Abstract
Aims
Knowledge on the ecophysiology of cobalt hyperaccumulator species is limited. The nickel hyperaccumulator Rinorea cf. bengalensis from Borneo can accumulate high concentrations of cobalt in nature. This study investigates the cobalt accumulation potential of Rinorea cf. bengalensis in relation to nickel concentrations in soils and the subsequent tissue and cellular-level distributions of cobalt, nickel and major cations.
Methods
Seedlings of Rinorea cf. bengalensis were grown in mixed treatments on ultramafic soil containing a high concentration of available nickel. Cobalt and nickel salts were then added to the soil to study their interactions. The tissue and cellular-level distributions of cobalt, calcium, nickel, and potassium were investigated using synchrotron-based X-ray fluorescence microscopy.
Results
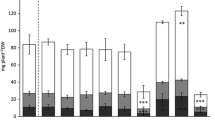
The maximum foliar cobalt concentration reached 1200 μg g−1. Accumulation of cobalt competed with nickel accumulation although nickel seems to stimulate cobalt phloem translocation. Plants suffered toxicity in the treatment with the highest soil cobalt concentration. Cobalt and nickel have contrasting distribution patterns in the leaves of Rinorea cf. bengalensis, with cobalt mainly excreted on the surface of the leaves, whereas nickel is localised in foliar epidermal cells.
Conclusions
Rinorea cf. bengalensis can accumulate high concentrations of cobalt, but is intrinsically more tolerant to nickel. It does not rely on a similar sequestration mechanism for both metals, which could explain the lesser tolerance for cobalt. Nickel appears to be essential for the plant to tolerate high cobalt concentrations. Further studies intending to develop agronomic practices are needed to determine the viability of Rinorea cf. bengalensis for nickel-cobalt agromining.









Similar content being viewed by others
References
Baker AJM, Brooks RR (1989) Terrestrial higher plants which hyperaccumulate metallic elements—a review of their distribution, ecology and phytochemistry. Biorecovery 1:81–126
Ballard HE, de Paula-Souza J, Wahlert GA (2014) Violaceae. In: Kubitzki K (ed) The families and genera of vascular plants. Springer, Berlin, pp 303–322. https://doi.org/10.1007/978-3-642-39417-1_25
Bani A, Echevarria G, Sulçe S, Morel JL (2015) Improving the agronomy of Alyssum murale for extensive phytomining: a five-year field study. Int J Phytoremediat 17:117–127. https://doi.org/10.1080/15226514.2013.862204
Blamey FPC, Joyce DC, Edwards DG, Asher CJ (1986) Role of trichomes in sunflower tolerance to manganese toxicity. Plant Soil 91:171–180. https://doi.org/10.1007/BF02181785
Bouman R, van Welzen P, Sumail S, Echevarria G, Erskine PD, van der Ent A (2018) Phyllanthus rufuschaneyi: a new nickel hyperaccumulator from Sabah (Borneo Island) with potential for tropical phytomining. Bot Stud 59:9. https://doi.org/10.1186/s40529-018-0225-y
Boyd RS (2007) The defense hypothesis of elemental hyperaccumulation: status, challenges and new directions. Plant Soil 293:153–176. https://doi.org/10.1007/s11104-007-9240-6
Boyd RS, Martens SN (1992) The raison d’être for metal hyperaccumulation by plants. In: Baker AJM, Proctor J, Reeves RD (eds) The vegetation of ultramafic (serpentine) soils. Intercept Limited, Andover, pp 279–289
Brooks RR, Wither ED (1977) Nickel accumulation by Rinorea bengalensis (Wall.) O.K. J Geochem Explor 7:295–300. https://doi.org/10.1016/0375-6742(77)90085-1
Brooks RR, Lee J, Reeves RD, Jaffré T (1977) Detection of nickeliferous rocks by analysis of herbarium specimens of indicator plants. J Geochem Explor 7:49–57. https://doi.org/10.1016/0375-6742(77)90074-7
