Abstract
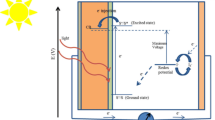
The present work is involved in the computational and experimental studies of organic dyes and their applications as dye-sensitized solar cells (DSSCs). This comprised the study of three hydrazone-based sensitizers (E)-2-cyano-N′-((2-hydroxynaphthalen-1-yl)methylene)acetohydrazide (CHMA), (E)-2-cyano-N′-(4-(dimethylamino)benzylidene)acetohydrazide (CDBA), (E)-N′-(anthracen-9-ylmethylene)-2-cyanoacetohydrazide (AMCH) that have been prepared and confirmed by means of several analytical procedures like Fourier transform infrared, UV–visible and nuclear magnetic resonance techniques to investigate the best possible selection for DSSCs by computational and experimental techniques. The computational methods are applied to optimize the structures of prepared organic dyes via density functional theory (DFT) method at B3LYP/6-311G(p,d) level of theory. The time-dependent DFT (TD-B3LYP/6-311G**) was used with and without solvent to find out the absorption spectra and matched with the experimental data and the electro-optical and reorganization energies of prepared dyes were further investigated. The results revealed that the prepared dyes would be better sensitizers for DSSCs because of small highest-occupied molecular orbital–lowest-unoccupied molecular orbital energy gap. Moreover, on the basis of the above results, we fabricated the devices via the doctor blade method to study the photovoltaic performance with the prepared dyes (CHMA, CDBA and AMCH). The dye AMCH exhibited the maximum efficiency with commercial TiO2.







Similar content being viewed by others
References
Potocnik J 2007 Science 315 810
Oregan B and Grätzel M 1991 Nature 353 737
Wu Y, Zhu W-H, Zakeeruddin S M and Grätzel M 2015 ACS Appl. Mater. Interfaces 7 9307
Ye M, Wen X, Wang M, Iocozzia J, Zhang N, Lin C et al 2015 Mater. Today 18 155
Wu M and Ma T 2014 J. Phys. Chem. C 118 16727
Gong J, Sumathy K, Qiao Q and Zhou Z 2017 Renew. Sust. Energ. Rev. 68 234
Hagfeldt A, Boschloo G, Sun L, Kloo L and Pettersson H 2010 Chem. Rev. 110 6595
Ji Z, Natu G and Wu Y 2013 ACS Appl. Mater. Interfaces 5 8641
Ngo K T, Lee N A, Pinnace S D and Rochford J 2017 Chem. Eur. J. 23 7621
Liang M and Chen J 2013 Chem. Soc. Rev. 42 3453
Urbani M, Grätzel M, Nazeeruddin M K and Torres T 2014 Chem. Rev. 114 12330
Giribabu L and Kanaparthi R K 2013 Curr. Sci. 104 847
Wang C L, Lan C M, Hong S H, Wang Y F, Pan T Y, Chang C W et al 2012 Energy Environ. Sci. 5 6933
Green M A 2001 Prog. Photovoltics 9 123
Calogero G, Bartolotta A, Marco G D, Carlo A D and Bonaccorso F 2015 Chem. Soc. Rev. 44 3244
Yao Z, Zhang M, Wu H, Yang L, Li R and Wang P 2015 J. Am. Chem. Soc. 137 3799
Calogero G, Yum J-H, Sinopoli A, Marco G D, Grätzel M and Nazeeruddin M K 2012 Sol. Energy 86 1563
Calogero G, Sinopoli A, Citro I, Marco G D, Petrov V, Diniz A M et al 2013 Photochem. Photobiol. Sci. 12 883
Koumura N, Wang Z-S, Mori S, Miyashita M, Suzuki E and Hara K 2006 J. Am. Chem. Soc. 128 14256
Mishra A, Fischer M K R and Bäuerle P 2009 Angew. Chem. 48 2474
Feldt S M, Gibson E A, Gabrielsson E, Sun L, Boschloo G and Hagfeldt A 2010 J. Am. Chem. Soc. 132 16714
Yella A, Lee H-W, Tsao H N, Yi C, Chandiran A K, Nazeeruddin M K et al 2011 Science 334 629
Gómez-Ortíz N, Vázquez-Maldonado I, Pérez-Espadas A, Mena-Rejón G, Azamar-Barrios J and Oskam G 2010 Sol. Energy Mater. Sol. Cells 94 40
Wongcharee K, Meeyoo V and Chavadej S 2007 Sol. Energy Mater. Sol. Cells 91 566
Li S-L, Jiang K-J, Shao K-F and Yang L-M 2006 Chem. Commun. 2792
Hara K, Sato T, Katoh R, Furube A, Yoshihara T, Murai M et al 2005 Adv. Funct. Mater. 15 246
Lygaitis R, Getautis V and Grazulevicius J V 2008 Chem. Soc. Rev. 37 770
Kim S and Yoon J-Y 2004 Sci. Synth. 27 671
