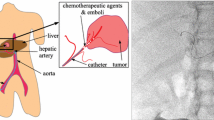
Abstract
Purpose
The fusion of pre/intraoperative images may improve catheter manipulation during radioembolization (RE) interventions by adding relevant information. The objective of this work is to propose and evaluate the performance of a RE guidance strategy relying on structure-driven intensity-based registration between preoperative CTA and intraoperative X-ray images.
Methods
The navigation strategy is decomposed into three image fusion steps, supporting the catheter navigation from the femoral artery till reaching the injection site (IS). During the pretreatment assessment intervention, the aorta and the origins of its side branches are projected on the intraoperative 2D fluoroscopy following a 3D/2D bone-based registration process, to assist the celiac trunk access. Subsequently, a similar approach consisting in projecting the hepatic vasculature on intraoperative DSA through 3D/2D vessel-based registration is performed to assist the IS location. Lastly, the selected IS is reproduced during the treatment intervention by employing 2D/2D image-based registration between pretreatment and treatment fluoroscopic images.
Results
The three fusion steps were independently evaluated on subsets of 20, 19 and 5 patient cases, respectively. Best results were obtained with gradient difference as similarity measure and with a delimited preoperative vascular structure for vessel-based registration. The approach resulted in qualitatively appropriate anatomical correspondences when projecting the preoperative structures on intraoperative images. With the best configuration, the registration steps showed accuracy and feasibility in aligning data, with global mean landmarks errors of 1.59 mm, 2.32 mm and 2.17 mm, respectively, a computation time that never exceeded 5 s, 25 s and 11 s, respectively, and a user interaction limited to manual initialization of the 3D/2D registration.
Conclusion
An image fusion-based approach has been specifically proposed for RE procedures guidance. The catheter manipulation strategy based on the fusion of pre- and intraoperative images has the potential to support different steps of the RE clinical workflow and to guide the overall procedure.












Similar content being viewed by others
Code availability
The code implemented for this study is not publicly available.
References
World Health Organization. https://www.who.int/news-room/fact-sheets/detail/cancer
Jafargholi Rangraz E, Coudyzer W, Maleux G, Baete K, Deroose CM, Nuyts J (2019) Multi-modal image analysis for semi-automatic segmentation of the total liver and liver arterial perfusion territories for radioembolization. EJNMMI Res 9:19
Spahr N, Thoduka S, Abolmaali N, Kikinis R, Schenk A (2019) Multimodal image registration for liver radioembolization planning and patient assessment. Int J Comput Assist Radiol Surg 14(2):215–225
Garin E, Rolland Y, Edeline J, Icard N, Lenoir L, Laffont S, Mesbah H, Breton M, Sulpice L, Boudjema K, Rohou T, Raoul JL, Clément B, Boucher E (2015) Personalized dosimetry and intensification concept with 90Y-loaded glass microsphere radioembolization induce prolonged overall survival in hepatocellular carcinoma patients with portal vein thrombosis. J Nucl Med 56(3):339–346
Puppala S (2019) Technical update on transcatheter arterial chemoembolization. Hepatoma Res. https://doi.org/10.20517/2394-5079.2019.28
Clifford MA, Banovac F, Levy E, Cleary K (2002) Assessment of hepatic motion secondary to respiration for computer assisted interventions. Comput Aided Surg Off J Int Soc Comput Aided Surg 7(5):291–299
Venkatraman V, Horn MHV, Weeks S, Bullitt E (2004) Liver motion due to needle pressure, cardiac, and respiratory motion during the TIPS procedure. In: Medical Image Computing and Computer-Assisted Intervention—MICCAI 2004, pp 66–72
Blackall JM, King AP, Penney GP, Adam A, Hawkes DJ (2001) A statistical model of respiratory motion and deformation of the liver. In: Medical Image Computing and Computer-Assisted Intervention—MICCAI 2001. Springer, Berlin, pp 1338–1340
Rohlfing T, Maurer CR, O’Dell WG, Zhong J (2004) Modeling liver motion and deformation during the respiratory cycle using intensity-based nonrigid registration of gated MR images. Med Phys 31(3):427–432
Ambrosini P, Ruijters D, Niessen WJ, Moelker A, van Walsum T (2015) Continuous roadmapping in liver TACE procedures using 2D–3D catheter-based registration. Int J Comput Assist Radiol Surg 10(9):1357–1370
Ambrosini P, Smal I, Ruijters D, Niessen WJ, Moelker A, Van Walsum T (2017) A hidden Markov model for 3D Catheter Tip Tracking With 2D X-ray catheterization sequence and 3D rotational angiography. IEEE Trans Med Imaging 36(3):757–768
