Abstract
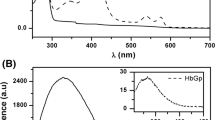
The aim of the present work was the biophysical characterization of the Amynthas gracilis hemoglobin (HbAg). The oxy-HbAg optical absorption data, with Soret and Q bands centered at 415, 540 and 575 nm, were stable and unchanged at pH 7.0. An increase in pH promotes decrease in the intensity in the optical absorption bands, suggesting an oligomeric dissociation and partial oxidation. Identical stability at pH 7.0 was observed in DLS results that presented a hydrodynamic diameter of 28 nm, characteristic of the whole oligomer. DLS shows that HbAg undergoes oligomeric dissociation and an aggregation/denaturation process that corroborates spectroscopic data. Our results showed that the monomer d presents four isoforms with molecular mass (MM) ranging from 16,244 to 16,855 Da; the trimer subunit presents two isoforms, (abc)1 and (abc)2, with MM of 51,415 ± 20 Da and 51,610 ± 14 Da, respectively, and a less intense species, at 67,793 Da, assigned to the tetramer abcd. Monomeric chains a, obtained from reduction of the disulfide-bonded trimer abc, present four isoforms with MM 17,015 Da, 17,061 Da, 17,138 Da and 17,259 Da. DLS and LSI revealed an isoeletric point (pI) of oxy-HbAg of 6.0 ± 0.3 and 5.5, respectively. Data analysis by IEF–SDS–PAGE revealed that the pI of oxy-HbAg is 6.11, correlating with DLS and LSI data. These studies indicate that oxy-HbAg is very stable, at pH 7.0, and has differing properties from orthologous giant hemoglobins.







Similar content being viewed by others
References
Bachega JFR, Maluf FV, Andi B, Pereira HD, Carazzollea MF, Orville AM, Tabak M, Brandao-Neto J, Garratt RC, Reboredo EH (2015) The structure of the giant haemoglobin from Glossoscolex paulistus. Acta Crystallogr Sect D Struct Biol 71:1257–1271. https://doi.org/10.1107/S1399004715005453
Bradford MM (1976) A rapid and sensitive method for quantification of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1006/abio.1976.9999
Carvalho FAO, Carvalho JWP, Santiago PS, Tabak M (2011a) Further characterization of the subunits of the giant extracellular hemoglobin of Glossoscolex paulistus (HbGp) by SDS–PAGE electrophoresis and MALDI-TOF–MS. Process Biochem 46:2144–2151. https://doi.org/10.1016/j.procbio.2011.08.013
Carvalho FAO, Santiago PS, Borges JC, Tabak M (2011b) Molecular masses and sedimentation coefficients of extracellular hemoglobin of Glossoscolex paulistus: alkaline oligomeric dissociation. Int J Biol Macromol 48:183–193. https://doi.org/10.1016/j.ijbiomac.2010.11.002
Carvalho FAO, Carvalho JWP, Alves FR, Tabak M (2013a) PH effect upon HbGp oligomeric stability: characterization of the dissociated species by AUC and DLS studies. Int J Biol Macromol 59:333–341. https://doi.org/10.1016/j.ijbiomac.2013.04.070
Carvalho JWP, Alves FR, Batista T, Carvalho FAO, Santiago PS, Tabak M (2013b) Sodium dodecyl sulfate (SDS) effect on the thermal stability of oxy-HbGp: dynamic light scattering (DLS) and small angle X-ray scattering (SAXS) studies. Colloids Surf B 111:561–570. https://doi.org/10.1016/j.colsurfb.2013.06.050
Carvalho JWP, Carvalho FAO, Santiago PS, Tabak M (2013c) Thermal denaturation and aggregation of hemoglobin of Glossoscolex paulistus in acid and neutral media. Int J Biol Macromol 54:109–118. https://doi.org/10.1016/j.ijbiomac.2012.11.022
