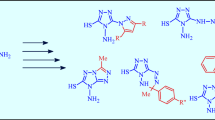
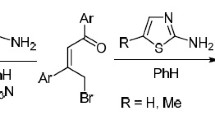
Alkylation of indolizinethione at the sulfur atom by the action of RCOCH2Br (R = Me, Ar, CO2Me, OEt) leads to thioethers, and subsequent closure of the thiazine ring of which leads to the formation of stable antiaromatic thiazino[4,3,2-cd]indolizines.



Similar content being viewed by others
References
(a) Taurins, A. Chemistry of Heterocyclic Compounds: Special Topics In Heterocyclic Chemistry; Wiley, 1976, Vol. 30, p. 245. (b) Windgassen, R. J., Jr.; Saunders, W. H.; Boekelheide. V. J. Am. Chem. Soc. 1959, 81, 1459.
(a) Kanemasa, S.; Nakamura, S.; Kajigaeshi, S. Chem. Lett. 1980, 9, 947. (b) Kanemasa, S.; Kobira, S.; Kajigaeshi, S. Chem. Lett. 1980, 9, 951. (c) Padwa, A.; Austin, D. J.; Precedo, L.; Zhi, L. J. Org. Chem. 1993, 58, 1140.
Babaev, E. V.; Vasilevich, N. I.; Ivushkina, A. S. Beilstein J. Org. Chem. 2005, 1. https://doi.org/10.1186/1860-5397-1-9.
Pozharskii, А. F. In Teoreticheskie osnovy khimii geterotsiklov (Theoretical Fundamentals of Heterocycle Chemistry [in Russian]); Moscow: Khimiya, 1985, p. 36.
Kurata, K.; Awaya, H.; Gotou, H.; Tominaga, Y.; Matsuda, Y.; Kobayashi, G. Chem. Pharm. Bull. 1985, 33, 3034.
Babaev, E. V; Nevskaya, A. A.; Dlynnikh, I. V. Chem. Heterocycl. Compd. 2015, 51, 269. [Khim. Geterotsikl. Soedin. 2015, 51, 269.]
Gevald, K.; Jansch, H. J. J. Prakt. Chem. 1976, 318, 313.
(a) Farrugia, L. J. J. Appl. Crystallogr. 1999, 32, 837. (b) Sheldrick, G. M. Acta Crystallogr., Sect. C: Struct. Chem. 2015, C71, 3.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, 2020, 56(7), 942–948
Rights and permissions
About this article
Cite this article
Babaev, E.V., Nevskaya, A.А., Dlynnikh, I.V. et al. Synthesis of antiaromatic thiazinoindolizines based on electrophilic cyclizations of indolizine-5-thione. Chem Heterocycl Comp 56, 942–948 (2020). https://doi.org/10.1007/s10593-020-02754-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-020-02754-w