Bargellini I, Turini F, Bozzi E, Lauretti D, Cicorelli A, Lunardi A, Cioni R, Bartolozzi C (2013) Image fusion of preprocedural CTA with real-time fluoroscopy to guide proper hepatic artery catheterization during transarterial chemoembolization of hepatocellular carcinoma: a feasibility study. Cardiovasc Intervent Radiol 36(2):526–530
Guevara JG, Peterlik I, Berger M-O, Cotin S (2018) Biomechanics-based graph matching for augmented CT-CBCT. Int J Comput Assist Radiol Surg 13(6):805–813
Kaladji A, Lucas A, Kervio G, Haigron P, Cardon A (2010) Sizing for endovascular aneurysm repair: clinical evaluation of a new automated three-dimensional software. Ann Vasc Surg 24(7):912–920
Duménil A, Kaladji A, Castro M, Göksu C, Lucas A, Haigron P (2016) A versatile intensity-based 3D/2D rigid registration compatible with mobile C-arm for endovascular treatment of abdominal aortic aneurysm. Int J Comput Assist Radiol Surg 11(9):1713–1729
Kim K, Park S, Hong H, Shin YG (2005) Fast 2D–3D registration using GPU-based preprocessing. In: Proceedings of 7th international workshop on enterprise networking and computing in healthcare industry, 2005. HEALTHCOM 2005, pp 139–143
Kubias A, Deinzer F, Feldmann T, Paulus D (2007) Extended global optimization strategy for rigid 2D/3D image registration. In: Computer analysis of images and patterns, Berlin, pp 759–76794
Sailer AM, Wagemans BA, Das M, de Haan MW, Nelemans PJ, Wildberger JE, Schurink GW (2015) Quantification of respiratory movement of the aorta and side branches. J Endovasc Ther Off J Int Soc Endovasc Spec 22(6):905–911
Raheem A, Carrell T, Modarai B, Penney G (2010) Non-rigid 2D–3D image registration for use in endovascular repair of abdominal aortic aneurysms. Presented at the Medical Image Understanding and Analysis, pp 153–157
Penney GP, Weese J, Little JA, Desmedt P, Hill DLG, Hawkes DJ (1998) A comparison of similarity measures for use in 2D–3D medical image registration. In: Wells WM, Colchester A, Delp S (eds) Medical Image Computing and Computer-Assisted Intervention—MICCAI’98. Springer, Berlin, pp 1153–1161
Lalys F, Favre K, Villena A, Durrmann V, Colleaux M, Lucas A, Kaladji A (2018) A hybrid image fusion system for endovascular interventions of peripheral artery disease. Int J Comput Assist Radiol Surg 13(7):997–1007
Wehrenberg-Klee E, Gandhi RT, Ganguli S (2019) Patient selection and clinical outcomes of Y90 in hepatocellular carcinoma. Tech Vasc Interv Radiol 22(2):70–73
Wondergem M, Smits ML, Elschot M, de Jong HW, Verkooijen HM, van den Bosch MA, Nijsen JF, Lam MG (2013) 99 mTc-macroaggregated albumin poorly predicts the intrahepatic distribution of 90Y resin microspheres in hepatic radioembolization. J Nucl Med. https://doi.org/10.2967/jnumed.112.117614
Kafrouni M, Allimant C, Fourcade M, Vauclin S, Guiu B, Mariano-Goulart D, Bouallègue FB (2019) Analysis of differences between 99mTc-MAA SPECT- and 90Y-microsphere PET-based dosimetry for hepatocellular carcinoma selective internal radiation therapy. EJNMMI Res 9(1):62
Knesaurek K, Machac J, Muzinic M, DaCosta M, Zhang Z, Heiba S (2010) Quantitative comparison of yttrium-90 (90Y)-microspheres and technetium-99m (99mTc)-macroaggregated albumin SPECT images for planning 90Y therapy of liver cancer. Technol Cancer Res Treat 9(3):253–262
Varnavas A, Carrell T, Penney G (2013) Increasing the automation of a 2D-3D registration system. IEEE Trans Med Imaging 32(2):387–399
Acknowledgements
This work was partially supported by the French National Association for Research and Technology (ANRT, Grant No. 2017/1639), and by the French National Research Agency (ANR) in the framework of the Investissement d’Avenir Program through Labex CAMI (ANR-11- LABX-0004).
Author information
Authors and Affiliations
Contributions
H.H and P.H conceived the presented idea. H.H collected the data, carried out the experiments and performed the analysis. F.L and P.H verified the analytical method. P.H supervised the findings of the work. H.H wrote the manuscript with support from P.H, F.L, Y.R and A.P. All authors discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval
This was an observational study for which anonymous retrospective data were used. It was performed in accordance with the ethical standards.
Consent to participate
Informed consent was obtained from all individual patients included in the study.
Consent for publication
Informed consent was obtained from all patients for whom data are included in this article.
Availability of data and materials
The data that support the findings of this study are available from the corresponding author, H.H, upon reasonable request.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hammami, H., Lalys, F., Rolland, Y. et al. Catheter navigation support for liver radioembolization guidance: feasibility of structure-driven intensity-based registration. Int J CARS 15, 1881–1894 (2020). https://doi.org/10.1007/s11548-020-02250-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11548-020-02250-8