Carvalho FAO, Carvalho JWP, Biazin E, Santiago PS, Tabak M (2014a) Characterization of Rhinodrilus alatus hemoglobin (HbRa) and its subunits: evidence for strong interaction with cationic surfactants DTAB and CTAC. Comp Biochem Physiol B 167:23–29. https://doi.org/10.1016/j.cbpb.2013.09.007
Carvalho JWP, Carvalho FAO, Batista T, Santiago PS, Tabak M (2014b) Cetyltrimethylammonium chloride (CTAC) effect on the thermal stability of oxy-HbGp: dynamic light scattering (DLS) and small angle X-ray scattering (SAXS) studies. Colloids Surf B 118:14–24. https://doi.org/10.1016/j.colsurfb.2014.03.021
Carvalho JWP, Carvalho FAO, Santiago PS, Tabak M (2016) Thermal denaturation and aggregation of hemoglobin of Rhinodrilus alatus (HbRa): DLS and SAXS studies. Eur Biophys J 45:549–563. https://doi.org/10.1007/s00249-016-1121-6
James SW, Shih HT, Chang HW (2007) Seven new species of Amynthas (Clitellata: Megascolecidae) and new earthworm records from Taiwan. J Nat Hist 39:1007–1028. https://doi.org/10.1080/00222930400001434
Lamy JN, Green BN, Toulmond A, Wall JS, Weber RE, Vinogradov SN (1996) Giant hexagonal bilayer hemoglobins. Chem Rev 96:3113–3124. https://doi.org/10.1021/cr9600058
Martin PD, Kuchumov AR, Green BN, Oliver RWA, Braswell EH, Wall JS, Vinogradov SN (1996) Mass spectrometric composition and molecular mass of Lumbricus terrestris hemoglobin: a refined model of its quaternary structure. J Mol Biol 255:154–169. https://doi.org/10.1006/jmbi.1996.0013
Muzzelo C, Neely C, Shah P, Abdulmalik O, Elmer J (2018) Prolonging the shelf life of Lumbricus terrestris erythrocruorin for use as a novel blood substitute. Artif Cells Nanomed Biotechnol 46:39–46. https://doi.org/10.1080/21691401.2017
Oliveira MS, Moreira LM, Tabak M (2007) Partial characterization of giant extracellular hemoglobin of Glossoscolex paulistus: a MALDI-TOF–MS study. Int J Biol Macromol 40:429–436. https://doi.org/10.1016/j.ijbiomac.2006.10.005
Otzen D (2011) Protein–surfactant interactions: a tale of many states. Biochim Biophys Acta Proteins Proteomics 1814:562–591. https://doi.org/10.1016/j.bbapap.2011.03.003
Ownby DW, Zhu H, Schneider K, Beavis RC, Chait BT, Riggs AF (1993) The extracellular hemoglobin of the earthworm, Lumbricus terrestris. Determination of subunit stoichiometry. J Biol Chem 268:13539–13547
Poli AL, Moreira LM, Imasato H (2011) Autoxidation of giant extracellular hemoglobin of Glossoscolex paulistus: Molecular mechanism and oligomeric implications. Spectrochim Acta Part A Mol Biomol Spectrosc 82:306–315. https://doi.org/10.1016/j.saa.2011.07.053
Riccio A, Vitagliano L, Prisco G, Zagari A, Mazzarella L (2002) The crystal structure of a tetrameric hemoglobin in a partial hemichrome state. PNAS 23:9801–9806. https://doi.org/10.1073/pnas.132182099
Rousselot M, Delpy E, La Rochelle CD, Lagente V, Pirow R, Rees J, Hagege A, Le Guen D, Hourdez S, Zal F (2006) Arenicola marina extracellular hemoglobin: a new promising blood substitute. Biotechnol J 1:333–345. https://doi.org/10.1002/biot.200500049
Royer WE, Sharma H, Strand K, Knapp JE, Bhyravbhatla B (2006) Lumbricus erythrocruorin at 3.5 Å resolution: architecture of a megadalton respiratory complex. Structure 14:1167–1177. https://doi.org/10.1016/j.str.2006.05.011
