Abstract
Main conclusion
Overexpression of the tobacco lipid transfer protein (NtLTP1) gene in transgenic orange mint resulted in enhanced accumulation of monoterpenes in the cavity of head cells of glandular trichomes, which resulted in enhanced emission of monoterpenes from transgenic orange mints.
Abstract
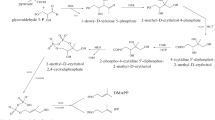
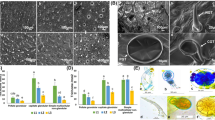
Plants in the genus Mentha (Lamiaceae) produce volatile oils that accumulate in peltate glandular trichomes in the aerial parts of plants. A lipid transfer protein (NtLTP1) in tobacco showed glandular trichome-specific expression and supported the secretion of diterpenoid lipids from head cells of glandular trichomes (Choi et al., Plant J 70:480–491,2012). Here, we constructed transgenic orange mint (Mentha × piperita f. citrata) overexpressing the tobacco NtLTP1 gene via Agrobacterium-mediated transformation. Transgenic lines of orange mint overexpressing NtLTP1 were confirmed by genomic PCR and RT-PCR. Immunoblotting analysis using an NtLTP1 polyclonal antibody showed clear dark spots at the position of the lipid exudates from tobacco glandular trichomes and the squeezed out lipids from the glandular trichomes of transgenic orange mint. Heads of glandular trichomes in transgenic plants overexpressing the NtLTP1 gene showed a larger diameter than those of the wild-type control. The enhanced size of trichome heads in transgenic orange mint was confirmed by scanning electron microscopy. Volatile components were extracted from wild-type and transgenic orange mint by solid-phase microextraction (SPME) and analyzed by headspace–gas chromatography–mass spectrometry (HS/GC/MS). Linalyl acetate was the most abundant component among the eleven identified monoterpenes in the volatile compounds extracted from both the wild-type and transgenic lines of orange mint. Overexpression of NtLTP1 in transgenic orange mint plants resulted in enhanced emission of volatile monoterpenoids compared with that of volatile monoterpenoids in the wild-type control plants.








Similar content being viewed by others
Abbreviations
- HS/SPME:
-
Headspace/solid-phase microextraction
- HS–GC–MS:
-
Headspace–gas chromatography–mass spectrometry
- LTP:
-
Lipid transfer protein
- PCR:
-
Polymerase chain reaction
- UHR-SEM:
-
Ultrahigh-resolution scanning electron microscopy
References
Adhikari PB, Han JY, Ahn CH, Choi YE (2019) Lipid transfer proteins (AaLTP3 and AaLTP4) are involved in sesquiterpene lactone secretion from glandular trichomes in Artemisia annua. Plant and Cell Physiol 60:2826–2836
Cassab GI, Varner JE (1989) Tissue printing on nitrocellulose paper: a new method for immunolocalization of proteins, localization of enzyme activities and anatomical analysis. Cell Biol Int Rep 13:147–152
Chiapparino A, Maeda K, Turei D, Saez-Rodriguez J, Gavin AC (2016) The orchestra of lipid-transfer proteins at the crossroads between metabolism and signaling. Prog Lipid Res 61:30–39
Choi YE, Lim S, Kim HJ, Han JY, Lee MH, Yang Y, Kim JA, Kim YS (2012) Tobacco NtLTP1, a glandular-specific lipid transfer protein, is required for lipid secretion from glandular trichomes. Plant J 70:480–491
Crouzet J, Roland J, Peeters E, Trombik T, Ducos E, Nader J, Boutry M (2013) NtPDR1, a plasma membrane ABC transporter from Nicotiana tabacum, is involved in diterpene transport. Plant Mol Biol 82:181–192
Diemer F, Caissard JC, Moja S, Calchat JC, Jullien F (2001) Altered monoterpene composition in transgenic mint following the introduction of 4S-limonene synthase. Plant Physiol Biochem 39:603–614
Echeverría E (2000) Vesicle-mediated solute transport between the vacuole and the plasma membrane. Plant Physiol 123:1217–1226
Edstam MM, Viitanen L, Salminen TA, Edqvist J (2011) Evolutionary history of the non- specific lipid transfer proteins. Mol Plant 4:947–964
Glenn W, Turner GW, Gershenzon J, Croteau RB (2000) Development of peltate glandular trichomes of peppermint. Plant Physiol 124:665–680
Gochev V, Wlcek K, Buchbauer G, Stoyanova A, Dobreva A, Schmidt E, Jirovetz L (2008) Comparative evaluation of antimicrobial activity and composition of rose oils from various geographic origins, in particular Bulgarian rose oil. Nat Prod Commun 3:1063–1068
Greenspan P, Mayer EP, Fowler SD (1985) Nile Red: a selective fluorescent stain for intracellular lipids droplets. J Cell Biol 100:965–973
Harada E, Kim JA, Meyer AJ, Hell R, Clemens S, Choi YE (2010) Expression profiling of tobacco leaf trichomes identifies genes for biotic and abiotic stresses. Plant Cell Physiol 51:1627–1637
Jirovetz L, Buchbauer G, Schmidt E (2007) Purity, antimicrobial activities and olfactoric evaluations of geraniol/nerol and various of their derivatives. J Essent Oil Res 19:288–291
Kader JC (1996) Lipid-transfer proteins in plants. Annu Rev Plant Physiol Plant Mol Biol 47:627–654
Khayyat S, Sameeh M (2018) Bioactive epoxides and hydroperoxides derived from naturally monoterpene geranyl acetate. Saudi Pharm J 26:14–19
