Abstracts
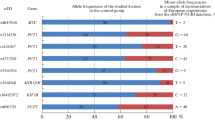
Recently, it has been shown that dysfunction of mitochondria is an important component of the molecular mechanisms of the development of many neurodegenerative diseases. These include multiple sclerosis, a chronic autoimmune and neurodegenerative disease of the central nervous system, which is characterized by clinical heterogeneity. The role of genetic variability of mitochondrial DNA in the development of various clinical forms of multiple sclerosis is poorly understood. The aim of present study was to analyze the association of ten mitochondrial DNA single nucleotide polymorphisms and the nine most common European mitochondrial haplogroups (H, J, K, U, T, I, V, W and X) with a severe and relatively rare multiple sclerosis disease form—primary progressive multiple sclerosis. 110 patients with primary progressive multiple sclerosis and 406 healthy controls were enrolled in the study, all ethnic Russians. For the first time association of the m.12308*G (rs2853498) variant (P = 0.024) and haplogroup U (P = 0.0004, passes the adjustment for multiple comparisons: Pcorr = 0.0076) with primary progressive multiple sclerosis was shown. Comparison of these data with the results of our previous study [1], that was focused on the role of mitochondrial genome variability in susceptibility to the most common form of multiple sclerosis, relapsing-remitting multiple sclerosis, leads to the conclusion that two different mitochondrial haplogroups, U and J, are involved in the development of two different clinical forms of multiple sclerosis. The results may contribute to the identification of new targets for the treatment of primary progressive multiple sclerosis, for which there is no effective pathogenetic treatment at the moment.
Similar content being viewed by others
REFERENCES
Kozin M.S., Kulakova O.G., Kiselev I.S., Balanovsky O.P., Boyko A.N., Favorova O.O. 2018. Variants of mitochondrial genome and risk of multiple sclerosis development in Russians. Acta Naturae.10 (4), 79–86.
Sawcer S., Franklin R.J.M., Ban M. 2014. Multiple sclerosis genetics. Lancet Neurol.13 (7), 700–709.
Boyko A., Smirnova N., Petrov S., Gusev E. 2016. Epidemiology of MS in Russia: A historical review. Mult. Scler. Demyelinat. Disord. 1, 13.
Huitema M.J.D., Schenk G.J. 2018. Insights into the mechanisms that may clarify obesity as a risk factor for multiple sclerosis. Curr. Neurol. Neurosci. Rep. 18 (4), 18.
Sand I.B.K., Lublin F.D. 2013. Diagnosis and differential diagnosis of multiple sclerosis. Continuum (Minneap. Minn.). 19 (4), 922–943.
Koch M., Kingwell E., Rieckmann P., Tremlett H. 2009. The natural history of primary progressive multiple sclerosis. Neurology. 73 (23), 1996–2002.
IMSGC. 2019. Peripheral immune cells and microglia in susceptibility. Science. 365 (6460), eaav7188.
IMSGC. 2011. Genetic risk and a primary role for cell-mediated immune mechanisms in multiple sclerosis. Nature.476 (7359), 214–219.
Martinelli-Boneschi F., Esposito F., Brambilla P., Lindström E., Lavorgna G., Stankovich J., Rodegher M., Capra R., Ghezzi A., Coniglio G., Colombo B., Sorosina M., Martinelli V., Booth D., Oturai A.B., et al. 2012. A Genome-Wide Association Study in progressive multiple sclerosis. Mult. Scler. 18 (10), 1384–1394.
Kiselev I., Bashinskaya V., Baulina N., Kozin M., Popova E., Boyko A., Favorova O., Kulakova O. 2019. Genetic differences between primary progressive and relapsing-remitting multiple sclerosis: the impact of immune-related genes variability. Mult. Scler. Relat. Disord.29, 130–136.
Campbell G., Mahad D.J. 2018. Mitochondrial dysfunction and axon degeneration in progressive multiple sclerosis. FEBS Lett. 592 (7), 1113–1121.
Kozin M.S., Kulakova O.G., Favorova O.O. 2018. Involvement of mitochondria in neurodegeneration in multiple sclerosis. Biochemistry (Moscow). 83 (7), 813–830.
Tranah G.J., Santaniello A., Caillier S.J., D’Alfonso S., Boneschi F.M., Hauser S.L., Oksenberg J.R. 2015. Mitochondrial DNA sequence variation in multiple sclerosis. Neurology.85 (4), 325–330.
Hudson G., Gomez-Duran A., Wilson I.J., Chinnery P.F. 2014. Recent mitochondrial DNA mutations increase the risk of developing common late-onset human diseases. PLoS Genet.10 (5), e1004369.
Giacalone G., Clarelli F., Osiceanu A.M., Guaschino C., Brambilla P., Sorosina M., Liberatore G., Zauli A., Esposito F., Rodegher M., Ghezzi A., Galimberti D., Patti F., Barizzone N., Guerini F., et al. 2015. Analysis of genes, pathways and networks involved in disease severity and age at onset in primary-progressive multiple sclerosis. Mult. Scler.21 (11), 1431–1442.
