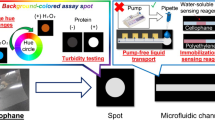
Abstract
The uncontrolled volume of sample has a significant influence on the outcome of the colorimetric detection method performed on paper-based microfluidic devices. In a reliable colorimetric assay, the results should be completely dependent on the concentration of analyte in the sample rather than its volume. However, this effect is usually overlooked in the development of paper-based microfluidic devices as a simple, inexpensive, portable and user-friendly medical diagnostic system. In this work, a volume-independent micro paper-based analytical device has been introduced in which the need for precise controlling of sample volume is eliminated. We have shown that the proposed device is only sensitive to the concentration of analyte (glucose) in the sample instead of its volume. We have also demonstrated that the proposed device addresses the reported problem of colour heterogeneity and is relatively insusceptible to the variation of reagents volume. An inexpensive smart phone camera is used for capturing the images and a portable 3D printed tool is designed to eliminate the environmental illuminations. To simulate the real condition, human plasma samples are used throughout the experiments.










Similar content being viewed by others
Data availability
All data and materials are truthfully available.
Code availability
Not applicable.
References
Abe K, Suzuki K, Citterio D (2008) Inkjet-printed microfluidic multianalyte chemical sensing paper. Anal Chem 80:6928–6934
Cai L, Wang Y, Wu Y et al (2014) Fabrication of a microfluidic paper-based analytical device by silanization of filter cellulose using a paper mask for glucose assay. Analyst 139:4593–4598
Carrilho E, Martinez AW, Whitesides GM (2009) Understanding wax printing: a simple micropatterning process for paper-based microfluidics. Anal Chem 81:7091–7095
Cate DM, Dungchai W, Cunningham JC et al (2013) Simple, distance-based measurement for paper analytical devices. Lab Chip 13:2397–2404
Chen X, Chen J, Wang F et al (2012) Determination of glucose and uric acid with bienzyme colorimetry on microfluidic paper-based analysis devices. Biosens Bioelectron 35:363–368
de Freitas SV, de Souza F, cio R, Rodrigues Neto JC, et al (2018) uncovering the formation of color gradients for glucose colorimetric assays on microfluidic paper-based analytical devices by mass spectrometry imaging. Anal Chem 90:11949–11954
de Tarso GP, Cardoso TMG, Garcia CD et al (2014) A handheld stamping process to fabricate microfluidic paper-based analytical devices with chemically modified surface for clinical assays. Rsc Adv 4:37637–37644
Dungchai W, Chailapakul O, Henry CS (2010) Use of multiple colorimetric indicators for paper-based microfluidic devices. Anal Chim Acta 674:227–233
Evans, Elizabeth and Gabriel, Ellen Flàvia Moreira and Coltro, Wendell Karlos Tomazelli and Garcia CD (2014) Rational selection of substrates to improve color intensity and uniformity on microfluidic paper-based analytical devices. Analyst 139:2127–2132
Evans E, Gabriel EFM, Benavidez TE et al (2014b) Modification of microfluidic paper-based devices with silica nanoparticles. Analyst 139:5560–5567
Figueredo F, Garcia PT, Cortón E, Coltro WKT (2016) Enhanced analytical performance of paper microfluidic devices by using Fe3O4 nanoparticles, MWCNT, and graphene oxide. ACS Appl Mater Interfaces 8:11–15. https://doi.org/10.1021/acsami.5b10027
Gabriel EFM, Garcia PT, Cardoso TMG et al (2016) Highly sensitive colorimetric detection of glucose and uric acid in biological fluids using chitosan-modified paper microfluidic devices. Analyst 141:4749–4756
Houghtaling J, Liang T, Thiessen G, Fu E (2013) Dissolvable bridges for manipulating fluid volumes in paper networks. Anal Chem 85:11201–11204
Im SH, Kim KR, Park YM et al (2016) An animal cell culture monitoring system using a smartphone-mountable paper-based analytical device. Sens Actuators B Chem 229:166–173. https://doi.org/10.1016/j.snb.2016.01.121
Jiang Y, Zhao H, Lin Y et al (2010) Colorimetric detection of glucose in rat brain using gold nanoparticles. Angew Chemie Int Ed 49:4800–4804. https://doi.org/10.1002/anie.201001057
Klonoff DC (2014) Point-of-care blood glucose meter accuracy in the hospital setting. Diabetes Spectr 27:174–179. https://doi.org/10.2337/diaspect.27.3.174
Lenk G, Pohanka A, Stemme G et al (2013) A disposable chip enabling metering in dried blood spot sampling. Micro TAS 2013:281–283
Lewis GG, DiTucci MJ, Phillips ST (2012) Quantifying analytes in paper-based microfluidic devices without using external electronic readers. Angew Chemie Int Ed 51:12707–12710
Lopez-Ruiz N, Curto VF, Erenas MM et al (2014) Smartphone-based simultaneous pH and nitrite colorimetric determination for paper microfluidic devices. Anal Chem 86:9554–9562
Lu Y, Shi W, Jiang L et al (2009) Rapid prototyping of paper-based microfluidics with wax for low-cost, portable bioassay. Electrophoresis 30:1497–1500
