Abstract
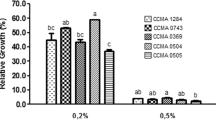
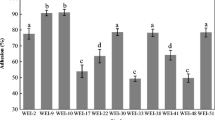
In this study, the presence of plasmids responsible for carbohydrate fermentation and antibiotic resistance and the stability of these plasmids in artificial gastric juice were investigated in 20 Lactobacillus plantarum strains with probiotic properties. Plasmid curing was performed with novobiocin, acriflavine and elevated incubation temperature to identify plasmids encoded with carbohydrate fermentation and antibiotic resistance genes and to compare them with artificial gastric juice. Plasmid profiling of the strains revealed that 100% of the strains were harbouring plasmids in varying sizes and numbers. The plasmid number of the potential probiotic strains ranged between 1 and 4, and the plasmid size ranged between 5.779 and 16.138 kb. The potential probiotic strains could not survive in the artificial gastric juice at pH 2.0. Although the strains maintained their viability in an artificial gastric juice at pH 2.5 and 3.0, and their derivatives lost their plasmids at a high rate (100%). Similarly, high levels of cured derivatives were obtained with 8 µg/mL novobiocin and 100 µg/mL acriflavine applications, and 24 h incubation at 43 °C. All the experiments were also performed to compare with two L. plantarum-type strains containing plasmids responsible for tetracycline and tetracycline + erythromycin resistances. Artificial gastric juice and other plasmid curing treatments caused a high-frequency loss in the antibiotic resistances of type strains. Determining plasmid stability in artificial gastric juice is a novel approach. Plasmid stability in the gastrointestinal tract is important for maintaining the plasmid-encoded probiotic properties.


Similar content being viewed by others
References
Abriouel H, Pérez Montoro B, de la Fuente Ordoñez JJ, Lavilla Lerma L, Knapp CW, Benomar N (2019) New insights into the role of plasmids from probiotic Lactobacillus pentosus MP-10 in Aloreña table olive brine fermentation. Sci Rep 9:1–19. https://doi.org/10.1038/s41598-019-47384-1
Aloǧlu H, Öner Z (2006) Assimilation of cholesterol in broth, cream, and butter by probiotic bacteria. Eur J Lipid Sci Technol 108:709–713. https://doi.org/10.1002/ejlt.200600137
Anonymous (2020) National Centre for Biotechnology Information. https://www.ncbi.nlm.nih.gov/genome/plasmids/1108
Balgir PP, Kaur B, Kaur T, Daroch N, Kaur G (2013) In vitro and in vivo survival and colonic adhesion of pediococcus acidilactici MTCC5101 in human gut. Biomed Res Int 2013:583850. https://doi.org/10.1155/2013/583850
Başyiğit G (2004) Usage possibilities of some lactic acid bacteria as probiotic. Süleyman Demirel University, Graduate School of Natural and Applied Sciences, MSc. Thesis, Isparta
Başyiğit G (2009) Genotyping identification of some Lactobacillus sp. strains and determination of their phages resistances. Süleyman Demirel University, Graduate School of Natural and Applied Sciences, PhD. Thesis, Isparta
Başyiğit Kiliç G, Karahan AG (2010) Identification of lactic acid bacteria isolated from the fecal samples of healthy humans and patients with dyspepsia, and determination of their pH, bile, and antibiotic tolerance properties. J Mol Microbiol Biotechnol 18:220–229. https://doi.org/10.1159/000319597
Başyiğit Kiliç G, Akpinar D (2013) The effects of different levels of β-glucan on yoghurt manufactured with Lactobacillus plantarum strains as adjunct culture. J Food Agric Environ 11:281–287. https://doi.org/10.1234/4.2013.3845
Başyiğit Kılıç G, Kuleaşan H, Sömer VF, Akpınar D (2013) Determining potential probiotic properties of human originated Lactobacillus plantarum strains. Biotechnol Bioprocess Eng 18:479–485. https://doi.org/10.1007/s12257-012-0785-8