Polman C.H., Reingold S.C., Banwell B., Clanet M., Cohen J.A., Filippi M., Fujihara K., Havrdova E., Hutchinson M., Kappos L., Lublin F.D., Montalban X., O’Connor P., Sandberg-Wollheim M., Thompson A.J., et al. 2011. Diagnostic criteria for multiple sclerosis: 2010 revisions to the McDonald criteria. Ann. Neurol.69 (2), 292–302.
van Oven M., Kayser M. 2009. Updated comprehensive phylogenetic tree of global human mitochondrial DNA variation. Hum. Mutat. 30 (2), E386–E394.
Zifa E., Daniil Z., Skoumi E., Stavrou M., Papadimitriou K., Terzenidou M., Kostikas K., Bagiatis V., Gourgoulianis K.I., Mamuris Z. 2012. Mitochondrial genetic background plays a role in increasing risk to asthma. Mol. Biol. Rep.39 (4), 4697–4708.
Gaur R.K. 2014. Amino acid frequency distribution among eukaryotic proteins. IIOAB J.5 (2), 6–11.
Pulkes T., Sweeney M.G., Hanna M.G. 2000. Increased risk of stroke in patients with the A12308G polymorphism in mitochondria. Lancet. 356 (9247), 2068–2069.
Schulmann A., Ryu E., Goncalves V., Rollins B., Christiansen M., Frye M.A., Biernacka J., Vawter M.P. 2019. Novel complex interactions between mitochondrial and nuclear DNA in schizophrenia and bipolar disorder. Mol. Neuropsychiatry. 5 (1), 13–27.
Covarrubias D., Bai R.K., Wong L.J.C., Leal S.M. 2008. Mitochondrial DNA variant interactions modify breast cancer risk. J. Hum. Genet.53 (10), 924–928.
van der Walt J.M, Dementieva Y.A., Martin E.R., Scott W.K., Nicodemus K.K., Kroner C.C., Welsh-Bohmer K.A., Saunders A.M., Roses A.D., Small G.W., Schmechel D.E., Doraiswamy P.M., Gilbert J.R., Haines J.L., Vance J.M., Pericak-Vance M.A. 2004. Analysis of European mitochondrial haplogroups with Alzheimer disease risk. Neurosci. Lett.365 (1), 28–32.
Rollins B., Martin M.V., Sequeira P.A., Moon E.A., Morgan L.Z., Watson S.J., Schatzberg A., Akil H., Myers R.M., Jones E.G., Wallace D.C., Bunney W.E., Vawter M.P. 2009. Mitochondrial variants in schizophrenia, bipolar disorder, and major depressive disorder. PLoS One. 4 (3), e4913.
Ban M., Elson J., Walton A., Turnbull D., Compston A., Chinnery P., Sawcer S. 2008. Investigation of the role of mitochondrial DNA in multiple sclerosis susceptibility. PLoS One. 3 (8), e2891.
Sun D., Wei Y., Zheng H.X., Jin L., Wang J. 2019. Contribution of mitochondrial DNA variation to chronic disease in East Asian populations. Front. Mol. Biosci. 6, 128.
Gómez-Durán A., Pacheu-Grau D., López-Gallardo E., Díez-Sánchez C., Montoya J., López-Pérez M.J., Ruiz-Pesini E. 2010. Unmasking the causes of multifactorial disorders: OXPHOS differences between mitochondrial haplogroups. Hum. Mol. Genet. 19 (17), 3343–3353.
Iwanowski P., Losy J. 2015. Immunological differences between classical phenothypes of multiple sclerosis. J. Neurol. Sci.349 (1–2), 10–14.
ACKNOWLEDGMENTS
The authors are deeply grateful to the neurologists N.N. Babicheva, L.I. Volkova, A.V. Karaeva, D.S. Kasatkin, D.S. Korobko, N.A. Malkova, S.A. Sivertseva, N.N. Spirina, N.N. Spirin, E.L. Turova, and F.A. Khabirov for participating in the formation of a group of PPMS patients.
Funding
This work was supported by the government contract AAAA-A19-119042590026-5.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest. The authors report no potential conflict of interest.
Statement of compliance with standards of research involving humans as subjects. All procedures performed in this work comply with the ethical standards of the institutional committee for research ethics and the 1964 Helsinki Declaration and its subsequent changes or comparable ethical standards. Written consent was obtained from all participants in the experiments.
Additional information
Abbreviations: CI, confidence interval; PCR, polymerase chain reaction; PCR-RFLP, PCR-restriction fragment length polymorphism analysis; OR, odds ratio; MS, multiple sclerosis; PPMS, primary progressive MS; RRMS, relapsing-remitting MS; GWAS, Genome Wide Association Study; SNP, single nucleotide polymorphism.
Rights and permissions
About this article
Cite this article
Kozin, M.S., Kulakova, O.G., Kiselev, I.S. et al. Variability of the Mitochondrial Genome and Development of the Primary Progressing form of Multiple Sclerosis. Mol Biol 54, 535–540 (2020). https://doi.org/10.1134/S0026893320040081
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0026893320040081