Abstract
Natural pigments are derived from sources such as plant, animal, insects, and fungi. The anthraquinones are natural pigments that have pharmacological activities. They have applications in several domains such as the pharmaceutical, cosmetic and textile industries. Anthraquinones occur naturally in many plants including Cassia tora of the Fabaceae family. The present study focused on the production of pigments (anthraquinones) from callus of Cassia tora cultured on medium supplemented with 6-benzylaminopurine and thidiazuron. The elicitor, salicylic acid, increased anthraquinone production in callus cultures whereas ethrel had minimum effect. Anthraquinone production increased twofold in callus inoculated in medium containing 10 mM salicylate. The yield obtained was 3709 ± 269.65 mg/kg dry weight of callus in Fraction B. The sun protection factor of the callus extracts was also assessed. The maximum sun protection factor observed was 38.
Key message
Growth analysis of Cassia tora callus, modulating callus growth and pigment (anthraquinones) production with the elicitors Ethrel and salicylic acid. Potential application of callus extract as Sun protectant.





Similar content being viewed by others
Data availability
All data generated or analysed during this study are included in this article and supplementary material.
References
Abreu Dutra E, Gonçalves A, da Costa OD, Rosa Maria Kedor-Hackmann E, M (2004) Determination of sun protection factor (SPF) of sunscreens by ultraviolet spectrophotometry. Rev Bras Ciências Farm Braz J Pharm Sci 40:381–385Inês Rocha Miritello Santoro
Anjusha S, Gangaprasad A (2017) Callus culture and in vitro production of anthraquinone in Gynochthodes umbellata (L.) Razafim. & B. Bremer (Rubiaceae). Ind Crops Prod 95:608–614. https://doi.org/10.1016/j.indcrop.2016.11.021
Bakoriya R, Soni KK, Thomas T (2015) Isolation and structural elucidation of bioactive compound from ethanolic extract of Cassia tora leaves spasmolytic activity of a herbal drug isolated from tephrosia purpurea in guinea pigs. Asian J Chem 10:3749–3752. https://doi.org/10.14233/ajchem.2015.18952
Bulgakov VP, Tchernoded GK, Mischenko NP et al (2002) Effect of salicylic acid, methyl jasmonate, ethephon and cantharidin on anthraquinone production by Rubia cordifolia callus cultures transformed with the rolB and rolC genes. J Biotechnol 97:213–221. https://doi.org/10.1016/S0168-1656(02)00067-6
Dave H, Ledwani L (2012) A review on anthraquinones isolated from Cassia species and their applications. Indian J Nat Prod Resour 3:291–319
Delgado-Vargas F, Jiménez AR, Paredes-López O (2000) Natural pigments: carotenoids, anthocyanins, and betalains—characteristics, biosynthesis, processing, and stability. Crit Rev Food Sci Nutr 40:173–289. https://doi.org/10.1080/10408690091189257
Demirezer L, Uzun M (2016) Determination of sun protection factor (SPF) of Rumex crispus and main anthraquinones. Planta Med 81:S1–S381. https://doi.org/10.1055/s-0036-1596459
Fortini EA, Batista DS, de Castro KM et al (2020) Photoperiod modulates growth and pigments and 20-hydroxyecdysone accumulation in Brazilian ginseng [Pfaffia glomerata (Spreng.) Pedersen] grown in vitro. Plant Cell Tissue Organ Cult. https://doi.org/10.1007/s11240-020-01886-3
Gaikwad SA (2018) Phytochemical investigation of bioactive Emodin and quercetin in Cassia fistula and Cassia tora plant parts by HPTLC. J Pharmacogn Phytochem 7:892–897