Blaabjerg S, Artzi DM, Aabenhus R (2017) Probiotics for the prevention of antibiotic-associated diarrhea in outpatients—a systematic review and meta-analysis. Antibiotics. https://doi.org/10.3390/antibiotics6040021
Boranbayeva T, Karahan AG, Tulemissova Z, Myktybayeva R, Özkaya S (2020) Properties of a new probiotic candidate and Lactobacterin-TK2 against diarrhea in calves. Probiotics Antimicrob Proteins. https://doi.org/10.1007/s12602-020-09649-4
Caro L, Churchward G, Chandler M (1984) Study of plasmid replication in vivo, methods in microbiology. Academic Press, New York
Chin CS, Abdullah N, Tan WS, Ho YW (2005) Plasmid profiling and curing of Lactobacillus strains isolated from the gastrointestinal tract of chicken. J Microbiol 43:251–256
CLSI (2016) M100S: Performance Standards for Antimicrobial Susceptibility Testing, M100S, 26th Edition
Danielsen M, Wind A (2003) Susceptibility of Lactobacillus spp. to antimicrobial agents. Int J Food Microbiol 82:1–11. https://doi.org/10.1016/S0168-1605(02)00254-4
EFSA (2012) Guidance on the assessment of bacterial susceptibility to antimicrobials of human and veterinary importance. EFSA J 10:1–10. https://doi.org/10.2903/j.efsa.2012.2740
Egervärn M, Roos S, Lindmark H (2009) Identification and characterization of antibiotic resistance genes in Lactobacillus reuteri and Lactobacillus plantarum. J Appl Microbiol 107:1658–1668. https://doi.org/10.1111/j.1365-2672.2009.04352.x
El-Deeb N, Sharaf MM, El-Adawi H (2015) Antibacterial and plasmid curing activity of lactic acid bacteria against multidrug resistant bacteria strains. Int J Pharmacol 11:114–121. https://doi.org/10.3923/ijp.2015.114.121
Feng C, Zhang F, Wang B, Gao J, Wang Y, Shao Y (2019) Evaluation of kanamycin and neomycin resistance in Lactobacillus plantarum using experimental evolution and whole-genome sequencing. Food Control 98:262–267. https://doi.org/10.1016/j.foodcont.2018.11.030
Fernández M, Margolles A, Suárez JE, Mayo B (1999) Duplication of the β-galactosidase gene in some Lactobacillus plantarum strains. Int J Food Microbiol 48:113–123. https://doi.org/10.1016/S0168-1605(99)00031-8
Gevers D, Huys G, Devlieghere F, Uyttendaele M, Debevere J, Swings J (2000) Isolation and identification of tetracycline resistant lactic acid bacteria from pre-packed sliced meat products. Syst Appl Microbiol 23:279–284. https://doi.org/10.1016/S0723-2020(00)80015-6
Gevers D, Danielsen M, Huys G, Swings J (2003a) Molecular characterization of tet(M) genes in Lactobacillus isolates from different types of fermented dry sausage. Appl Environ Microbiol 69:1270–1275. https://doi.org/10.1128/AEM.69.2.1270-1275.2003
Gevers D, Huys G, Swings J (2003b) In vitro conjugal transfer of tetracycline resistance from Lactobacillus isolates to other Gram-positive bacteria. FEMS Microbiol Lett 225:125–130. https://doi.org/10.1016/S0378-1097(03)00505-6
Jacobsen L, Wilcks A, Hammer K, Huys G, Gevers D, Andersen SR (2007) Horizontal transfer of tet(M) and erm(B) resistance plasmids from food strains of Lactobacillus plantarum to Enterococcus faecalis JH2-2 in the gastrointestinal tract of gnotobiotic rats. FEMS Microbiol Ecol 59:158–166. https://doi.org/10.1111/j.1574-6941.2006.00212.x
Jamuna M, Kolanchiammal R, Jeevaratnam K (2010) Plasmid-associated bacteriocin production in Lactobacillus strains isolated from some traditional fermented foods. Glob J Biotechnol Biochem 5:175–181
Jewell B, Collins-Thompson DL (1989) Characterization of chloramphenicol resistance in Lactobacillus plantarum caTC2. Curr Microbiol 19:343–346. https://doi.org/10.1007/BF01570879
Kandler O, Weiss N (1986) Regular, non-sporing gram-positive rods. In: Sneath HA, Mair NS, Sharpe ME, Holt JG (eds) Bergey’s manual of systematic bacteriology. Williams and Wilkins, Baltimore, pp 1208–1234
