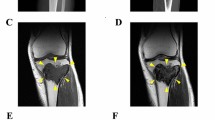
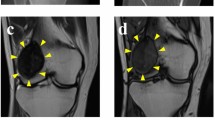
Abstract
Giant cell tumor of bone (GCTB) is a rare osteolytic bone tumor, accounting for approximately 5% of all primary bone tumors. GCTB is characterized by unique giant cells. It is also characterized by recurrent mutations in the histone tail of the histone variant H3.3, H3F3A, on chromosome 1, therapeutic implications of which have not been established yet. There are few effective standardized treatments for GCTB, and a novel therapy has long been required. Patient-derived cancer cells have facilitated the understanding of mechanisms underlying the etiology and progression of multiple cancers. Thus far, only 10 GCTB cell lines have been reported, and none of them are publicly available. The aim of this study was to develop an accessible patient-derived cell line of GCTB, which could be used as a screening tool for drug development. Here, we describe the establishment of a cell line, designated NCC-GCTB1-C1, from the primary tumor tissue of a male patient with GCTB on the right distal radius. NCC-GCTB1-C1 cells were maintained as a monolayer culture for over 23 passages for 7 months. These cells exhibited continuous growth, as well as spheroid formation and invasive ability. Using an oncology agent screen, we tested the effect of anticancer drugs on the proliferation of NCC-GCTB1-C1 cells. The cells displayed a remarkable response to romidepsin and vincristine. Thus, we established a novel GCTB cell line, NCC-GCTB1-C1, which could be a useful tool for studying GCTB tumorigenesis and the efficacy of anticancer drugs.




Similar content being viewed by others
References
Eckardt JJ, Grogan TJ. Giant cell tumor of bone. Clin Orthop Relat Res. 1986;1:45–58.
McGrath PJ. Giant-cell tumour of bone: an analysis of fifty-two cases. J Bone Joint Surg Br. 1972;54:216–29.
Bridge JA, Neff JR, Mouron BJ. Giant cell tumor of bone: chromosomal analysis of 48 specimens and review of the literature. Cancer Genet Cytogenet. 1992;58:2–13.
Mendenhall WM, Zlotecki RA, Scarborough MT, Gibbs CP, Mendenhall NP. Giant cell tumor of bone. Am J Clin Oncol. 2006;29:96–9.
Goldenberg RR, Campbell CJ, Bonfiglio M. Giant-cell tumor of bone: an analysis of two hundred and eighteen cases. J Bone Joint Surg Am. 1970;52:619–64.
Bertoni F, Present D, Sudanese A, Baldini N, Bacchini P, Campanacci M. Giant-cell tumor of bone with pulmonary metastases. Six case reports and a review of the literature. Clin Orthop Relat Res. 1988;1:275–85.
Siebenrock KA, Unni KK, Rock MG. Giant-cell tumour of bone metastasising to the lungs: a long-term follow-up. J Bone Joint Surg Br. 1998;80:43–7.
van der Heijden L, Dijkstra PD, van de Sande MA, et al. The clinical approach toward giant cell tumor of bone. Oncologist. 2014;19:550–61.
Behjati S, Tarpey PS, Presneau N, et al. Distinct H3F3A and H3F3B driver mutations define chondroblastoma and giant cell tumor of bone. Nat Genet. 2013;45:1479–82.
Presneau N, Baumhoer D, Behjati S, et al. Diagnostic value of H3F3A mutations in giant cell tumour of bone compared to osteoclast-rich mimics. J Pathol Clin Res. 2015;1:113–23.
Cleven AH, Höcker S, Briaire-de Bruijn I, Szuhai K, Cleton-Jansen AM, Bovée JV. Mutation analysis of H3F3A and H3F3B as a diagnostic tool for giant cell tumor of bone and chondroblastoma. Am J Surg Pathol. 2015;39:1576–83.
Szenker E, Ray-Gallet D, Almouzni G. The double face of the histone variant H3.3. Cell Res. 2011;21:421–34.
Huang C, Zhu B. H33 turnover: a mechanism to poise chromatin for transcription, or a response to open chromatin? BioEssays News Rev Mol Cell Dev Biol. 2014;36:579–84.
Errani C, Ruggieri P, Asenzio MA, et al. Giant cell tumor of the extremity: a review of 349 cases from a single institution. Cancer Treat Rev. 2010;36:1–7.
Boriani S, Sudanese A, Baldini N, Picci P. Sarcomatous degeneration of giant cell tumours. Ital J Orthop Traumatol. 1986;12:191–9.
Wu PF, Tang JY, Li KH. RANK pathway in giant cell tumor of bone: pathogenesis and therapeutic aspects. Tumour Biol. 2015;36:495–501.
Highlights of prescribing information of XGEVA (denosumab). [updated; cited 20 February 2020]. Available from: https://www.accessdata.fda.gov/drugsatfda_docs/label/2013/125320s094lbl.pdf
Tsukamoto S, Tanaka Y, Mavrogenis AF, Kido A, Kawaguchi M, Errani C. Is treatment with denosumab associated with local recurrence in patients with giant cell tumor of bone treated with curettage? A systematic review. Clin Orthop Relat Res. 2020;478:1076–85.