Chaney RL, Reeves RD, Baklanov IA, Centofanti T, Broadhurst CL, Baker AJM, Angle JS, van der Ent A, Roseberg RJ (2014) Phytoremediation and phytomining: using plants to remediate contaminated or mineralized environments, chap. 15. In: Rajakaruna N, Boyd RS, Harris T (eds) Plant ecology and evolution in harsh environments. Nova Science, New York, pp 365–391
Chen C, Huang D, Liu J (2009) Functions and toxicity of nickel in plants: recent advances and future prospects. Clean 37:304–313. https://doi.org/10.1002/clen.200800199
De Hertogh AA, Mayeux PA, Evans HJ (1964) The relationship of cobalt requirement to propionate metabolism in rhizobium. J Biol Chem 239:2446–2453
Dixon NE, Gazzola TC, Blakeley RL, Zermer B (1975) Jack bean urease (EC 3.5.1.5). A metalloenzyme. A simple biological role for nickel? J Am Chem Soc 97(14):4131–4133
Guillot S, Hattori K (2013) Serpentinites: essential roles in geodynamics, arc volcanism, sustainable development, and the origin of life. Elements 9:95–98. https://doi.org/10.2113/gselements.9.2.95
Homer FA, Morrison RS, Brooks RR, Clemens J, Reeves RD (1991) Comparative-studies of nickel, cobalt, and copper uptake by some nickel hyperaccumulators of the genus Alyssum. Plant Soil 138:195–205
Hörger AC, Fones HN, Preston GM (2013) The current status of the elemental defense hypothesis in relation to pathogens. Front Plant Sci 4:395. https://doi.org/10.3389/fpls.2013.00395
Jones MWM, Kopittke PM, Casey LW, Reinhardt J, Pax F, Blamey C, van der Ent A (2020) Assessing radiation dose limits for X-ray fluorescence microscopy analysis of plant specimens. Ann Bot 125:599–610. https://doi.org/10.1093/aob/mcz195
Kabata-Pendias A (2010) Trace elements in soils and plants. CRC Press/Taylor & Francis Group 4th edition, Boca Raton
Kabeya FI, Pongrac P, Lange B, Faucon MP, Van Elteren JT, Šala M, Šelih VS, Eeckhoudt EV, Verbruggen N (2018) Tolerance and accumulation of cobalt in three species of Haumaniastrum and the influence of copper. Environ Exp Bot 149:27–33. https://doi.org/10.1016/j.envexpbot.2018.01.018
Keeling SM, Stewart RB, Anderson CW, Robinson BH (2003) Nickel and cobalt phytoextraction by the hyperaccumulator Berkheya coddii: implications for polymetallic phytomining and phytoremediation. Int J Phytoremediation 5:235–244. https://doi.org/10.1080/713779223
Kirkham R, Dunn PA, Kucziewski A, Siddons DP, Dodanwela R, Moorhead G, Ryan CG, De Geronimo G, Beuttenmuller R, Pinelli D, Pfeffer M, Davey P, Jensen M, Paterson D, de Jonge MD, Kusel M, McKinlay J (2010) The Maia spectroscopy detector system: engineering for integrated pulse capture, low-latency scanning and real-time processing. AIP Conf Proc 1234:240–243. https://doi.org/10.1063/1.3463181
Küpper H, Lombi E, Zhao FJ, Wieshammer G, McGrath SP (2001) Cellular compartmentation of nickel in the hyperaccumulators Alyssum lesbiacum, Alyssum bertolonii and Thlaspi goesingense. J Exp Bot 52:2291–2300. https://doi.org/10.1093/jexbot/52.365.2291
Lange B, van der Ent A, Baker AJ, Echevarria G, Mahy G, Malaisse F, Meerts P, Pourret O, Verbruggen N, Faucon MP (2017) Copper and cobalt accumulation in plants: a critical assessment of the current state of knowledge. New Phytol 213:537–551. https://doi.org/10.1111/nph.14175
Li YM, Chaney R, Brewer E, Roseberg R, Angle JS, Baker AJM, Reeves R, Nelkin J (2003) Development of a technology for commercial phytoextraction of nickel: economic and technical considerations. Plant Soil 249:107–115. https://doi.org/10.1023/A:1022527330401
Liu J, Reid RJ, Smith FA (2000) The mechanism of cobalt toxicity in mung beans. Physiol Plant 110:104–110. https://doi.org/10.1034/j.1399-3054.2000.110114.x