Godard S, Slacanin I, Viret O, Gindro K (2009) Induction of defence mechanisms in grapevine leaves by emodin- and anthraquinone-rich plant extracts and their conferred resistance to downy mildew. Plant Physiol Biochem 47:827–837. https://doi.org/10.1016/J.PLAPHY.2009.04.003
Gupta P, Sharma S, Saxena S (2015) Biomass yield and steviol glycoside production in callus and suspension culture of Stevia rebaudiana treated with proline and polyethylene glycol. Appl Biochem Biotechnol 176:863–874. https://doi.org/10.1007/s12010-015-1616-0
Han L, Piao XC, Jiang J et al (2019) A high production of flavonoids and anthraquinones via adventitious root culture of Oplopanax elatus and evaluating antioxidant activity. Plant Cell Tissue Organ Cult 137:173–179. https://doi.org/10.1007/s11240-018-01543-w
Han YS, Van Der Heijden R, Verpoorte R (2001) Biosynthesis of anthraquinones in cell cultures of the Rubiaceae. Plant Cell Tissue Organ Cult 67:201–220. https://doi.org/10.1023/A:1012758922713
Jang DS, Lee GY, Kim YS et al (2007) Anthraquinones from the seeds of Cassia tora with inhibitory activity on protein glycation and aldose reductase. Biol Pharm Bull 30:2207–2210. https://doi.org/10.1248/bpb.30.2207
Kang S-H, Lee W, Lee C et al (2020) De novo transcriptome sequence of Senna tora provides insights into anthraquinone biosynthesis. PLoS ONE 15:e0225564. https://doi.org/10.1371/journal.pone.0225564
Kim YM, Lee CH, Kim HG, Lee HS (2004) Anthraquinones isolated from Cassia tora (Leguminosae) seed show an antifungal property against phytopathogenic fungi. J Agric Food Chem 52:6096–6100. https://doi.org/10.1021/jf049379p
Krishnan SRRS, Siril EA (2018) Elicitor mediated adventitious root culture for the large-scale production of anthraquinones from Oldenlandia umbellata L. Ind Crops Prod 114:173–179. https://doi.org/10.1016/j.indcrop.2018.01.069
Lee GY, Cho BO, Shin JY et al (2018) Tyrosinase inhibitory components from the seeds of Cassia tora. Arch Pharm Res 41:490–496. https://doi.org/10.1007/s12272-018-1032-4
Lee GY, Kim JH, Choi SK, Kim YH (2015) Constituents of the seeds of Cassia tora with inhibitory activity on soluble expoxide hydrolease. Bioorganic Med Chem Lett 25:5097–5101. https://doi.org/10.1016/j.bmcl.2015.10.014
Lee NH, Lee SM, Song DH et al (2013a) Antimicrobial effect of emodin isolated from Cassia tora Linn. Seeds against food-borne bacteria. J Appl Biol Chem 56:187–189. https://doi.org/10.3839/jabc.2013.030
Lee YH, Do KH (2004) Dyeing properties and colour fastness of cotton and silk fabrics dyed with Cassia tora L. extract. Fibers Polym 5:303–308. https://doi.org/10.1007/BF02875529
Lee YS, Ju HK, Kim YJ et al (2013b) Enhancement of anti-inflammatory activity of Aloe vera adventitious root extracts through the alteration of primary and secondary metabolites via salicylic acid elicitation. PLoS ONE 8:e82479. https://doi.org/10.1371/journal.pone.0082479
Maity TK, Dinda SC (2003) Purgative activity of cassia tora leaf extract and isolated aloeemodin. Indian J Nat Prod Resour 65:93–95
Malsawmtluangi C, Nath DK, Jamatia I et al (2013) Determination of sun protection factor (SPF) number of some aqueous herbal extracts. J Appl Pharm Sci 3:150–151. https://doi.org/10.7324/JAPS.2013.3925
Mazumder MU, Das K, Choudhury AD, Khazeo P (2018) Determination of sun protection factor (SPF) number of some hydroalcoholic vegetable extracts. PharmaTutor 6:41–45. https://doi.org/10.29161/pt.v6.i12.2018.41