Martinez AW, Phillips ST, Nie Z, Cheng C-M, Carrilho E, Wiley BJ, Whitesides GM (2010) Programmable diagnostic devices made from paper and tape. Lab Chip 10:2499–2504
Martinez AW, Phillips ST, Butte MJ, Whitesides GM (2007) Patterned paper as a platform for inexpensive, low-volume, portable bioassays. Angew Chemie Int Ed 46:1318–1320
Martinez AW, Phillips ST, Carrilho E et al (2008) Simple telemedicine for developing regions: camera phones and paper-based microfluidic devices for real-time, off-site diagnosis. Anal Chem 80:3699–3707
Martinez AW, Phillips ST, Whitesides GM, Carrilho E (2009) Diagnostics for the developing world: microfluidic paper-based analytical devices
Mohammadi S, Maeki M, Mohamadi RM et al (2015) An instrument-free, screen-printed paper microfluidic device that enables bio and chemical sensing. Analyst 140:6493–6499. https://doi.org/10.1039/c5an00909j
Nie J, Liang Y, Zhang Y, Le S, Li D, Zhang S (2013) One-step patterning of hollow microstructures in paper by laser cutting to create microfluidic analytical devices. Analyst 138:671–676
Nie J, Zhang Y, Lin L et al (2012) Low-cost fabrication of paper-based microfluidic devices by one-step plotting. Anal Chem 84:6331–6335. https://doi.org/10.1021/ac203496c
Nie Z, Deiss F, Liu X et al (2010) Integration of paper-based microfluidic devices with commercial electrochemical readers. Lab Chip 10:3163–3169
Nilghaz A, Shen W (2015) Low-cost blood plasma separation method using salt functionalized paper. Rsc Adv 5:53172–53179
Nilghaz A, Zhang L, Shen W (2015) Coffee stains on paper. Chem Eng Sci 129:34–41
Noiphung J, Songjaroen T, Dungchai W et al (2013) Electrochemical detection of glucose from whole blood using paper-based microfluidic devices. Anal Chim Acta 788:39–45. https://doi.org/10.1016/j.aca.2013.06.021
Organization WH, others (2016) Global report on diabetes 2016. ISBN 978 92 4 156525 7
Oyola-Reynoso S, Heim AP, Halbertsma-Black J et al (2015) Draw your assay: Fabrication of low-cost paper-based diagnostic and multi-well test zones by drawing on a paper. Talanta 144:289–293
Schilling KM, Lepore AL, Kurian JA, Martinez AW (2012) Fully enclosed microfluidic paper-based analytical devices. Anal Chem 84:1579–1585. https://doi.org/10.1021/ac202837s
Smith GT, Dwork N, Khan SA et al (2016) Robust dipstick urinalysis using a low-cost{,} micro-volume slipping manifold and mobile phone platform. Lab Chip 16:2069–2078. https://doi.org/10.1039/C6LC00340K
Songjaroen T, Dungchai W, Chailapakul O et al (2012) Blood separation on microfluidic paper-based analytical devices. Lab Chip 12:3392–3398
Tonyushkina K, Nichols JH (2009) Glucose meters: a review of technical challenges to obtaining accurate results. J Diabetes Sci Technol 3:971–980
Vella SJ, Beattie P, Cademartiri R et al (2012) Measuring markers of liver function using a micropatterned paper device designed for blood from a fingerstick. Anal Chem 84:2883–2891
Walsh J, Roberts R, Vigersky RA, Schwartz F (2012) New criteria for assessing the accuracy of blood glucose monitors meeting, October 28, 2011
Wei X, Tian T, Jia S et al (2016) Microfluidic distance readout sweet hydrogel integrated paper-based analytical device (μDiSH-PAD) for visual quantitative point-of-care testing. Anal Chem 88:2345–2352. https://doi.org/10.1021/acs.analchem.5b04294
Wexler DJ, Meigs JB, Cagliero E et al (2007) Prevalence of hyper-and hypoglycemia among inpatients with diabetes: a national survey of 44 US hospitals. Diabetes Care 30:367–369
Yamada K, Shibata H, Suzuki K, Citterio D (2017) Toward practical application of paper-based microfluidics for medical diagnostics: state-of-the-art and challenges. Lab Chip 17:1206–1249
Yang X, Forouzan O, Brown TP, Shevkoplyas SS (2012) Integrated separation of blood plasma from whole blood for microfluidic paper-based analytical devices. Lab Chip 12:274–280
Yu J, Ge L, Huang J, Wang S, Ge S (2011) Microfluidic paper-based chemiluminescence biosensor for simultaneous determination of glucose and uric acid. Lab Chip 11:1286–1291
Zhou M, Yang M, Zhou F (2014) Paper based colorimetric biosensing platform utilizing cross-linked siloxane as probe. Biosens Bioelectron 55:39–43
Zhu W-J, Feng D-Q, Chen M et al (2014) Bienzyme colorimetric detection of glucose with self-calibration based on tree-shaped paper strip. Sensors Actuators B Chem 190:414–418
Acknowledgments
Financial support provided by the Research Council of University of Tehran is gratefully acknowledged.
Funding
This work is supported with an INSF grant number 95848969.
Author information
Authors and Affiliations
Contributions
Not applicable.
Corresponding authors
Ethics declarations
Conflicts of interest
The authors declare that there is no conflict of interest regarding the publication of this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Choobbari, M.L., Rad, M.B., Jahanshahi, A. et al. A sample volume independent paper microfluidic device for quantifying glucose in real human plasma. Microfluid Nanofluid 24, 74 (2020). https://doi.org/10.1007/s10404-020-02382-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10404-020-02382-y