Abstract
Key message
Host mediated silencing of COM1 gene of Colletotrichum gloeosporioides disables appressorial differentiation and effectively prevents the development of Anthracnose disease in chilli and tomato.
Abstract
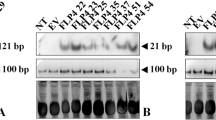
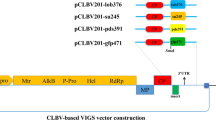
Anthracnose disease is caused by the ascomycetes fungal species Colletotrichum, which is responsible for heavy yield losses in chilli and tomato worldwide. Conventionally, harmful pesticides are used to contain anthracnose disease with limited success. In this study, we assessed the potential of Host-Induced Gene Silencing (HIGS) approach to target the Colletotrichum gloeosporioides COM1 (CgCOM1) developmental gene involved in the fungal conidial and appressorium formation, to restrict fungal infection in chilli and tomato fruits. For this study, we have developed stable transgenic lines of chilli and tomato expressing CgCOM1-RNAi construct employing Agrobacterium-mediated transformation. Transgenic plants were characterized by molecular and gene expression analyses. Production of specific CgCOM1 siRNA in transgenic chilli and tomato RNAi lines was confirmed by stem-loop RT-PCR. Fungal challenge assays on leaves and fruits showed that the transgenic lines were resistant to anthracnose disease-causing C. gloeosporioides in comparison to wild type and empty-vector control plants. RT-qPCR analyses in transgenic lines revealed extremely low abundance of CgCOM1 transcripts in the C. gloeosporioides infected tissues, indicating near complete silencing of CgCOM1 gene expression in the pathogen. Microscopic examination of the Cg-challenged leaves of chilli-CgCOM1i lines revealed highly suppressed conidial germination, germ tube development, appressoria formation and mycelial growth of C. gloeosporioides, resulting in reduced infection of plant tissues. These results demonstrated highly efficient use of HIGS in silencing the expression of essential fungal developmental genes to inhibit the growth of pathogenic fungi, thus providing a highly precise approach to arrest the spread of disease.






Similar content being viewed by others
References
Alkan N, Friedlander G, Ment D, Prusky D, Fluhr R (2015) Simultaneous transcriptome analysis of Colletotrichum gloeosporioides and tomato fruit pathosystem reveals novel fungal pathogenicity and fruit defense strategies. New Phytol 205:801–815. https://doi.org/10.1111/nph.13087
Barksdale TH (1972) Resistance in tomato to six anthracnose fungi. Phytopathology 62:660–663
Baulcombe D (2004) RNA silencing in plants. Nature 431:356–363
Baum JA et al (2007) Control of coleopteran insect pests through RNA interference. Nat Biotechnol 25:1322–1326
Bhadauria V, Wang L-X, Peng Y-L (2010) Proteomic changes associated with deletion of the Magnaporthe oryzae conidial morphology regulating gene COM1. Biol Direct 5:61
Bharti P, Jyoti P, Kapoor P, Sharma V, Shanmugam V, Yadav SK (2017) Host-induced silencing of pathogenicity genes enhances resistance to Fusarium oxysporum wilt in tomato. Mol Biotechnol 59:343–352
Bosland PW, Votava EJ (2012) Peppers: vegetable and spice Capsicums. CABI, Wallingford, pp 1–230
Cai Q et al (2018) Plants send small RNAs in extracellular vesicles to fungal pathogen to silence virulence genes. Science 360:1126–1129. https://doi.org/10.1126/science.aar4142
Chen C et al (2005) Real-time quantification of microRNAs by stem-loop RT-PCR. Nucleic Acids Res 33:e179–e179. https://doi.org/10.1093/nar/gni178
Chen W et al (2016) Host-induced silencing of Fusarium culmorum genes protects wheat from infection. J Exp Bot 67:4979–4991. https://doi.org/10.1093/jxb/erw263