Lopez-Moreno ML, Aviles LL, Perez NG, Irizarry BT, Perales O, Cedeno-Mattei Y, Roman F (2016) Effect of cobalt-ferrite nanoparticles on the growth and development of Solanum lycopersicum L. (tomato plants). Sci Total Environ 550:45–52. https://doi.org/10.1016/j.scitotenv.2016.01.063
Malik M, Chaney RL, Brewer EP, Angle JS (2000) Phytoextraction of soil cobalt using hyperaccumulator plants. Int J Phytorem 2:319–329. https://doi.org/10.1080/15226510008500041
Morrissey J, Baxter IR, Lee J, Li L, Lahner B, Grotz N, Kaplan J, Salt DE, Guerinot ML (2009) The ferroportin metal efflux proteins function in iron and cobalt homeostasis in Arabidopsis. Plant Cell 21:3326–3338. https://doi.org/10.1105/tpc.109.069401
Nkrumah P (2018) Hyperaccumulation of trace elements in native species from Sabah (Malaysia): new discoveries and practical considerations for viable tropical nickel agromining, PhD Thesis, Sustainable Minerals Institute, The University of Queensland
Nkrumah PN, Baker AJM, Chaney RL, Erskine PD, Echevarria G, Morel JL, van der Ent A (2016) Current status and challenges in developing nickel phytomining: an agronomic perspective. Plant Soil 406:55–69. https://doi.org/10.1007/s11104-016-2859-4
Nkrumah P, Echevarria G, Erskine PD, van der Ent A (2018a) Contrasting nickel and zinc hyperaccumulation in subspecies of Dichapetalum gelonioides from Southeast Asia. Sci Rep 8:9659. https://doi.org/10.1038/s41598-018-26859-7
Nkrumah PN, Echevarria G, Erskine PD, van der Ent A (2018b) Nickel hyperaccumulation in Antidesma montis-silam: from herbarium discovery to collection in the native habitat. Ecol Res 33:675–685. https://doi.org/10.1007/s11284-018-1601-5
Nkrumah PN, Echevarria G, Erskine PD, Chaney RL, Sumail S, van der Ent A (2019a) Soil amendments affecting nickel uptake and growth performance of tropical ‘metal crops’ used for agromining. J Geochem Explor 203:78–86. https://doi.org/10.1016/j.gexplo.2019.03.009
Nkrumah PN, Echevarria G, Erskine PD, Chaney RL, Sumail S, van der Ent A (2019b) Growth effects in tropical nickel-agromining ‘metal crops' in response to nutrient dosing. J Plant Nutr Soil Sci 182:715–728. https://doi.org/10.1002/jpln.201800468
Nkrumah PN, Echevarria G, Erskine PD, Chaney RL, Sumail S, van der Ent A (2019c) Effect of nickel concentration and soil pH on metal accumulation and growth in tropical agromining ‘metal crops’. Plant Soil 443:27–39. https://doi.org/10.1007/s11104-019-04200-z
Oven M, Grill E, Golan-Goldhirsh A, Kutchan TM, Zenk MH (2002) Increase of free cysteine and citric acid in plant cells exposed to cobalt ions. Phytochem 60:467–474. https://doi.org/10.1016/S0031-9422(02)00135-8
Proctor J (2003) Vegetation and soil and plant chemistry on ultramafic rocks in the tropical Far East. Perspect Plant Ecol Evol Syst 6:105–124. https://doi.org/10.1078/1433-8319-00045
Reeves RD, Baker AJM, Jaffré T, Erskine PD, Echevarria G, van der Ent A (2018) A global database for plants that hyperaccumulate metal and metalloid trace elements. New Phytol 218:407–411. https://doi.org/10.1111/nph.14907
Ryan CG (2000) Quantitative trace element imaging using PIXE and the nuclear microprobe. Int J Imag Syst Tech 11:219–230. https://doi.org/10.1002/ima.1007
Ryan CG, Jamieson DN (1993) Dynamic analysis: on-line quantitative PIXE microanalysis and its use in overlap-resolved elemental mapping. Nucl Instrum Meth B 77:203–214. https://doi.org/10.1016/0168-583X(93)95545-G
Ryan CG, Cousens D, Sie S, Griffin W (1990) Quantitative analysis of PIXE spectra in geoscience applications. Nucl Instrum Meth B 49:271–276. https://doi.org/10.1016/0168-583X(90)90259-W