Mbanga L, Mulenga M, Mpiana PT et al (2014) Determination of sun protection factor ( SPF ) of some body creams and lotions marketed in Kinshasa by ultraviolet spectrophotometry. Int J Adv Res Chem Sci 1:7–13
Mbatchou VC, Tchouassi DP, Dickson RA et al (2017) Mosquito larvicidal activity of Cassia tora seed extract and its key anthraquinones aurantio-obtusin and obtusin. Parasites and Vectors 10:562. https://doi.org/10.1186/s13071-017-2512-y
Mischenko NP, Fedoreyev SA, Glazunov VP et al (1999) Anthraquinone production by callus cultures of Rubia cordifolia. Fitoterapia 70:552–557. https://doi.org/10.1016/S0367-326X(99)00085-4
Mishchenko NP, Fedoreev SA, Bryukhanov VM et al (2007) Chemical composition and pharmacological activity of anthraquinones from Rubia cordifolia cell culture. Pharm Chem J 41:605–609. https://doi.org/10.1007/s11094-008-0021-1
Naik PM, Al-Khayri JM (2016) Abiotic and biotic elicitors—role in secondary metabolites production through in vitro culture of medicinal plants. In: Shanker AK, Shanker C (eds) Abiotic and Biotic Stress in Plants—Recent Advances and Future Perspectives. InTech, London, pp 247–277
Ochoa-Villarreal M, Howat S, Hong S et al (2016) Plant cell culture strategies for the production of natural products. BMB Rep 49:149–158. https://doi.org/10.5483/bmbrep.2016.49.3.264
Orbán N, Boldizsár I, Szucs Z, Dános B (2008) Influence of different elicitors on the synthesis of anthraquinone derivatives in Rubia tinctorum L. cell suspension cultures. Dye Pigment 77:249–257. https://doi.org/10.1016/j.dyepig.2007.03.015
Perassolo M, Smith ME, Giulietti AM, Rodríguez Talou J (2016) Synergistic effect of methyl jasmonate and cyclodextrins on anthraquinone accumulation in cell suspension cultures of Morinda citrifolia and Rubia tinctorum. Plant Cell Tissue Organ Cult 124:319–330. https://doi.org/10.1007/s11240-015-0896-y
Quevedo C, Perassolo M, Alechine E et al (2010) Increasing anthraquinone production by overexpression of 1-deoxy-d-xylulose-5-phosphate synthase in transgenic cell suspension cultures of Morinda citrifolia. Biotechnol Lett 32:997–1003. https://doi.org/10.1007/s10529-010-0250-8
Sahu J, Mohan Koley K, Sahu B (2017) Effect of supplementing Cassia tora leaf extract on immunological and haematological parameters of broiler birds. J Anim Res 2:293–298. https://doi.org/10.5958/2277-940X.2017.00042.0
Shah MR, George IA (2019) Increased biomass and pigment production from Cassia alata L. callus cultures and their potential as a textile dye. Ind Crops Prod 128:346–353. https://doi.org/10.1016/J.INDCROP.2018.11.018
Shahid M, Wertz J, Degano I et al (2019) Analytical methods for determination of anthraquinone dyes in historical textiles: a review. Anal Chim Acta 1083:58–87. https://doi.org/10.1016/J.ACA.2019.07.009
Shkryl YN, Veremeichik GN, Makhazen DS et al (2016) Increase of anthraquinone content in Rubia cordifolia cells transformed by native and constitutively active forms of the AtCPK1 gene. Plant Cell Rep 35:1907–1916. https://doi.org/10.1007/s00299-016-2005-z
Siva R, Mayes S, Behera SK, Rajasekaran C (2012) Anthraquinones dye production using root cultures of Oldenlandia umbellata L. Ind Crops Prod 37:415–419. https://doi.org/10.1016/J.INDCROP.2011.12.027
Sreelakshmi V, Abraham A (2016) Anthraquinones and flavonoids of Cassia tora leaves ameliorate sodium selenite induced cataractogenesis in neonatal rats. Food Funct 7:1087–1095. https://doi.org/10.1039/c5fo00905g