Cheng W et al (2015) Host-induced gene silencing of an essential chitin synthase gene confers durable resistance to Fusarium head blight and seedling blight in wheat. Plant Biotechnol J 13:1335–1345. https://doi.org/10.1111/pbi.12352
De Silva DD, Crous PW, Ades PK, Hyde KD, Taylor PWJ (2017) Life styles of Colletotrichum species and implications for plant biosecurity. Fungal Biol Rev 1–14:155–168. https://doi.org/10.1016/j.fbr.2017.05.001
Doyle JJ, Doyle JL (1990) Isolation of plant DNA from fresh tissue. Focus 12:13–15
Dubreuil G, Magliano M, Deleury E, Abad P, Rosso MN (2007) Transcriptome analysis of root-knot nematode functions induced in the early stages of parasitism. New Phytol 176:426–436. https://doi.org/10.1111/j.1469-8137.2007.02181.x
Eloy YRG, Vasconcelos IM, Barreto ALH, Freire-Filho FR, Oliveira JTA (2015) H2O2 plays an important role in the lifestyle of Colletotrichum gloeosporioides during interaction with cowpea (Vigna unguiculata L. Walp.). Fungal Biol 119:747–757. https://doi.org/10.1016/j.funbio.2015.05.001
Ghag SB, Shekhawat UKS, Ganapathi TR (2014) Host-induced post-transcriptional hairpin RNA-mediated gene silencing of vital fungal genes confers efficient resistance against Fusarium wilt in banana. Plant Biotechnol J 12:541–553. https://doi.org/10.1111/pbi.12158
Govindarajulu M, Epstein L, Wroblewski T, Michelmore RW (2015) Host-induced gene silencing inhibits the biotrophic pathogen causing downy mildew of lettuce. Plant Biotechnol J 13:875–883. https://doi.org/10.1111/pbi.12307
Hofgen R, Willmitzer L (1988) Storage of competent cells for Agrobacterium transformation. Nucleic Acids Res 16:9877
Huang G, Allen R, Davis EL, Baum TJ, Hussey RS (2006) Engineering broad root-knot resistance in transgenic plants by RNAi silencing of a conserved and essential root-knot nematode parasitism gene. Proc Natl Acad Sci USA 103:14302–14306. doi:https://doi.org/10.1073/pnas.0604698103
Hyde KD, Cai L, McKenzie EHC, Yang YL, Zhang JZ, Prihastuti H (2009) Colletotrichum: a catalogue of confusion Fungal Diversity 39:1–7
Jahan SN, Asman AK, Corcoran P, Fogelqvist J, Vetukuri RR, Dixelius C (2015) Plant-mediated gene silencing restricts growth of the potato late blight pathogen Phytophthora infestans. J Exp Bot 66:2785–2794. https://doi.org/10.1093/jxb/erv094
Jeger MJ, Bailey JA (1992) Colletotrichum: biology, pathology and control. Wallingford
Khatri M, Rajam MV (2007) Targeting polyamines of Aspergillus nidulans by siRNA specific to fungal ornithine decarboxylase gene. Med Mycol 45:211–220
Kim SH, Yoon JB, Do JW, Park HG (2008) A major recessive gene associated with anthracnose resistance to Colletotrichum capsici in chili pepper (Capsicum annuum L.). Breed Sci 58:137–141. https://doi.org/10.1270/jsbbs.58.137
Koch A, Kumar N, Weber L, Keller H, Imani J, Kogel K-H (2013) Host-induced gene silencing of cytochrome P450 lanosterol C14α-demethylase-encoding genes confers strong resistance to Fusarium species. Proc Natl Acad Sci USA 110:19324–19329. https://doi.org/10.1073/pnas.1306373110
Koch A et al (2020) Host-induced gene silencing involves transfer of dsRNA-derived siRNA via extracellular vesicles. bioRxiv. https://doi.org/10.1101/2020.02.12.945154
Li Y (2013) Anthracnose of tomato. The Connecticut Agricultural Experiment Station
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2–∆∆Ct method. Methods 25:402–408. doi:https://doi.org/10.1006/meth.2001.1262
