
D. W. Rogers, Concise Physical Chemistry, John Wiley & Sons, Brooklyn, 2013, 405 pp.
M. A. Leontovich, Vvedenie v termodinamiku. Statisticheskaya fizika [Introduction to Thermodynamics. Statistical Physics], Vysshaya Shkola, Moscow, 1983, p. 211 (in Russian).
V. P. Malyshev, A. M. Makasheva, Yu. S. Krasikova, Vestn. Karagandinskogo Un-ta, Ser. Fizika [Vestn. Karaganda Univ., Ser. Physics], 2018, 3, 42 (in Russian).
V. P. Malyshev, N. S. Bekturganov, A. M. Turdukozhaeva, Vyazkost’, tekuchest’ i plotnost’ veschestv kak mera ikh khaotizatsii [Viscosity, Fluidity, and Density of Substances as a Measure of their Randomization], Nauchnyi Mir, Moscow, 2012, 288 pp. (in Russian).
V. P. Malyshev, M. Zh. Tolymbekov, A. M. Turdukozhaeva, A. S. Kazhikenova, A. M. Akulov, Rasplavy [Melts], 2010, 6, 43 (in Russian).
V. P. Malyshev, A. M. Turdukozhaeva, Rasplavy [Melts], 2011, 6, 72 (in Russian).
V. P. Malyshev, A. M. Makasheva, Tsvetnye Metally [Non-Ferrous Metals], 2018, 10, 6 (in Russian).
R. E. Dickerson, H. B. Gray, G. P. Haight, Jr., Chemical Principles, the Benjamin/Cummings Publishing Co., Inc., California, 1979, 540 pp.
V. A. Rabinovitch, Z. Ya. Khazin, Kratkii khimicheskii spravochnik [Brief Chemical Reference Book], Khimiya, Leningrad, 1978, 316 pp. (in Russian).
A. Ya. Malkin, S. A. Patlazhan, V. G. Kulichikhin, Russ. Chem. Rev., 2019, 88, 319.
Author information
Authors and Affiliations
Corresponding author
Additional information
Based on the materials of the XXI Mendeleev Congress on General and Applied Chemistry (September 9–13, 2019, St. Petersburg, Russia).
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 7, pp. 1296–1305, July, 2020.
Rights and permissions
About this article
Cite this article
Malyshev, V.P., Makasheva, A.M. Relationship between the cluster theory of liquids and the Frenkel—Andrade viscosity model. Russ Chem Bull 69, 1296–1305 (2020). https://doi.org/10.1007/s11172-020-2901-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-020-2901-9