Ryan CG, Etschmann BE, Vogt S, Maser J, Harland CL, van Achterbergh E, Legnini D (2005) Nuclear microprobe-synchrotron synergy: towards integrated quantitative real-time elemental imaging using PIXE and SXRF. Nucl Instrum Meth B 231:183–188. https://doi.org/10.1016/j.nimb.2005.01.054
Ryan CG, Kirkham R, Hough RM, Moorhead G, Siddons DP, De Jonge MD, Paterson DJ, De Geronimo G, Howard DL, Cleverley JS (2010) Elemental X-ray imaging using the Maia detector array: the benefits and challenges of large solid-angle. Nucl Instrum Meth A 619:37–43. https://doi.org/10.1016/j.nima.2009.11.035
Ryan CG, Siddons DP, Kirkham R, Li ZY, de Jonge MD, Paterson DJ, Kuczewski A, Howard DL, Dunn PA, Falkenberg G, Boesenberg U, De Geronimo G, Fisher LA, Halfpenny A, Lintern MJ, Lombi E, Dyl KA, Jensen M, Moorhead GF, Cleverley JS, Hough RM, Godel B, Barnes SJ, James SA, Spiers KM, Alfeld M, Wellenreuther G, Vukmanovic Z, Borg S (2014) Maia X-ray fluorescence imaging: capturing detail in complex natural samples. X-ray optics and microanalysis. J Phys Conf Ser 499:012002. https://doi.org/10.1088/1742-6596/499/1/012002
Schroer CG, Boye P, Feldkamp JM, Patommel J, Samberg D, Schropp A, Schwab A, Stephan S, Falkenberg G, Wellenreuther G, Reimers N (2010) Hard X-ray nanoprobe at beamline P06 at PETRA III. Nucl Instrum Meth A 616:93–97. https://doi.org/10.1063/1.4952830
Siddons DP, Kirkham R, Ryan CG, De Geronimo G, Dragone A, Kuczewski AJ, Li ZY, Carini GA, Pinelli D, Beuttenmuller R, Elliott D, Pfeffer M, Tyson TA, Moorhead GF, Dunn PA (2014) Maia X-ray microprobe detector Array system. J Phys Conf Ser 499:012001–012010. https://doi.org/10.1088/1742-6596/499/1/012001
Tappero R, Peltier E, Gräfe M, Heidel K, Ginder-Vogel M, Livi KJT, Rivers ML, Marcus MA, Chaney RL, Sparks DL (2007) Hyperaccumulator Alyssum murale relies on a different metal storage mechanism for cobalt than for nickel. New Phytol 175:641–654. https://doi.org/10.1111/j.1469-8137.2007.02134.x
van der Ent A, Mulligan D (2015) Multi-element concentrations in plant parts and fluids of Malaysian nickel hyperaccumulator plants and some economic and ecological considerations. J Chem Ecol 41:396–408. https://doi.org/10.1007/s10886-015-0573-y
van der Ent A, Baker AJM, Reeves RD, Pollard AJ, Schat H (2013) Hyperaccumulators of metal and metalloid trace elements: facts and fiction. Plant Soil 362:319–334. https://doi.org/10.1007/s11104-012-1287-3
van der Ent A, Jaffré T, L’Huillier L, Gibson N, Reeves RD (2015a) The flora of ultramafic soils in the Australia-Pacific region: state of knowledge and research priorities. Aust J Bot 63:173–190. https://doi.org/10.1071/BT15038
van der Ent A, Erskine PD, Sumail S (2015b) Ecology of nickel hyperaccumulator plants from ultramafic soils in Sabah (Malaysia). Chemoecol 25:243–259. https://doi.org/10.1007/s00049-015-0192-7
van der Ent A, Baker AJM, Reeves RD, Chaney RL, Anderson CWN, Meech JA, Erskine PD, Simonnot MO, Vaughan J, Morel JL, Echevarria G, Fogliani B, Qiu RL, Mulligan DR (2015c) Agromining: farming for metals in the future? Environ Sci Technol 49:4773–4780. https://doi.org/10.1021/es506031u
van der Ent A, van Balgooy MMJ, van Welzen P (2016) Actephila alanbakeri (Phyllanthaceae): a new nickel hyperaccumulating species from localised ultramafic soils in Sabah (Malaysia). Bot Stud 57:6. https://doi.org/10.1186/s40529-016-0122-1
van der Ent A, Callahan DL, Noller BN, Mesjasz-Przybylowicz J, Przybylowicz WJ, Barnabas A, Harris HH (2017) Nickel biopathways in tropical nickel hyperaccumulating trees from Sabah (Malaysia). Sci Rep 7:41861. https://doi.org/10.1038/srep41861
van der Ent A, Mak R, de Jonge MD, Harris HH (2018) Simultaneous hyperaccumulation of nickel and cobalt in the tree Glochidion cf. sericeum (Phyllanthaceae): elemental distribution and chemical speciation. Sci Rep 8:9683. https://doi.org/10.1038/s41598-018-26891-7