Turcotte RE. Giant cell tumor of bone. Orthoped Clin N Am. 2006;37:35–51.
Tse LF, Wong KC, Kumta SM, Huang L, Chow TC, Griffith JF. Bisphosphonates reduce local recurrence in extremity giant cell tumor of bone: a case-control study. Bone. 2008;42:68–73.
Balke M, Campanacci L, Gebert C, et al. Bisphosphonate treatment of aggressive primary, recurrent and metastatic Giant Cell Tumour of Bone. BMC Cancer. 2010;10:462.
Nishisho T, Hanaoka N, Endo K, Takahashi M, Yasui N. Locally administered zoledronic Acid therapy for giant cell tumor of bone. Orthopedics. 2011;34:e312–e315315.
Kaban LB, Mulliken JB, Ezekowitz RA, Ebb D, Smith PS, Folkman J. Antiangiogenic therapy of a recurrent giant cell tumor of the mandible with interferon alfa-2a. Pediatrics. 1999;103:1145–9.
Kaban LB, Troulis MJ, Ebb D, August M, Hornicek FJ, Dodson TB. Antiangiogenic therapy with interferon alpha for giant cell lesions of the jaws. J Oral Maxillofac Surg. 2002;60:1103–11.
Sharma SV, Haber DA, Settleman J. Cell line-based platforms to evaluate the therapeutic efficacy of candidate anticancer agents. Nat Rev Cancer. 2010;10:241–53.
Lim J, Park JH, Baude A, et al. The histone variant H3.3 G34W substitution in giant cell tumor of the bone link chromatin and RNA processing. Sci Rep. 2017;7:13459.
Fellenberg J, Sähr H, Mancarella D, et al. Knock-down of oncohistone H3F3A-G34W counteracts the neoplastic phenotype of giant cell tumor of bone derived stromal cells. Cancer Lett. 2019;448:61–9.
Bairoch A. The cellosaurus, a cell-line knowledge resource. J Biomol Tech. 2018;29:25–38.
Yoshimatsu Y, Noguchi R, Tsuchiya R, et al. Establishment and characterization of NCC-CDS2-C1: a novel patient-derived cell line of CIC-DUX4 sarcoma. Hum Cell. 2020;33:427–36.
Capes-Davis A, Reid YA, Kline MC, et al. Match criteria for human cell line authentication: where do we draw the line? Int J Cancer. 2013;132:2510–9.
Shoemaker RH. The NCI60 human tumour cell line anticancer drug screen. Nat Rev Cancer. 2006;6:813–23.
Barretina J, Caponigro G, Stransky N, et al. The Cancer Cell Line Encyclopedia enables predictive modelling of anticancer drug sensitivity. Nature. 2012;483:603–7.
Garnett MJ, Edelman EJ, Heidorn SJ, et al. Systematic identification of genomic markers of drug sensitivity in cancer cells. Nature. 2012;483:570–5.
Tsherniak A, Vazquez F, Montgomery PG, et al. Defining a cancer dependency map. Cell. 2017;170:564–76.
Voissiere A, Jouberton E, Maubert E, et al. Development and characterization of a human three-dimensional chondrosarcoma culture for in vitro drug testing. PLoS ONE. 2017;12:e0181340.
Prince HM, Dickinson M. Romidepsin for cutaneous T-cell lymphoma. Clin Cancer Res. 2012;18:3509–15.
Carbone PP, Bono V, Frei E 3rd, Brindley CO. Clinical studies with vincristine. Blood. 1963;21:640–7.
Acknowledgements
We appreciate the technical support provided by Miss Yu Kuwata (Division of Rare Cancer Research, National Cancer Center). We would like to thank the Tochigi Cancer Center Operating Room Nurse Team and the Secretary of the Medical Office for their assistance in processing and transporting the samples. We would like to thank Editage (https://www.editage.jp) for providing English-language editing service and for their constructive comments on the manuscript.
Funding
This research was supported by the Japan Agency for Medical Research and Development Grant (20ck0106537h0001), titled “Study to Overcome the Limits of Cancer Genome-based Medicine Using Patient-derived ‘Rare Cancer’ Model”.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This study was approved by the ethics committee of the Tochigi Cancer Center and the National Cancer Center, and written informed consent was obtained from the patient.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
13577_2020_415_MOESM1_ESM.tif
Supplementary Figure 1 Single tandem repeat patterns in sequencing data from the original tumor and NCC-GCTB1-C1 cells (TIF 307 kb)
13577_2020_415_MOESM2_ESM.tif
Supplementary Figure 2 Images of invading NCC-GCTB1-C1 cells on the membrane of the upper chamber in transwell assays (TIF 535 kb)
13577_2020_415_MOESM3_ESM.tif
Supplementary Figure 3 Growth curves of agents that showed relatively higher inhibition of NCC-GCTB1-C1 cell proliferation (TIF 753 kb)
Rights and permissions
About this article
Cite this article
Noguchi, R., Yoshimatsu, Y., Ono, T. et al. Establishment and characterization of NCC-GCTB1-C1: a novel patient-derived cancer cell line of giant cell tumor of bone. Human Cell 33, 1321–1328 (2020). https://doi.org/10.1007/s13577-020-00415-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13577-020-00415-w