Kinoshita T, Shimazaki KI (1999) Blue light activates the plasma membrane H(+)-ATPase by phosphorylation of the C-terminus in stomatal guard cells. EMBO J 18:5548–5558
Krasnyansky S, May RA, Loskutov A, Ball TM, Sink KC (1999) Transformation of the limonene synthase gene into peppermint (Mentha piperita L.) and preliminary studies on the essential oil profiles of single transgenic plants. Theor Appl Genet 99:676–682
Lange BM, Wildung MR, Stauber EJ, Sanchez C, Pouchnik D, Croteau R (2000) Probing essential oil biosynthesis and secretion by functional evaluation of expressed sequence tags from mint glandular trichomes. Proc Natl Acad Sci USA 97:2934–2939
Lange BM, Mahmoud SS, Wildung MR, Turner GW, Davis EM, Lange I, Baker RC, Boydston RA, Croteau RB (2011) Improving peppermint essential oil yield and composition by metabolic engineering. Proc Natl Acad Sci USA 108:16944–16949
Lapczynski A, Foxenberg RJ, Bhatia SP, Letizia CS, Api AM (2008) Fragrance material review on nerol. Food Chem Toxicol 46:S241–S244
Lev S (2010) Non-vesicular lipid transport by lipid-transfer proteins and beyond. Nat Rev Mol Cell Biol 11:739–750
Liu F, Zhang X, Lu C, Zeng X, Li Y, Fu D, Wu G (2015) Non-specific lipid transfer proteins in plants: presenting new advances and an integrated functional analysis. J Exp Bot 66:5663–5681
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2 -ΔΔCT method. Methods 25:402–408
Mahmoud SS, Croteau RB (2001) Metabolic engineering of essential oil yield and composition in mint by altering expression of deoxyxylulose phosphate reductoisomerase and menthofuran synthase. Proc Natl Acad Sci USA 98:8915–8920
Mahmoud SS, Williams M, Croteau R (2004) Cosuppression of limonene-3-hydroxylase in peppermint promotes accumulation of limonene in the essential oil. Phytochemistry 65:547–554
McCaskill DG, Gershenzon J, Croteau R (1992) Morphology and monoterpene biosynthetic capabilities of secretory cell clusters isolated from glandular trichomes of peppermint (Mentha piperita L.). Planta 187:445–454
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15:473–497
Murray MJ, Lincoln DE (1970) The genetic basis of acyclic oil constituents in Mentha citrate Ehrh. Genetics 65:457–471
Robards AW, Stark M (1988) Nectar secretion in Abutilon: a new model. Protoplasma 142:79–91
Rohloff J (1999) Monoterpene composition of essential oil from peppermint (Mentha x piperita L.) with regard to leaf position using solid-phase microextraction and gas chromatography/mass spectrometry analysis. J Agric Food Chem 47:3782–3786
Sallaud C, Giacalone C, Töpfer R, Goepfert S, Bakaher N, Rösti S, Tissier A (2012) Characterization of two genes for the biosynthesis of the labdane diterpene Z-abienol in tobacco (Nicotiana tabacum) glandular trichomes. Plant J 72:1–17
Schuurink R, Tissier A (2020) Glandular trichomes: micro-organs with model status? New Phytol 225:2251–2266
Singh P, Pandey AK (2018) Prospective of essential oils of the genus Mentha as biopesticides. Front Plant Sci 9:1295
Taherpour AA, Khaef S, Yari A, Nikeafshar S, Fathi M, Ghambari S (2017) Chemical composition analysis of the essential oil of Mentha piperita L. from Kermanshah, Iran by hydrodistillation and HS/SPME methods. J Anal Sci Technol 8:11
Tian J, Zeng X, Zeng H, Feng Z, Miao X, Peng X (2013) Investigations on the antifungal effect of nerol against Aspergillus flavus causing food spoilage. Sci World J 2013:230795
Tissier A (2012) Glandular trichomes: what comes after expressed sequence tags? Plant J 70:51–68
Tissier A, Morgan JA, Dudareva N (2017) Plant volatiles: going ‘in’ but not ‘out’ of trichome cavities. Trends Plant Sci 22:930–938
Wagner GJ, Wang E, Shepherd RW (2004) New approaches for studying and exploiting an old protuberance, the plant trichome. Ann Bot 93:3–11
Wang B, Kashkooli AB, Sallets A, Ting HM, de Ruijter NCA, Olofsson L, Brodelius P, Pottier M, Boutry M, Bouwmeester H, van der Krol AR (2016) Transient production of artemisinin in Nicotiana benthamiana is boosted by a specific lipid transfer protein from A. annua. Metab Eng 38:159–169
Yu F, De Luca V (2013) ATP-binding cassette transporter controls leaf surface secretion of anticancer drug components in Catharanthus roseus. Proc Natl Acad Sci USA 110:15830–15835
Acknowledgements
This work was supported by the Rural Development Administration, Republic of Korea [Next-Generation Bio-Green 21 Program (PJ01344401 and PJ01369103)], and Kangwon National University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Anastasios Melis.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
425_2020_3447_MOESM1_ESM.docx
Supplementary file1 Supplemental Figure S1. DNA sequences of T-DNA regions of the binary vectors used in this experiment. (DOCX 19 kb)
Rights and permissions
About this article
Cite this article
Hwang, HS., Adhikari, P.B., Jo, HJ. et al. Enhanced monoterpene emission in transgenic orange mint (Mentha × piperita f. citrata) overexpressing a tobacco lipid transfer protein (NtLTP1). Planta 252, 44 (2020). https://doi.org/10.1007/s00425-020-03447-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00425-020-03447-6