van der Ent A, Erskine PD, Malaisse F, Mesjasz-Przybylowicz J, Przybylowicz WJ, Barnabas AD, Sosnicka M, Harris HH (2019) Abnormal concentrations of Cu-Co in Haumaniastrum katangense, Haumaniastrum robertii and Aeolanthus biformifolius: contamination or hyperaccumulation? Metallomics 11:586–596. https://doi.org/10.1039/c8mt00300a
van der Ent A, de Jonge MD, Mak R, Mesjasz-Przybylowicz J, Przybylowicz WJ, Barnabas AD, Harris HH (2020a) X-ray fluorescence elemental mapping of roots, stems and leaves of the nickel hyperaccumulators Rinorea cf. bengalensis and Rinorea cf. javanica (Violaceae) from Sabah (Malaysia), Borneo. Plant Soil 448:15–36. https://doi.org/10.1007/s11104-019-04386-2
van der Ent A, Erskine PD, Vinya R, Malaisse F, Mesjasz-Przybylowicz J, Przybylowicz WJ, Barnabas AD, Harris HH (2020b) Elemental distribution and chemical speciation of copper and cobalt in three metallophytes from Zambia. Metallomics in press
van der Pas L, Ingle RA (2019) Towards an understanding of the molecular basis of nickel hyperaccumulation in plants. Plants 8:11. https://doi.org/10.3390/plants8010011
Wahlert GA, Ballard HE (2012) A phylogeny of Rinorea (Violaceae) inferred from plastid DNA sequences with an emphasis on the African and Malagasy species. Syst Bot 37:964–973. https://doi.org/10.1600/036364412X656392
Yamaguchi T, Tsukada C, Takahama K, Hirotomo T, Tomioka R, Takenaka C (2019) Localization and speciation of cobalt and nickel in the leaves of the cobalt-hyperaccumulating tree Clethra barbinervis. Trees 33:521–532. https://doi.org/10.1007/s00468-018-1797-6
Zelano I, Cloquet C, van der Ent A, Echevarria G, Gley R, Landrot G, Pollastri S, Fraysse F, Montargès-Pelletier E (2020) Coupling nickel chemical speciation and isotope ratios to decipher nickel dynamics in the Rinorea cf. bengalensis-soil system in Malaysian Borneo. Plant Soil, in press. https://doi.org/10.1007/s11104-020-04541-0
Acknowledgements
This research was undertaken at P06 at the Deutsches Elektronen-Synchrotron, a member of the Helmholtz Association (HGF). We thank Jan Garrevoet for technical support during the experiment. We acknowledge the Laboratory of Excellence Ressources 21 for financial support. The research leading to this result has been supported by the project CALIPSOplus under the Grant Agreement 730872 from the EU Framework Programme for Research and Innovation HORIZON 2020. A. van der Ent was the recipient of a Discovery Early Career Researcher Award (DE160100429) from the Australian Research Council. A.L.D Paul is the recipient of an Australian Government Research Training Program Scholarship and UQ Centennial Scholarship at The University of Queensland, Australia.
Author information
Authors and Affiliations
Contributions
A.L.D.P, P.N.N, G.E., S. S and A.vdE conducted the fieldwork and collected the samples in Malaysia. A.vdE, G.E. and K. S conducted the synchrotron X-ray Fluorescence Microscopy (XFM) experiment. AvdE performed the XFM data processing and analysis. All authors contributed to writing the manuscript.
Corresponding author
Additional information
Responsible Editor: Fangjie Zhao.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 29 kb)
Rights and permissions
About this article
Cite this article
Paul, A.L.D., Nkrumah, P.N., Echevarria, G. et al. Cobalt hyperaccumulation in Rinorea cf. bengalensis (Violaceae) from Sabah: accumulation potential and tissue and cellular-level distribution of cobalt. Plant Soil 455, 289–303 (2020). https://doi.org/10.1007/s11104-020-04629-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-020-04629-7