Royer WE, Omartian MN, Knapp JE (2007) Low resolution crystal structure of Arenicola erythrocruorin: influence of coiled coils on the architecture of a megadalton respiratory protein. J Mol Biol 365:226–236. https://doi.org/10.1016/j.jmb.2006.10.016
Santiago PS, Moreira LM, de Almeida EV, Tabak M (2007) Giant extracellular Glossoscolex paulistus hemoglobin (HbGp) upon interaction with cethyltrimethylammonium chloride (CTAC) and sodium dodecyl sulphate (SDS) surfactants: dissociation of oligomeric structure and autoxidation. Biochim Biophys Acta-Gen Subj 1770:506–517. https://doi.org/10.1016/j.bbagen.2006.11.005
Santiago PS, Moura F, Moreira LM, Domingues MM, Santos NC, Tabak M (2008) Dynamic light scattering and optical absorption spectroscopy study of pH and temperature stabilities of the extracellular hemoglobin of Glossoscolex paulistus. Biophys J 94:2228–2240. https://doi.org/10.1529/biophysj.107.116780
Santiago PS, Carvalho FAO, Domingues MM, Carvalho JWP, Santos NC, Tabak M (2010) Isoelectric point determination for Glossoscolex paulistus extracellular hemoglobin: oligomeric stability in acidic pH and relevance to protein–surfactant interactions. Langmuir 26:9794–9801. https://doi.org/10.1021/la100060p
Stetefeld J, McKenna SA, Patel TR (2016) Dynamic light scattering: a practical guide and applications in biomedical sciences. Biophys Rev 8:409–427. https://doi.org/10.1007/s12551-016-0218-6
Tabak M, Carvalho FAO, Carvalho JWP, Bachega JFR, Santiago PS (2012) Recent new characterizations on the giant extracellular hemoglobin of Glossoscolex paulistus and some other giant hemoglobins from different worms. Stoichiom Res-Importance Quant Biomed 1:337–356. https://doi.org/10.5772/33962
Vinogradov SN (2004) The stoichiometry of the four linker subunits of Lumbricus terrestris hemoglobin suggests an asymmetric distribution. Micron 35:127–129. https://doi.org/10.1016/j.micron.2003.10.041
Voet D, Voet JG (2010) Biochemistry, 4th Edition, Kindle Edition, ISBN: 978-0-470-57095-1
Weber RE, Vinogradov SN (2001) Nonvertebrate hemoglobins: functions and molecular adaptations. Physiol Revi 81:569–628. https://doi.org/10.1152/physrev.2001.81.2.569
Zal F, Green BN, Lallier FH, Vinogradov SN, Toulmond A (1997) Quaternary structure of the extracellular haemoglobin of the lugorm Arenicola marina: a multi-angle-laser-light-scattering and electrospray-ionisation–mass-spectrometry analysis. Eur J Biochem 243:85–92. https://doi.org/10.1111/j.1432-1033.1997.85_1a.x
Acknowledgments
The authors are indebted to the Brazilian agencies FAPESP, CNPq, and CAPES for partial financial support. J. B. Oliveira is a recipient of a master degree grant from FAPESP (2016/10884-1). C. O. Souza is grateful for a doctoral grant from CAPES. I. Sebastião is a recipient of a doctoral Grant from CNPq. P. S. Santiago is grateful to FAPESP for a research Project (2015/11447-1).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Oliveira, J.B.S., Ramos, L., Souza, C.O. et al. Initial biophysical characterization of Amynthas gracilis giant extracellular hemoglobin (HbAg). Eur Biophys J 49, 473–484 (2020). https://doi.org/10.1007/s00249-020-01455-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00249-020-01455-8