Uribe-Romo F J, Doonan C J, Furukawa H, Oisaki K and Yaghi O M 2011 J. Am. Chem. Soc. 133 11478
Ulrich S and Dumy P 2014 Chem. Commun. 50 5810
Raue R, Brack A and Lange K H 1991 Angew. Chem. 30 1643
Laurinaviciute R, Mimaite V, Ostrauskaite J, Grazulevicius J V and Jankauskas V 2014 Synth. Met. 197 1
Michaleviciute A, Buika G, Grazulevicius J V, Tran-Van F, Chevrot C and Jankauskas V 2007 Mol. Cryst. Liq. Cryst. 468 107
Shen P, Liu X, Jiang S, Huang Y, Yi L, Zhao B et al 2011 Org. Electron. 12 1992
Shen P, Liu X, Jiang S, Wang L, Yi L, Ye D et al 2012 Dyes Pigm. 92 1042
Aich R, Tran-Van F, Goubard F, Beouch L, Michaleviciute A, Grazulevicius J et al 2008 Thin Solid Films 516 7260
Li L-L and Diau E W-G 2013 Chem. Soc. Rev. 42 291
Birel Ö, Nadeem S and Duman H 2017 J. Fluoresc. 27 1075
Ladomenou K, Kitsopoulos T N, Sharma G D and Coutsolelos A G 2014 RSC Adv. 4 21379
Salzner U, Lagowski J B, Pickup P G and Poirier R A 1998 Synth. Met. 96 177
Koch W, Holthausen M C and Kaupp M 2001 Angew. Chem. 113 989
Irfan A and Al-Sehemi A G 2012 J. Chem. Soc. Pak. 34 350
Nazeeruddin M K, De Angelis F, Fantacci S, Selloni A, Viscardi G, Liska P et al 2005 J. Am. Chem. Soc. 127 16835
Liu Z 2008 J. Mol. Struct. Theochem. 862 44
Sánchez-Carrera R S, Coropceanu V, da Silva Filho D A, Friedlein R, Osikowicz W, Murdey R et al 2006 J. Phys. Chem. B 110 18904
Abbas G, Irfan A, Mir M and Khan A F 2013 J. Mol. Struct. 1050 10
Irfan A, Al-Sehemi A G, Muhammad S, Chaudhry A R, Al-Assiri M S, Jin R et al 2015 C. R. Chim. 18 1289
Al-Sehemi A G, Irfan A and Assiri A M 2012 Theor. Chem. Acc. 131 1199
Becke A D 1993 J. Chem. Phys. 98 5648
Lee C, Yang W and Parr R G 1988 Phys. Rev. B: Condens. Matter 37 785
Stephens P J, Devlin F J, Chabalowski C F and Frisch M J 1994 J. Phys. Chem. 98 11623
Lynch B J, Fast P L, Harris M and Truhlar D G 2000 J. Phys. Chem. A 104 4811
Perdew J P, Chevary J A, Vosko S H, Jackson K A, Pederson M R, Singh D J et al 1992 Phys. Rev. B: Condens. Matter 46 6671
Walsh P J, Gordon K C, Officer D L and Campbell W M 2006 J. Mol. Struct. Theochem. 759 17
Cleland D M, Gordon K C, Officer D L, Wagner P and Walsh P J 2009 Spectrochim. Acta Part A 74 931
Zhang C R, Liang W Z, Chen H S, Chen Y H, Wei Z Q and Wu Y Z 2008 J. Mol. Struct. Theochem. 862 98
Sun J, Song J, Zhao Y and Liang W Z 2007 J. Chem. Phys. 127 234107
Matthews D, Infelta P and Grätzel M 1996 Sol. Energy Mater. Sol. Cells 44 119
Al-Sehemi A G, Irfan A and Assiri M A 2014 Chin. Chem. Lett. 25 609
Frisch M J, Trucks G W, Schlegel H B, Scuseria G E, Robb M A, Cheeseman J R et al 2009 Gaussian 09 revision D.01, Wallingford, CT: Gaussian, Inc.
Prima E C, Hidayat N N, Yuliarto B and Dipojono H K 2017 Spectrochim. Acta Part A 171 112
Ekanayake P, Kooh M R, Kumara N T, Lim A, Petra M I, Voo N Y et al 2013 Chem. Phys. Lett. 585 121
Liang M, Xu W, Cai F, Chen P, Peng B, Chen J et al 2007 J. Phys. Chem. C 111 4465
Guo M, Diao P, Ren Y-J, Meng F, Tian H and Cai S-M 2005 Sol. Energy Mater. Sol. Cells 88 23
Han D, Zhang G, Chai H, Zhang X and Zhao L 2013 J. Lumin. 138 223
Irfan A, Al-Sehemi A G and Al-Assiri M S 2013 J. Mol. Graph. Model 44 168
Palomares E, Clifford J N, Haque S A, Lutz T and Durrant J R 2003 J. Am. Chem. Soc. 125 475
Al-Sehemi A G, Irfan A, Al-Melfi M A M, Al-Ghamdi A A and Shalaan E 2014 J. Photochem. Photobiol. A 292 1
Acknowledgement
We thank King Abdul Aziz City of Science and Technology (KACST) for their financial support to Shuhrah Ali S Allami with Grant Number 1-17-01-010-0018.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
AL-SEHEMI, A.G., ALLAMI, S.A.S. & KALAM, A. Design and synthesis of organic dyes with various donor groups: promising dyes for dye-sensitized solar cells. Bull Mater Sci 43, 224 (2020). https://doi.org/10.1007/s12034-020-02198-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12034-020-02198-0