Karahan N, Işler M, Koyu A, Karahan AG, Başyiǧit Kiliç G, Çiriş IM, Sütçü R, Onaran I, Çam H, Keskin M (2012) Effects of probiotics on methionine cholinedeficient diet-induced steatohepatitis in rats. Turkish J Gastroenterol 23:110–121. https://doi.org/10.4318/tjg.2012.0330
Karthikeyan V, Santosh SW (2010) Comparing the efficacy of plasmid curing agents in Lactobacillus acidophilus. Benef Microbes 1:155–158. https://doi.org/10.3920/BM2009.0038
Mathur S, Singh R (2005) Antibiotic resistance in food lactic acid bacteria—a review. Int J Food Microbiol 105:281–295. https://doi.org/10.1016/J.IJFOODMICRO.2005.03.008
Mayo B, Hardisson C, Brana AF (1989) Selected characteristics of several strains of Lactobacillus plantarum. Microbioigia 5:105–122
McHugh GL, Swartz MN (1977) Elimination of plasmids from several bacterial species by novobiocin. Antimicrob Agents Chemother 12:423–426. https://doi.org/10.1128/AAC.12.3.423
McKay LL, Baldwin KA, Zottola EA (1972) Loss of lactose metabolism in lactic streptococci. Appl Microbiol 23:1090–1096. https://doi.org/10.1128/aem.23.6.1090-1096.1972
Mohite JA, Soman YP, Mujumdar SS (2016) Herbal extraction and plasmid curing (curing of multiple drug resistance in bacteria using herbal extracts). World J Pharm Res 5:1554–1565
Sağlam H (2013) Tanımlanmış Lactobacillus plantarum suşlarının plazmit profilleri ve bunların bazı özelliklerinin belirlenmesi. Süleyman Demirel Üniversitesi, Fen Bilimleri Enstitüsü, Gıda Mühendisliği Ana Bilimdalı, Isparta
Sağlam H, Karahan AG (2017) Plasmids in lactic acid bacteria and their properties. Kafkas Univ. Inst Nat Appl Sci J 10:252–285
Salminen S, Ouwehand A, Benno Y, Lee YK (1999) Probiotics: how should they be defined? Trends Food Sci Technol. https://doi.org/10.1016/S0924-2244(99)00027-8
Senol A, Isler M, Karahan AG, Kilic GB, Kuleasan H, Kaya S, Keskin M, Goren I, Saritas U, Aridogan BC, Delibas N (2011) Preventive effect of probiotics and α-tocopherol on ethanol-induced gastric mucosal injury in rats. J Med Food 14:173–179. https://doi.org/10.1089/jmf.2010.0040
Şenol A, Işler M, Karahan AG, Başyiğit Kiliç G, Kuleaşan H, Gören I, Saritaş Ü, Kaya S, Çiriş M, Aktürk O, Aridoǧan BC, Demirin H, Çakmakçi LM (2011) Effect of probiotics on aspirin-induced gastric mucosal lesions. Turkish J Gastroenterol. https://doi.org/10.4318/tjg.2011.0151
Shimizu-Kadota M (1987) Properties of lactose plasmid pLY101 in Lactobacillus casei. Appl Environ Microbiol 53:2987–2991. https://doi.org/10.1128/aem.53.12.2987-2991.1987
Spengler G, Molnar A, Schelz Z, Amaral L, Sharples D, Molnar J (2006) The mechanism of plasmid curing in bacteria. Curr Drug Targets 7:823–841. https://doi.org/10.2174/138945006777709601
Trevors JT (1986) Plasmid curing in bacteria. FEMS Microbiol Lett 32:149–157. https://doi.org/10.1016/0378-1097(86)90286-7
van den Nieuwboer M, van Hemert S, Claassen E, de Vos WM (2016) Lactobacillus plantarum WCFS1 and its host interaction: a dozen years after the genome. Microb Biotechnol 9:452–465. https://doi.org/10.1111/1751-7915.12368
Wang T, Lee BH (1997) Plasmids in Lactobacillus. Crit Rev Biotechnol 17:227–272. https://doi.org/10.3109/07388559709146615
Acknowledgements
This research was funded by Süleyman Demirel University Scientific Research Projects Coordination Unit with Project No. 1876-D-09.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Erko Stackebrandt.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sağlam, H., Karahan, A.G. Plasmid stability of potential probiotic Lactobacillus plantarum strains in artificial gastric juice, at elevated temperature, and in the presence of novobiocin and acriflavine. Arch Microbiol 203, 183–191 (2021). https://doi.org/10.1007/s00203-020-02017-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00203-020-02017-4