Abstract
Twenty-four aryl benzyl sulfides, diaryl sulfides and dibenzyl sulfides were synthesized by four methods and characterized by 1H NMR, FT-IR and Gas chromatography. The reaction conditions of different synthesis methods were studied from the aspects of time, solvent, base and dispersant. The molecular structures of benzylphenyl sulfide (2S), (4-tert-butylbenzyl)(4-methylphenyl) sulfide (4S), (4-methylbenzyl)(4-methylphenyl) sulfide (9S), di(4-methylphenyl) sulfide (11S), (3,5-dimethylphenyl)(4-methyl phenyl) sulfide (15S), and dibenzyl sulfide (19S) [22] have been determined by single-crystal X-ray crystallography. Compounds 2S and 15S crystallize in the monoclinic space group P21/c, with a = 12.278(3), b = 15.894(3), c = 5.6056(11) Å, β = 94.532(2)°, and Z = 4 for 2S, and a = 9.800(9), b = 7.950(7), c = 16.690(15) Å, β = 100.890(12)°, and Z = 4 for 15S. The unit cell of 4S has a triclinic Pī symmetry with the cell parameters a = 6.0436(10), b = 8.7871(14), c = 15.535(2) Å, α = 81.921(2)°, β = 81.977(2)°, γ = 80.889(2)°, and Z = 2. Compounds 9S and 11S both crystallize in the orthorhombic space group P212121, with a = 6.188(3), b = 8.041(4), c = 26.005(14) Å, and Z = 4 for 9S, and a = 5.835(2), b = 8.010(3), c = 25.131(9) Å, and Z = 4 for 11S.
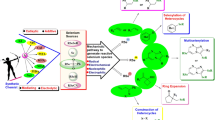
Graphic Abstract
Twenty-four aryl sulfide compounds with different substituents were synthesized and characterized, and the molecular structures of six different sulfide compounds have been determined by single-crystal X-ray crystallography.











Similar content being viewed by others
References
Choghamarani AG, Seydyosefi Z, Tahmasbi B (2018) Appl Organomet Chem 32:e4396–4404
Ghaderi A (2016) Tetrahedron 72:4758–4782
Salom-Roig X, Bauder C (2020) Synthesis-Stuttgart 52(7): 964–978
Wojaczyńska E (2010) J Wojaczyński Chem Rev 110:4303–4356
Rozen S (2014) Acc Chem Res 47:2378–2389
Percec V, Bae JY, Hill DH (1995) J Org Chem 60(21):6895–6903
Choghamarani AG, Heidarnezhad Z, Tahmasbi B (2019) ChemistrySelect 4:8860–8869
Velasco N, Virumbrales C, Sanz R (2018) Organic Lett 20(10):2848–2852
Firouzabadi H, Iranpoor N, Samadi A (2014) Tetrahedron Lett 55:1212–1217
O’Mahony GE, Ford A, Maguire AR (2012) J Org Chem 77:3288–3296
Migita T, Shimizu T, Asami Y, Shiobara J, Kato Y, Kosugi M (1980) Bull Chem Soc Jpn 53:1385–1389
She J, Jiang Z, Wang YG (2009) Tetrahedron Lett 50:593–596
SMART and SAINT+ for Windows NT Version 6.02a. Bruker Analytical X-ray Instruments Inc., Madison, Wisconsin, USA, 1998.
Sheldrick GM (1996) Sadabs. University of Göttingen, Göttingen, Germany
G.M. Sheldrick. Shelxtl (version 5.1), Software Reference Manual, Bruker AXS Inc., Madison, Wisconsin, USA, 1997.
Sheldrick GM (2015) Acta Cryst C 71:3–8
Zhang XY, Song HB, Li QS, Liu XF, Tang LF (2007) Polyhedron 26:3743–3749
Yan YG, Xie MJ, Chen YR, Wu QX, Yu ZL (1999) J. Sichuan Univ. (Nat. Sci. Ed.) 36(3):535–540
Gogoi P, Hazarika S, Sarma MJ, Barman P (2014) Tetrahedron 70(41):7484–7489
Ramirez AA, Cross ED, Bierenstiel M (2014) Dalton Trans 43(8):3104–3113
Kumar S, Kandasamy K, Singh HB, Butcher RJ (2004) New J Chem 28:640–645
Hansson C (2006) Acta Cryst E62:o2377–o2379
Lee E, Ju H, Kim S, Park KM, Lee SS (2015) Cryst. Growth Des 15(11):5427–5436
Acknowledgements
This project was supported by the National Natural Science Foundation of China (21372007).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interests
The authors declare that they have no conflict of interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhou, WY., Chen, M., Zhang, PZ. et al. Methods, Syntheses and Characterization of Diaryl, Aryl Benzyl, and Dibenzyl Sulfides. J Chem Crystallogr 51, 301–310 (2021). https://doi.org/10.1007/s10870-020-00859-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10870-020-00859-w