Madhulatha P, Pandey R, Hazarika P, Rajam MV (2007) High transformation frequency in Agrobacterium mediated genetic transformation of tomato by using polyamines and maltose in shoot regeneration medium. Physiol Mol Biol Plants 13:191–198
Mahto BK, Sharma P, Rajam MV, Reddy PM, Dhar-Ray S (2018) An efficient method for Agrobacterium-mediated genetic transformation of chilli pepper (Capsicum annuum L.). Indian J Plant Physiol 23:573–581. https://doi.org/10.1007/s40502-018-0389-1
Mamta R, Rajam MV (2016) Targeting chitinase gene of Helicoverpa armigera by host-induced RNA interference confers insect resistance in tobacco and tomato. Plant Mol Biol 90:281–292. https://doi.org/10.1007/s11103-015-0414-y
Mao Y-B et al (2007) Silencing a cotton bollworm P450 monooxygenase gene by plant-mediated RNAi impairs larval tolerance of gossypol. Nat Biotechnol 25:1307–1313
Moraes SRG, Tanaka FAO, Massola J, Nelson SMJ (2013) Histopathology of Colletotrichum gloeosporioides on guava fruits (Psidium guajava L.). Rev Brasil Fruticult 35:657–664
Nelson SC (2008) Mango anthracnose (Colletotrichum gloeosporiodes). Plant Dis 48:1–9
Nowara D et al (2010) HIGS: Host-induced gene silencing in the obligate biotrophic fungal pathogen Blumeria graminis. Plant Cell 22:3130–3141. https://doi.org/10.1105/tpc.110.077040
O’Connell R, Herbert C, Sreenivasaprasad S, Khatib M, Esquerre-Tugaye M, Dumas B (2004) A novel Arabidopsis-Colletotrichum pathosystem for the molecular dissection of plant-fungal interactions. Mol Plant Microbe Interact 17:272–282. https://doi.org/10.1094/mpmi.2004.17.3.272
O’Connell RJ et al (2012) Lifestyle transitions in plant pathogenic Colletotrichum fungi deciphered by genome and transcriptome analyses. Nat Genet 44:1060–1065
Pakdeevaraporn P, Wasee S, Taylor PWJ, Mongkolporn O (2005) Inheritance of resistance to anthracnose caused by Colletotrichum capsici in Capsicum. Plant Breed 124:206–208. https://doi.org/10.1111/j.1439-0523.2004.01065.x
Panwar V, Jordan M, McCallum B, Bakkeren G (2017) Host-induced silencing of essential genes in Puccinia triticina through transgenic expression of RNAi sequences reduces severity of leaf rust infection in wheat. Plant Biotechnol J 16:1013–1023. https://doi.org/10.1111/pbi.12845
Panwar V, McCallum B, Bakkeren G (2013) Endogenous silencing of Puccinia triticina pathogenicity genes through in planta-expressed sequences leads to the suppression of rust diseases on wheat. Plant J 73:521–532. https://doi.org/10.1111/tpj.12047
Peng J, Xia Z, Chen L, Shi M, Pu J, Guo J, Fan Z (2014) Rapid and efficient isolation of high-quality small RNAs from recalcitrant plant species rich in polyphenols and polysaccharides. PLOS ONE 9:e95687. https://doi.org/10.1371/journal.pone.0095687
Phoulivong S, Cai L, Chen H, McKenzie EHC, Abdelsalam K, Chukeatirote E, Hyde KD (2010) Colletotrichum gloeosporioides is not a common pathogen on tropical fruits. Fungal Divers 44:33–43. https://doi.org/10.1007/s13225-010-0046-0
Prathibha VH, Rao AM, Ramesh S, Nanda C (2013) Estimation of fruit quality parameters in anthracnose infected chilli fruits. Int J Agric Food Sci Technol 4:57–60
Raruang Y et al (2020) Host induced gene silencing targeting Aspergillus flavus aflM reduced aflatoxin contamination in transgenic maize under field conditions. Front Microbiol. https://doi.org/10.3389/fmicb.2020.00754
Reynolds A, Leake D, Boese Q, Scaringe S, Marshall WS, Khvorova A (2004) Rational siRNA design for RNA interference. Nat Biotechnol 22:326
Seo H, Park S, Oh B, Back K, Han O, Kim J, Kim YS (2014) Overexpression of a defensin enhances resistance to a fruit-specific anthracnose fungus in pepper. PLOS ONE 9:e97936