Thakur M, Bhattacharya S, Khosla PK, Puri S (2019) Improving production of plant secondary metabolites through biotic and abiotic elicitation. J Appl Res Med Aromat Plants 12:1–12. https://doi.org/10.1016/J.JARMAP.2018.11.004
Tilwari A, Chauhan D, Sharma R, Singh R (2016) Assessment of Genetic Variations Among Medicinal Plant Cassia tora from different geographic regions of central india using RAPD markers. Med Aromat Plants, https://doi.org/10.4172/2167-0412.1000276
Tsimogiannis D, Oreopoulou V (2019) Classification of phenolic compounds in plants. In: Watson RR (ed) Polyphenols in plants, second edition. Academic Press, London, pp 263–284
Vats S, Kamal R (2014) Identification of flavonoids and antioxidant potential of Cassia tora L. Am J Drug Discov Dev 4:50–57. https://doi.org/10.3923/ajdd.2014.50.57
Verma NR, Nandani D, Batra A (2013) Isolation and identification of quercetin and emodin from Cassia tora L. An Int J Ann Phytomed 2:96–104
Wagner HB, S, Bladt (2001) Plant drug analysis: a thin layer chromatography atlas, 2nd edn. Springer, Dordrecht
Wink M (2013) Evolution of secondary metabolites in legumes (Fabaceae). S Afr J Bot 89:164–175. https://doi.org/10.1016/J.SAJB.2013.06.006
Xu LY, Li X, Cui YQ, et al (2018) Antibacterial activity of anthraquinone from cassia seed on spiced pig head. In: Proceedings of the IOP Conference Series: Materials Science and Engineering. IOP Publishing
Zhou X, Song B, Jin L et al (2006) Isolation and inhibitory activity against ERK Phosphorylation of hydroxyanthraquinones from rhubarb. Bioorg Med Chem Lett 16:563–568. https://doi.org/10.1016/j.bmcl.2005.10.047
Author information
Authors and Affiliations
Contributions
All authors contributed to the conception and design of the study. Experiments and data collection was carried out by M.S. The data was analysed by M.S. and I.G. The first draft of the manuscript was written by M.S. and I.G. commented on previous versions of the manuscript. Both the authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Danny Geelen.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
11240_2020_1913_MOESM1_ESM.tif
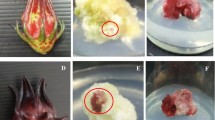
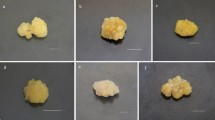
Supplementary file 1 Fig. 1 Seeds, callus and extract of Cassia tora. (a) Cassia tora seeds used for germination of seedlings (b) 44th Day callus obtained from 51BT medium. (c) Methanolic extract of Cassia tora callus (TIF 8302 kb)
11240_2020_1913_MOESM2_ESM.tif
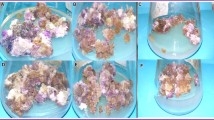
Supplementary file 2 Fig. 2 Cassia tora callus types (a) Sporadic organogenesis of shoots in hypocotyl explants treated with Kn (b) Root organogenesis in hypocotyl explants treated with IAA (c) Roots with friable yellow callus in root explants treated with TDZ (d) Green brown callus in cotyledonary explant treated with TDZ (e) Compact white callus in hypocotyl treated with 2-iP (f) White green brown callus in hypocotyl explants treated with BAP(TIF 8295 kb)
Rights and permissions
About this article
Cite this article
Shah, M., George, I.A. Pigment elicitation and sun protection factor of callus induced from Cassia tora seedling explants. Plant Cell Tiss Organ Cult 143, 201–210 (2020). https://doi.org/10.1007/s11240-020-01913-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-020-01913-3