Singh N, Mukherjee SK, Rajam MV (2020) Silencing of the ornithine decarboxylase gene of Fusarium oxysporum f. sp. lycopersici by host-induced RNAi confers resistance to Fusarium wilt in tomato. Plant Mol Biol Rep. https://doi.org/10.1007/s11105-020-01205-2
Song Y, Thomma BPHJ (2018) Host-induced gene silencing compromises Verticillium wilt in tomato and Arabidopsis. Mol Plant Pathol 19:77–89. https://doi.org/10.1111/mpp.12500
Takahara H, Dolf A, Endl E, O’Connell R (2009) Flow cytometric purification of Colletotrichum higginsianum biotrophic hyphae from Arabidopsis leaves for stage-specific transcriptome analysis. Plant J 59:672–683. https://doi.org/10.1111/j.1365-313X.2009.03896.x
Thakare D, Zhang J, Wing RA, Cotty PJ, Schmidt MA (2017) Aflatoxin-free transgenic maize using host-induced gene silencing. Sci Adv 3:e1602382. https://doi.org/10.1126/sciadv.1602382
Than PP, Prihastuti H, Phoulivong S, Taylor PWJ, Hyde KD (2008) Chilli anthracnose disease caused by Colletotrichum species. J Zhejiang Univ Sci B 9:764–778. https://doi.org/10.1631/jzus.B0860007
Varkonyi-Gasic E, Wu R, Wood M, Walton EF, Hellens RP (2007) Protocol: a highly sensitive RT-PCR method for detection and quantification of microRNAs. Plant Methods 3:12–12. https://doi.org/10.1186/1746-4811-3-12
Vaucheret H, Beclin C, Fagard M (2001) Post-transcriptional gene silencing in plants. J Cell Sci 114:3083–3091
Voorrips RE, Finkers R, Sanjaya L, Groenwold R (2004) QTL mapping of anthracnose (Colletotrichum spp.) resistance in a cross between Capsicum annuum and C. chinense. Theoret Appl Genet 109:1275–1282. https://doi.org/10.1007/s00122-004-1738-1
Yang J, Zhao X, Sun J, Kang Z, Ding S, Xu J-R, Peng Y-L (2010) A novel protein Com1 is required for normal conidium morphology and full virulence in Magnaporthe oryzae. Mol Plant Microbe Interact 23:112–123. doi:https://doi.org/10.1094/mpmi-23-1-0112
Yin C, Jurgenson JE, Hulbert SH (2011) Development of a host-induced RNAi system in the wheat stripe rust fungus Puccinia striiformis f. sp. tritici. Mol Plant Microbe Interact 24:554–561. https://doi.org/10.1094/mpmi-10-10-0229
Yogindran S, Rajam MV (2015) RNAi for crop improvement. In: Bahadur B, Venkat Rajam M, Sahijram L, Krishnamurthy KV (eds) In Plant biology and biotechnology: Volume II: plant genomics and biotechnology. Springer, New Delhi. pp 623–637. https://doi.org/10.1007/978-81-322-2283-5_31
Zhou B, Bailey A, Niblett CL, Qu R (2016) Control of brown patch (Rhizoctonia solani) in tall fescue (Festuca arundinacea Schreb.) by host induced gene silencing. Plant Cell Rep 35:791–802. doi:https://doi.org/10.1007/s00299-015-1921-7
Acknowledgements
The research work was financed by the Department of Biotechnology (Grant No. BT/PR5399/AGR/36/722/2012), Government of India, to SD and MVR. BKM gratefully acknowledges Department of Science and Technology-INSPIRE (DST-INSPIRE-IF120813), Government of India, for providing fellowship.
Author information
Authors and Affiliations
Contributions
BKM: Conducted the experiments and wrote manuscript. AS: RNA isolation and RT-PCR. MP: Stem-loop RT-PCR. MVR: RNAi vector. SD-R: Conceptualization, Experimental planning. PMR: Designing of experiments and wrote the manuscript.
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mahto, B.K., Singh, A., Pareek, M. et al. Host-induced silencing of the Colletotrichum gloeosporioides conidial morphology 1 gene (CgCOM1) confers resistance against Anthracnose disease in chilli and tomato. Plant Mol Biol 104, 381–395 (2020). https://doi.org/10.1007/s11103-020-01046-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11103-020-01046-3